Q: 41. A nitric acid solution containing 71.0% HNO3 (by mass) has a density of 1.42 g/mL. Ho many moles…
A: Given that mass percent of nitric acid is 71% and density is 1.42 g/m L.
Q: 16. Consider the generic chemical reaction:2 A + 3B -> 3 CHow many moles of B are required to…
A: Given: Moles of the reactant A (n): 6 moles, 2 moles, 7 moles, 11 moles Moles of B are needed to be…
Q: B. Weight of Na2SO4 Sample 0.3512 g C. Total Vol. of Precipitant (BaCl2) used WITH 1М ВаCI2 PLEASE…
A:
Q: A concentrated perchloric acid solution has a density of 1.67 g/mL at 25°C and is 11.7 M. What is…
A: Given data is as follows: The density of the perchloric acid solution = 1.67 g/mL The molarity of…
Q: Consider the following chemical reaction: KI (aq) + Pb(NO3)2(aq) -----------> PbI2(s) + KNO3(aq)…
A: Given reaction, KI (aq) + Pb(NO3)2(aq) -----> PbI2(s) + KNO3(aq) Determine molar ratio of PbI2 /…
Q: seM Determine the mass (in g) of O2lg) that was consumed during this
A: A combustion reaction is a type of chemical reaction in which a compound and an oxidant is reacted…
Q: What is a combustion reaction? Give an example.
A: Combustion reaction is one of the important class of reactions. In this reaction, substance reacts…
Q: ygen nitial appearance of Mg Shiny lisht Sroy ribbonot mekal 2. Evidence of chemical reaction Hea t…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: A 0.640 g sample of a metal, M, reacts completely with sulfuric acid according to M(s) + H,SO,(aq) –…
A: As per equation 1 mol of metal M gives 1 mol of H2 gas First find the moles of H2 gas by using…
Q: 4.3.4 Which of the following is the correct net ionic equationfor the reaction of HF and LiOH?
A: Solution- let us consist the moleculer equation for the combination of HF and LiOH-HF and LiOH…
Q: (а) (b) NaN3 KCN ? H20 ? HOCH,CH,OH (с) (d) HCN 1. LIAIH4 ? HOCH,CH,OH ? 2. Н,о°
A:
Q: H,0, H* .COOH H,PO, 1. Оy 1. BH, KMnO4 KMNO4 2. H,O, OH 2. Zn, H2O
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: a. KO2(s) + H20(1) → KOH(aq)+ O2 (g) + H2O2(aq) b. Fe, O3 (s) + HNO3(aq) → Fe(NO3)3 (aq) + H2O(1) c.…
A: Using hit trial method In this method we have to equal all elements both side of arrow by…
Q: AgNO3 + CaCl2 --> AgCl + Ca(NO3)2 When properly balanced, coefficient of silver nitrate is:
A:
Q: tilanes, Cll, is a spe ihai iKSCin in fnita. Ii is sla: kratwn "lad agur berDE i is ieune in bl:ad…
A: Molality (m): Molality is the number of moles of solute present in one kilogram of solvent.
Q: a. Al(s) + MnO, (aq) → MnO,(s) + Al(OH), (aq) b. Cl,(g) →CI (aq) + OCI (aq) c. NO, (aq) + Al(s)…
A:
Q: d. For the reaction 2NO(g) → N2(g) +02(g) Species S J/(mol-K) NO(g) 210.6 N2 (9) 02(g) 191.6 205.0…
A: The change in entropy, ∆S° is the difference between the sum of the entropies of the products and…
Q: Wine is approximately 12% ethanol (CH3CH2OH) by volume. Ethanol has a molar mass of 46.06 g/moland a…
A:
Q: Use the oxidation number method to balance the followingequations by placing coefficients in the…
A: I am supposed to ans only the 3 sub parts sonols do repost the same for the rest.
Q: The solubility differs from concentration because concentration is amount of solute per volume of…
A: Solution is the homogeneous mixture of two or more substances which consists of two phases solute…
Q: Consider the following reaction Ca(s) + 2HCI(aq) --> CaCl,(aq) + H2(8) How many moles of HCl are…
A: Calcium metal reacts with hydrogen chloride to form calcium chloride and hydrogen gas. The equation…
Q: Consider the reaction of zinc metal with hydrochloric acid, HCl(aq). Zn(s) + 2 HCl(aq) --> H2(g) +…
A: Balanced chemical equation: Zn(s) + 2 HCl(aq) ----> H2(g) + ZnCl2(aq) From the above equation:…
Q: E. What type of reaction is the following? 2KCIO3 2KCI + 302 E. What type of reaction is the…
A:
Q: A 12.3 g sample of an alloy of Mg and Al produces 1.66 mol of H2 when added to excess HCI. What…
A: Given, Mass of the sample = 12.3 g Mass of H2 gas produced = 1.66 mole What is the percentage of…
Q: a 10.000 g sample of hydrated nickel iodate Ni(IO3)2 * H20 is heated in order to driver off the…
A:
Q: Methanol (CH3OH) is a liquid at room temperature with a density of 7.91 ×102 kg/m3 . In a certain…
A: (a) Methanol combust with oxygen to give carbon dioxide and water. Hence it is a combustion…
Q: Given the following equation: 2 AGNO3 + CaCl2 --> 2 AgCI(s) + Ca(NO3)2 What is the net ionic…
A: 2 AgNO3 (aq) + CaCl2 (aq) ==> 2AgCl (s) + Ca(NO3)2 (aq)
Q: se ( x Edward V x O procedur x G traductor x E Imt rgt c x Course H X Part The x Part A A X…
A: 2.4 moles of ethane undergoes combustion reaction with 3.5 moles of oxygen. Here we have to…
Q: A 0.355-L soft drink sample contains 0.071 mol of sucrose (table sugar). What is the molar…
A: Given, moles of sucrose (table sugar) = 0.071 mol Volume of the soft drink sample = 0.355 L…
Q: 1) 1 eq HCI ? 2) 1 eq HBr Br CI Br CI Br CI Br CI Br Br CI I II III IV V %3I
A: Given reaction is : Predict the major product of the reaction = ? Options are :
Q: A classic chemistry class demonstration is the burning of magnesium ribbon with a Bunsen burner. In…
A: The limiting reagents the starting reactant molecules that are used completely with the course of…
Q: A 0.355 M K2SO4 solution can be prepared by
A: Dilution is a process via which the concentration of a solution is decreased by adding extra…
Q: What is net ionic for CuSo4+NH4Cl ? Does CUCl2 Considered Insoluable ?
A: The reactants given are 1) CuSO4 (aq) 2) NH4Cl (aq) Since the 2 salts will react and undergo…
Q: a) HNO, + S b) CrCl, + MnO, + H;0 c) KMNO, + HCI + H,S NO, + H,SO, + H;0 - MnCly + H;CrO → KCI +…
A: We are given three redox reaction we have to show the change in oxidation states and hence balance…
Q: CrOs HCl PCC CH;(CH,),CH,OH CH;Cl, (solvent)
A: PCC (Pyridinium chlorochromate) is an oxidizing agent with reduces the primary and secondary alcohol…
Q: & or ob OEt oor DEt Cl₂ H₂O* NaOH, H₂O, A 1. NaOEt, EtOH 2. H₂O* 1. LDA, THF, -78 °C 2. 1₂ NaOH, H₂O…
A:
Q: F25gof a 23% by mass magnesium chloride (MW= 95.21 g/mole) solution is reacted with 125.0 mL da125M…
A: Here the problem is solved by mole ratio and the concept of limiting reagent. First it is determined…
Q: A sample of calcium oxalate and magnesium oxalate weighing 0.6240 g is heated and converted to…
A: A chemical reaction is symbolic representation of the conversion of substances to new substances.In…
Q: Consider the reaction: 2 Al(s) +3H2SO4(aq) --> Al2(SO4)3(aq) + 3H2(g) (molar mass of aluminum…
A: • No. of moles = Given massMolar mass• Mass = No. of moles x Molar mass• No. of moles =…
Q: The solution is made by dissolving 25 g fructose in water to make 100 g of the solution. What is…
A: %W/W of the solution is Mass of solute/mass of solution × 100% mole of the solution = mass of…
Q: Weight of Na2SO4 Sample 0.3512 g C. Total Vol. of Precipitant (BaCl2) used WITH 1M BaCl2…
A: Given that the weight of Na2SO4 sample is 0.3512 g.
Q: For ench renction! AJ urite word equntion b) vrite the balanced molecUlas equton C) write he total…
A: 1) The reactants given are AgNO3 (aq) i.e. silver nitrate and Na2CO3 (aq) i.e. sodium carbonate.
Q: c) List An ore with the mass of 1.52 g is analyzed for the manganese content (Mn) by converting the…
A: Ans.
Q: MgO(s) + CO2(g) ⇌ MgCO3(s) [MgCO3(s)] [MgO(s)][ CO2(g)]…
A: Reaction given as : MgO(s) + CO2(g) ⇌ MgCO3(s)
Q: 25.015g 775225g Mass of 3.0% H2O2 solution Mass of H2O2 in 3.0% solution Moles H2O2 decomposed mol
A: Molar mass of H2O2 = 2 (Atomic mass of H) + 2 (Atomic mass of O) = 2 ( 1 g/mol ) + 2 ( 16 g/mol )…
Q: HBr (aq) + KOH (aq) KBr (aq) + H2O (1) PbCO3 (s)→ PbO (s) + CO2 (3) Zn + FESO4ZNSO4 + Fe
A: Interpretation: We have to classify them as neutralization , gas forming or redox reaction.
Q: 3. A lactory is pumpingihiercury bydroxide (Hg(OH)inta the Vocal lek a1,250 kilogram sample bf lake…
A:
Q: B. A chemist adds water to 25 mL of a 2.4 M solution of NaOH until the final volume is 3.02 L. What…
A: Dilution of the solution: On dilution of the solution with the pure water, the mole of solute…
Q: Consider the reaction: 2KCIO3 2KCI+302 1. How many moles of KCIO3 are required to produce 22.8 moles…
A: The given data : a) The number of the moles of Oxygen gas = 22.8 moles b) The moles of KClO3 = 18.8…
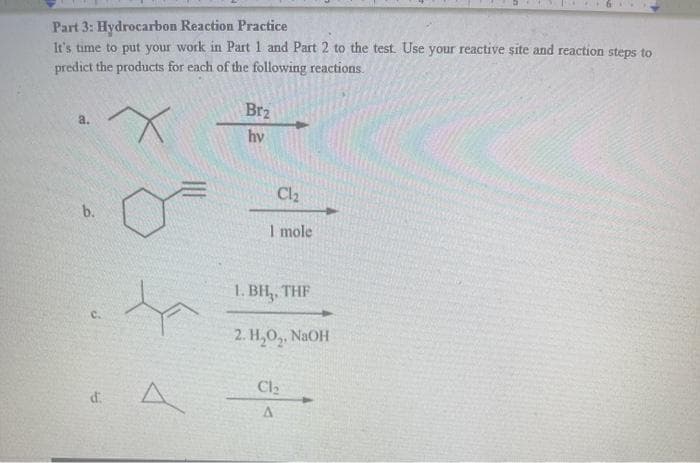
Step by step
Solved in 2 steps with 2 images

- This reaction must have a ∆G<0 at SATP: Xe(g)+F2(g)→XeF2(g) 2Na(s)+Cl2(g)→2NaCl(s) 2NaF(s)+I2(s)→2NaI(s)+F2(g) MgSO4(s)→Mg(s)+S(s)+2O2(g)Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2OProf. Neiman and Prof. James were first to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?
- Find out feasible reaction in high temperature gas-phase from following reactions? explain reasoning in all cases- (a) CuBr2(g) + 2NaF(g) --> CuF2(g) + 2NaBr(g) (b) TiF4(g) + 2TiI2(g)--> TiI4(g) + 2TiF2(g) (c) CuI2(g) + 2CuF(g) --> CuF2(g) + 2CuI(g) (d) CoF2(g) + HgBr2(g) --> CoBr2(g) + HgF2(g)Reaction of interest : S2O82-(aq) + 3I- (aq)→ 2SO42-(aq) + I3-(aq). rate= k[S2O82-]1[I-]1.Average reaction rate between 0 and 100 us? Average reaction rate between 200 and 300 us?
- UCI Chemistry researchers, Prof. F. Sherwood Rowland and Dr. Mario Molina werefirst to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?-OAc can be used for Sn2 as well How would this be incorporated into the answerPls show eork, will rate, D,E,F

