Q: What main type of forces must be disrupted between (CH3)2CO molecules when I2 dissolves? hydrogen bo...
A: Intermolecular forces are basically the type of forces that help in binding the different atoms of a...
Q: Use alkynes as starting materials and intermediates in one-step and multistep syntheses.
A: Hydrocarbons are classified into different group – alkanes, alkene and alkynes. The compounds contai...
Q: Using the “Like dissolves like” principle of solubility, assign each solute to the solvent in which ...
A: The solubility of a compound depends on the nature of the solute and the solvent. If the nature of b...
Q: 2. What is the stere Br Hy Br and H. CI CI
A: Stereoisomerism is the kind of isomerism, in which molecules have same molecular formula, but differ...
Q: A 7.298 g sample of copper reacts with oxygen, forming a copper oxide. The final mass of the copper ...
A: The formula of a compound can be determined by using the following steps Find the number of moles o...
Q: Hi there! Can someone please help me with this question? Thank you so much!
A: In the given compound label all lone pair electron and pi bond electron
Q: How do I express each number to significant figures or scientific notation ?
A: a. Number of Significant Figures: 6The Significant Figures are 5 5 0 0 8 8 b. Number of Significant ...
Q: Determine order of reaction for reversible reactions?
A: Determine the order of reaction for reversible reactions?
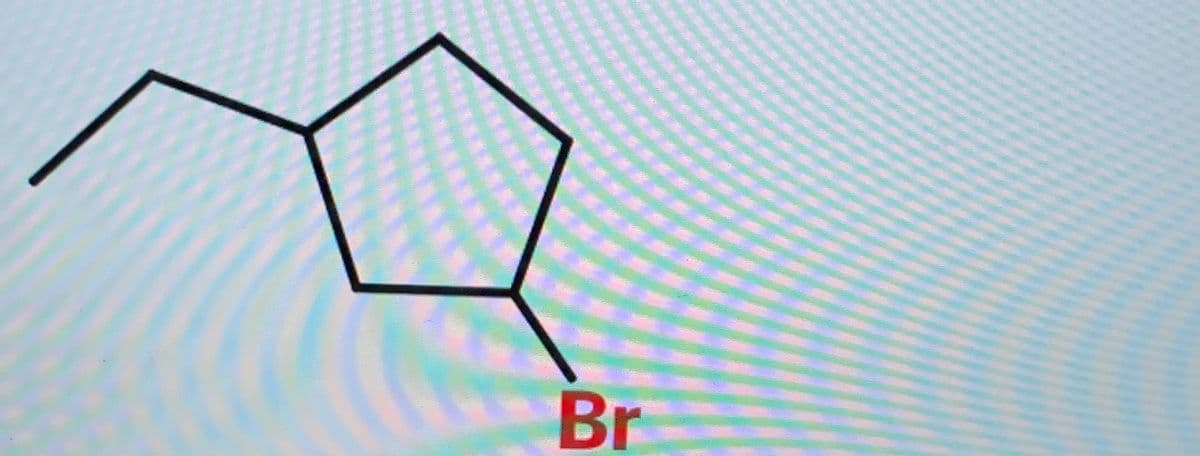
Q: what would be the proper name for this molecule?
A: To write the name (IUPAC) of given compound we have to follow the following steps :- (1) Identify t...
Q: Water produces Hydrogen gas and Oxygen gas when subjected to electrolysis. A. Convert the given pro...
A: Electrolysis is the process in which electric current is passed through any substance to effect a ch...
Q: Balance the equations according to the half-reaction method. Zn(s) + NO3−(aq) → Zn2+(aq) + N2(g) (in...
A:
Q: Consider Se, Sc and O elements. Arrange them in order of increasing ionization energy. Briefly expla...
A: Ionization energy of elements increases from left to right of the period in the periodic table due t...
Q: What is the mole fraction of solute in a 3.77 m aqueous solution?
A:
Q: An ionic compound crystallizes with 1 ion at each corner, 1 ion at each face, and 8 ions in the body...
A:
Q: How do I figure this out?
A:
Q: What are all the reagents needed to complete 1)step 1 & 2)step 2 of Methamphetamine synthesis st...
A:
Q: 3-ethyl-2,2,4-trimethylpentane
A: A carbon atom (in a compound) to which four different types of substituents (atoms or groups of atom...
Q: Cholesterol is an important compound in the body, but in excess has been implicated as a cause of he...
A: Given data : Mass of cholestrol = 4.82 gms Mass of Benzene = 112 gms Tfreez...
Q: 2. Draw the products for the following reaction conditions using the starting material provided. 1. ...
A: The given compound is an alpha beta-unsaturated ketone. The nucleophile can give the addition reacti...
Q: A metal has atoms with a radius of 156 pm. It crystallizes in a body-entered cubic unit cell. It has...
A: We know the expression of density for solid is, d= (Z × M)/a3.NA d= density of solid=7.74 g/cm3 Z=nu...
Q: Construct the expression for Kc for the following reaction. 2 Cu+(aq) + Zn(s) <---> 2 Cu(s) + ...
A: Kc:- It may be defined as the equilibrium constant when the concentration of reactants and products ...
Q: A metal object has a mass of 8.370 g. When it was placed in a graduated cylinder containing 22.0 mL ...
A: Density = mass/volume
Q: What is the specific heat capacity of K if 1030 J were required to heat 48.15 g of it from 25.0A°C t...
A: Given Amount of heat, (q) = 1030J Mass of K, (m) = 48.15 g Initial temperature, (T1) = 25 •C Final ...
Q: The solution's osmotic
A: Osmotic pressure is one of the four colligative properties of the solutions. Osmosis can be de\fined...
Q: For the following reaction, draw a complete reaction mechanism using curvy arrows being sure to show...
A: Solution Two sensible sources of binary compound-like reactivity ar the complicated metal hydrides a...
Q: Chemistry Question
A: Given equation: 2C +2H2 →C2H4 ∆G=∆Go+RTlnKeq here ∆Go=∆Ho-T∆S0 ∆Go=52.47 x 1000 -298(-53.5) ...
Q: 3.5 mole of NH3 are ____ grams I don’t understand the formula to answer this question
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: 4. A sulfuric acid solution containing 571.6 g H2SO4 per liter of solution has a density of 1.329 g/...
A: Mass percentage of a solution/mixture is the ration of mass of solute/element/compound of interest t...
Q: The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an acti...
A:
Q: Which of the following statements is FALSE? OA Bromobenzene is less reactive than benzene in an elec...
A: All the electron donating substituents on the ring activates the ring towards electrophilic aromatic...
Q: Round 3.075 to 3 significant figures and put in scientific notation.
A: Rounding significant figures :- * If the rightmost digit to be removed is greater that five then pre...
Q: The combustion of fuel in your car engine requires oxygen gas, which is supplied as air (21% oxygen ...
A: Given data : Volume of air = 2.71 L Pressure = 1 atm Temperature = 25°C = 298 K Ac...
Q: Draw the structure corresponding to the following name. acetic formic anhydride
A: The structure of acetic formic anhydride has to be given.
Q: The solubilities of three salts in water are shown in 1000 the graph. Pb(NO,, 900 800 K,Cr.O, For ea...
A:
Q: How much heat (in kJ) is evolved in converting 1.00 mol of steam at 145°C to ice at -50°C? The heat ...
A: Given: The number of moles of steam is 1.00 mol. The initial temperature of the steam and the final ...
Q: 1. IPROH, cat. H+ n-Bu 2. .CI Me CI Me NEtz, CH2CI2
A: Under acidic condition, epoxide opening occurs from more substituted carbon of epoxide Oxidation of ...
Q: A 75.0 g piece of Ag metal is heated to 80.0 °C and dropped into 50.0 g of water at 23.2 °C. The fin...
A: Given, Mass of Ag(silver) =75.0g It is heated to 80.0 °C. Final temperature of silver rod after drop...
Q: Methamphetamine is a long-lasting, potent stimulant sold as a street drug. The synthesis is quite si...
A: According to the question, we need to synthesis Methamphetamine (a street drug) from the given keton...
Q: The following chemical reaction takes place in aqueous solution: 2 AgNO3(aq)+Na,S(aq) →Ag,S(s)+2NaNO...
A: To write the net ionic equation the following steps are involved 1. Write the balanced equation 2....
Q: The concentration of an HCl solution is 0.567 g HCl/g of HCl solution (the solution containing BOTH ...
A: Given the concentration of an HCl solution is 0.567 g HCl/g of the solution it means that 1 g of HCl...
Q: Explain why the center of the band is used to calculate the Rf value for a cation rather than the le...
A: Retention Value (Rf value) is the distance moved by solute to the distance moved by solvent. These d...
Q: What is the molality, m, of a solution that contains 28.193 g of NaCl (58.44 g/mol) in 542.229 mL of...
A: Given: Mass of NaCl= 28.193 g Molar mass of NaCl= 58.44g/mol Moles of solute ,NaCl = mass of NaCl /m...
Q: 2. Show the Boyle temperature for a van der Waals gas is T Rb Hint: (1- a)-N1+a
A: Boyle's Law states that there is an inverse relation between the pressure of the given mass of the i...
Q: Explain the following using the principles of colligative properties. Antifreeze or a coolant is a ...
A: For part 1.) Antifreeze or a coolant is a liquid mixture containing water and a glycol compound that...
Q: Explain how Boyle’s law, Charles’s law, Avogadro’s law, andDalton’s law all follow from kinetic mole...
A: The kinetic theory of gases is a basic, historically important model of thermodynamic action of gase...
Q: Chemistry Question
A: NaH is in the form of – Na+ and H-
Q: Draw the perspective structure . Lone pairs must be written
A: Lone pair:- These are the non-bonding electrons, which are present on atoms. Lone pair makes an at...
Q: The unit of volume in the metric system is the liter, which consists of 1000 milliliters. How many l...
A: given that 1lt = 1000 milliliter The following conversion are given below:
Q: At 20°C, an aqueous solution that is 20.0% ammonium chloride by mass has a density of 1.0567 g/mL. ...
A: The question is based on concept of solution preparation and molarity concept. in first step, we cal...
Q: The picture below is a representation of H(g) and O2(g) in a sealed container. Ozia • Hag 2H2(g) + O...
A: Hydrogen and oxygen react to form water. The molecular formula of hydrogen is H2 and oxygen is O2 an...
what's the proper name for this molecule?
Step by step
Solved in 2 steps with 2 images


