Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 53AP
Related questions
Question
Briefly explain why OtBu- sometimes favored over hydroxide as an elimination reagent?
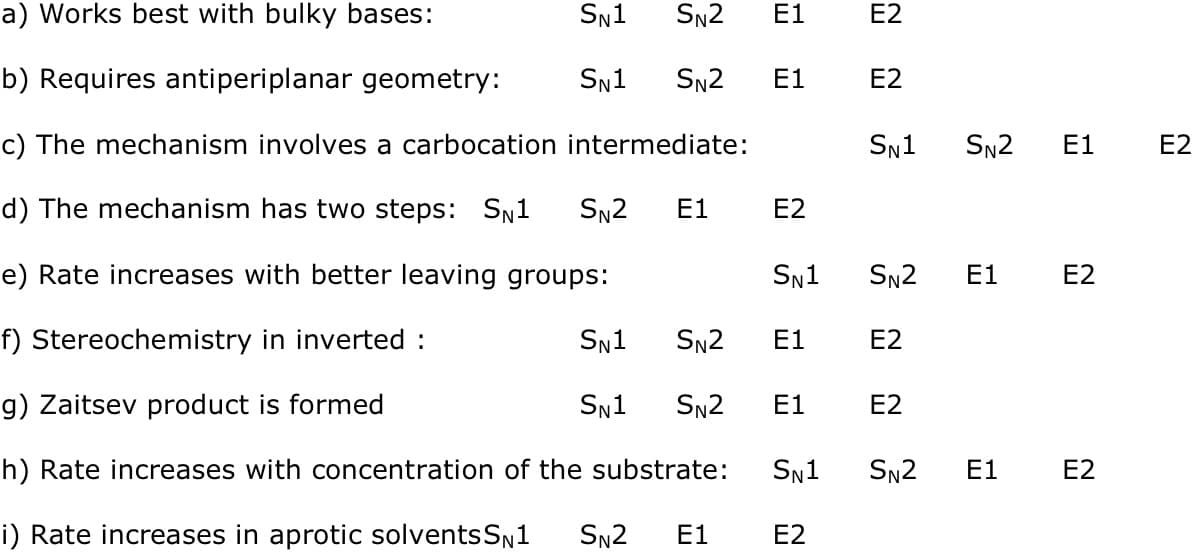
Transcribed Image Text:a) Works best with bulky bases:
SN1
SN2
E1
E2
b) Requires antiperiplanar geometry:
SN1
SN2
E1
Е2
c) The mechanism involves a carbocation intermediate:
SN1
SN2
E1
E2
d) The mechanism has two steps: SN1
SN2
E1
E2
e) Rate increases with better leaving groups:
SN1
SN2
Е1
E2
f) Stereochemistry in inverted :
SN1
SN2
E1
Е2
g) Zaitsev product is formed
SN1
SN2
E1
Е2
h) Rate increases with concentration of the substrate:
SN1
SN2
Е1
Е2
i) Rate increases in aprotic solventsSN1
SN2
Е1
E2
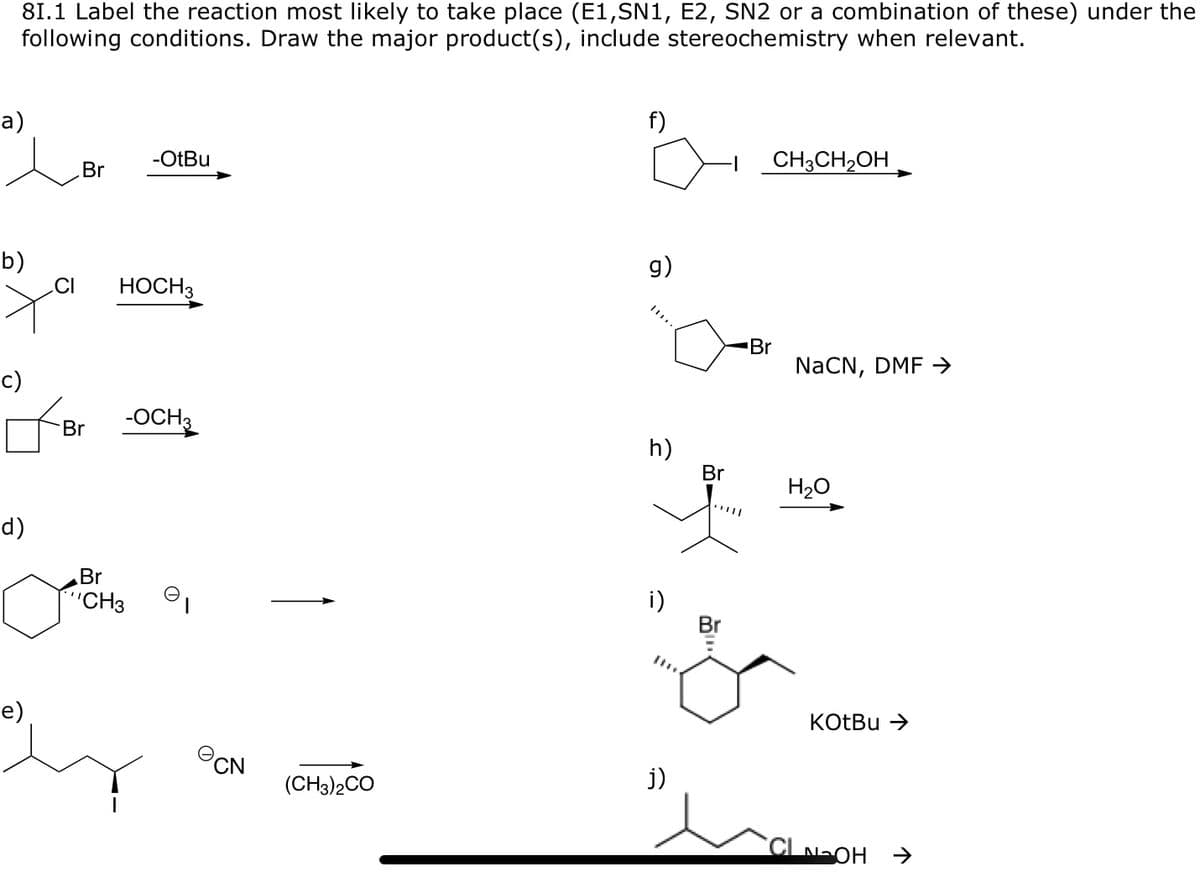
Transcribed Image Text:81.1 Label the reaction most likely to take place (E1,SN1, E2, SN2 or a combination of these) under the
following conditions. Draw the major product(s), include stereochemistry when relevant.
а)
f)
Br
-OtBu
CH3CH2OH
b)
CI
g)
НОСН
Br
NaČN, DMF →
Br
-OCH3
h)
Br
H20
d)
Br
"CH3
i)
Br
e)
KOTBU >
PCN
(CH3)2CO
j)
CLNOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning