Q: Draw a chiral alkene with the formula C6H12
A: Draw chiral alkene with the formula C6H12 ?
Q: Using the VSEPR Theory, draw the following compounds; represent them as covalent bonds. a) H2SO4 b...
A:
Q: convert and use sig figs 2.75kg to oz.
A: We have to convert the given quantity in oz
Q: Calculate the wavelength, in nanometers, of the spectral line produced when an electron in a hydroge...
A:
Q: Select the choice that best describes the relationship of the pair of compounds. H3C H. H3C CH3 H3C ...
A: The relationship between compounds is described on the basis of connectivity of bonds.
Q: 73. CH3OH can be synthesized by the reaction: CH3OH(g) CO(g) + 2 H2(g) What volume of H2 gas (in L),...
A: This question can be solved using law of multiple proportions and ideal gas law.
Q: OH KMNO4 a) H2O/MEOH, 0°c OH Br- Bra Oxone OH b) Acetone/H20, 0° HO. OH KMNO4 с) H2O/MEOH, 0°C OH Fo...
A: Concept: 1. an Alken react with KMnO4 to form cis- diol. 2. Oxone react with Alken and form a...
Q: Perform the following conversions and use sig figs 2.75kg to oz
A: Given values is 2.75 kg.
Q: Calculate the ∆G in kJ/mol for a reaction at room temperature (~25.00°C) that has a Keq = 1000.0. Us...
A: We have to calculate the ∆G.
Q: Write the net ionic equation for the following molecular equation. Cr(NO3)2(aq) + (NH4)½CO3(aq) →CrC...
A:
Q: What is the molar mass of sucrose (C12H22O11)?
A: Given :- Chemical formula of sucrose = C12H22O11 To be calculated :- Molar mass of sucrose
Q: 4Pd (CH, CH,),N organohalogen alkene
A: See answer below.
Q: Arrange the ions from largest to smallest. Largest ionic radius to Smallest ionic radius. O^2- , F...
A: The trend of ionic radius is given below:
Q: exothermic Is this reaction exothermic, endothermic, or neither? O endothermic O neither If you said...
A:
Q: The compound cobalt(II) nitrate is a strong electrolyte. Write the reaction when solid cobalt(II) ni...
A:
Q: Which of these four compounds is most likely to glve rise to the proton NMR spectrum below?
A: Given is 1Hnmr spectrum, which has 2 signals Given.
Q: N2(g)+3H2(g)→2NH3(g) A) If you have 4.00 gg of H2H2, how many grams of NH3NH3 can be produced? B) ...
A: N2(g) + 3H2(g) →2NH3(g) 1 mol N2 reacts with 3 mol H2 produces 2 mol NH3 molar mass of N2 = 28.0 g...
Q: the molar mass of the metal.
A:
Q: For the substituted cyclohexane compound shown, identify the atoms that are trans to the bromo subst...
A: Here fluorine is axially above so those atom which are below are trans to bromine
Q: What is the balanced net ionic equation for this reaction. РЫ (NO,),(аq) + 2HCI(aq) PBCI, (s) + 2HN0...
A:
Q: Order the element Rb , Na , C , F in order of increasing ionizing energy. Explain your answer
A: Ionization Or ionizing energy is the energy required to remove electron from valence shell.
Q: Show the dipole moment of each bond and then predict the overall molecule as either being polar or n...
A: Polar and non polar nature of molecule is determined by net dipole moment.
Q: Radium Isotope Ra-226 a) Fully show the decay scheme of this isotope as it is transformed into radon...
A: The radioactive decay reaction takes place when the nucleus of the isotope is unstable. The stabilit...
Q: In [Ni(DMG)2], is DMG a chelating ligands and what is the type of this ligand depend on the number o...
A: The type of ligand is depends upon number of donor sites by ligand to the central metal ion.
Q: For many purposes we can treat methane (CH,) as an ideal gas at temperatures above its boiling point...
A:
Q: Liquid octane (CH, (CH,) CH,) will react with gaseous oxygen (O,) to produce gaseous carbon dioxide ...
A:
Q: Is this reaction exothermic, endothermic, or neither? If you said the reaction was exothermic or end...
A:
Q: The solubility of PbF, is measured and found to be 0.526 g/L. Use this information to calculate a Ke...
A: We have to calculate the Ksp for lead fluoride.
Q: If 2.50 g of aluminum metal is heated in Fluorine gas it’s found that 5.28g will combine with Alumin...
A: Given data, Mass of aluminium = 2.50 g Mass of F = 5.28 g
Q: • Part A Electromagnetic radiation with a wavelength of 641 nm appears as orange light to the human ...
A: Given data Photon energy : Ep=3.10×10-19 Jphoton Energy emit from laser in one pulse : E=1.3×10-2 Jp...
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A: The amount of hydrochloric acid taken = 1.1 g The amount of sodium hydroxide taken = 0.79 g We have ...
Q: of decreasing acid strength u
A: Acid is proton donor while base is proton accepter.
Q: When Vinylcyclopentane is reacted with H2O in presence of acid, 1-cyclopentylethanol is the major pr...
A:
Q: The ideal gas constant (R) has a value of 0.0820578 L atm mol^-1 K^-1. Determine the value of the ga...
A: We have to calculate the gas constant in SI unit.
Q: Calculate the energy required to heat 676.0 g of cyclohexane from 16.1 °C to 36.6 °C. Assume the spe...
A: Specific heat capacity (C) :- The amount of heat required to rise the temperature of one gram of a s...
Q: What product do you expect from the reaction between 3-benzyl-1H-Indole with allyl bromide in presen...
A:
Q: Explain the gas laws in terms of the Kinetic Molecular Theory
A: Various gas laws can be explained in terms of Kinetic Molecular Theory.
Q: A student dissolves 13.8 g of lithium chloride (LİCI)in 200. g of water in a well-insulated open cup...
A: Given the mass of LiCl taken = 13.8 g mass of solvent water taken, m = 200 g temperature rise, ∆T = ...
Q: Draw the step by step structures to obtain the Lewis dot structures and balance the charges of the f...
A: Ans AlF3 here Al has 3 valance electrons and 7 has 1 valance electrons 3 + 7(3) = 24 electrons And...
Q: Aluminum sulfate is analyzed, and the sample contains 0.0667 moles of sulfate ions. How many moles ...
A: Given;
Q: ance the following equations, name reactants and products, name reactions type and prove oxidation/r...
A: The reaction is provided in the next step:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A: In the given acid base neutralization reaction, NaOH is acting as a limiting reagent which will deci...
Q: Give the symbol of the element that has the least metallic character in Group 8A (18). Give the sym...
A:
Q: An arctic weather balloon is filled with 16.6L of helium gas inside a prep shed. The temperature i...
A: The new volume is calculated by using ideal gas equation PV = nRT.
Q: Predict the product for the reaction: Draw the product Select Draw Rings More C CI H-CI
A: Given reaction is, hydrohalogenation reaction.
Q: The compound zinc fluoride is a strong electrolyte. Write the reaction when solid zinc fluoride is p...
A: Zinc fluoride is an ionic compound, it is an inorganic compound with Chemical formula of ZnF2. It ca...
Q: Radium Isotope Ra-226 a) Fully show the decay scheme of this isotope as it is transformed into radon...
A: The conversion of a heavier and unstable nucleus into the lighter nuclei along with the liberation o...
Q: X=[Ar]4s23d5X=[Ar]4s23d5, Y=[Ar]4s23d104p1Y=[Ar]4s23d104p1, Z=[Ar]4s23d104p6 metal non metal...
A: Given the electronic configuration of X is X: [Ar] 4s2 3d5, the electronic configuration of Y is Y: ...
Q: How many acidic hydrogens are present in the molecule shown below? None 2 4 3 1
A: We have to calculate the number of acidic hydrogen.
Q: If the reaction mixture containing p-aminophenol, distilled water, and acetic anhydride was heated t...
A:
draw chair confiration
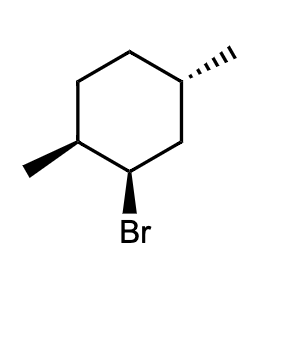
Step by step
Solved in 3 steps with 2 images


