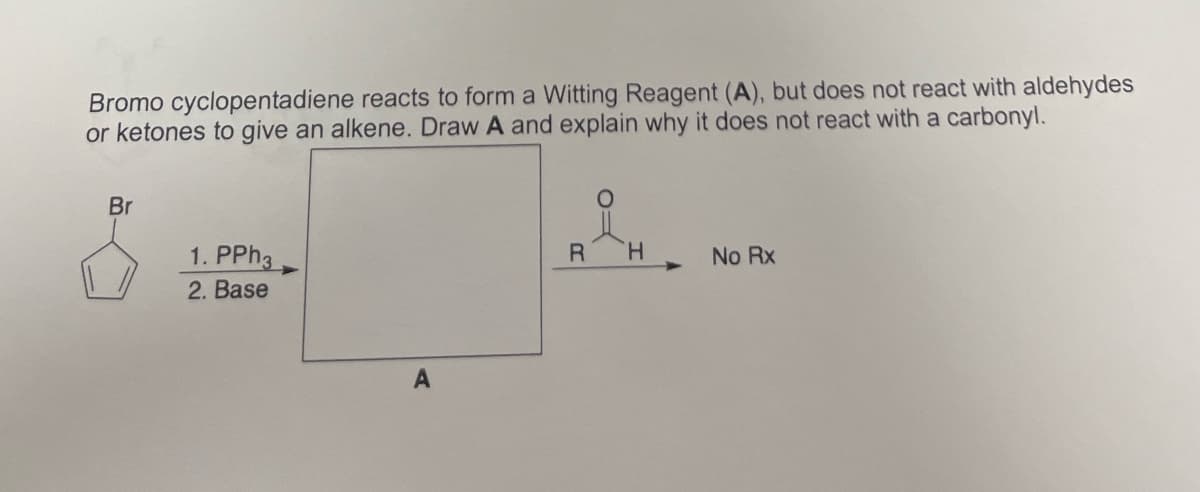
Bromo cyclopentadiene reacts to form a Witting Reagent (A), but does not react with aldehydes or ketones to give an alkene. Draw A and explain why it does not react with a carbonyl. Br 1. PPh3 2. Base A R H No Rx
Q: Which of the following carbocations would you expect to rearrange? If you expect rearrangement, draw…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1×10 -14 -8 = (6.4×10³ +x)x
A: Step 1: Given equation is: 1×10−14=(6.4×10−8+x)x x2+(6.4×10−8)x−10−14=0 ⇒x2+1086.4x−10−141=0 Tip:…
Q: Starting with diethyl malonate, and using any other reagents of your choice, show how you would…
A:
Q: Please don't provide handwritten solution ....
A: GivenMassM1 =0.125 kg (unknown)M2=0.285 kg (water)M3=0.150kg(calorimeter)Temparature,T1=…
Q: None
A: Step 1:Explanation Step 2: Step 3: Step 4:
Q: Check the box under each compound that exists as a pair of cis/trans isomers. If none of them do,…
A: Step 1: Step 2: Step 3: Step 4:
Q: The base protonation constant K of allantoin (C4H4NO3NH2) is 9.12 × 106. Calculate the pH of a 0.44…
A: Step 1: Step 2: Step 3: Step 4:
Q: The reaction C2H5 + OH →C2H5OH + I was studied, and the following rates were obtained. Temperature,…
A: The objective of the question is to detrmine the followingslopey-interceptPearson…
Q: For the following structure, identify the spatial relationship between two or more hydrogens that…
A: Step 1: Step 2: Step 3: Step 4:
Q: need help determining the final product organic and inorganic
A: The final product is given and the mechanism is also shown. The N of semicarbazide which is more…
Q: Cations Which of the following is considered a cation? Na+ CI- CI Na
A: Step 1: SolutionThe cation is -----> Na+ Explanation: we know that the chemical ions with a…
Q: 4. Calculate the average volume of sodium hydroxide used for the back titration. 5. Calculate the…
A: A reaction is a balanced reaction if it contains the same number of atoms on both sides of the…
Q: Payalben
A: The objective of the question is to propose a mechanism for the biosynthesis of terpene Eucalyptol…
Q: Draw the structure of all products of the mechanism below. H H H + N 21 OH H H C=C H H Click and…
A: By following the curved arrow mechanism, the product formed can be given as
Q: H3Cm Br cyclohexane I CH3
A: The objective of the question is to find out the both conformer of the given cyclohexane molecule…
Q: A 131.7 mg sample of powdered Carbamazepine tablets was boiled with ethanol, filtered and then made…
A: Step :
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: i.) Ester hydrolysis can occur in acidic or basic conditions. Predict the product AND show the…
A: c)This reaction proceeds without the need for an external acid or base catalyst because water itself…
Q: 2) For each of the carbocations shown below, include a mechanism that shows its rearrangement to the…
A: The objective of the question is to determine the rearrangement of given carbocations to their most…
Q: The great expert Hand written solution is not allowed. Please give explanation.
A: Shown below is the peptide histidylglycylglutamylisoleucine.The name suggests that there is a…
Q: Consider the pair of reactions below to answer the following question(s). 70% H₂O a. (CH3)3C-C1…
A: The objective of the question is to determine which of the two given reactions is faster and to…
Q: For the question, balance the following chemical equations with proper coefficient(s). For example,…
A: Balancing the chemical equation step by step: Given equation: Pb(NO₃)₂ + NaCl → PbCl₂ + NaNO₃ 1.…
Q: A chemist adds 265.0 mL of a 0.0539uM copper 2 sulfate (CuSO 4 ) solution to a reaction. Calculate…
A: Step 1:This is a conversion problem. It is easier for me to convert the given to its standard units…
Q: Draw the first compund in Fischer structure and 3D perspective structure( as if it was a molecular…
A:
Q: Please correct answer and don't use hend raiting
A:
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking for the structure of the amino acid tyrosine in its completely protonated…
Q: 7. What is the equilibrium constant, K, at 25°C for this redox reaction under standard conditions?…
A: The objective of this question is to calculate the equilibrium constant, K, for the given redox…
Q: 7. Predict the products for the reactions below. 1) excess EtMgBr 2) H20 excess NH3 ОН pyridine 1)…
A: Step 1: a) Propanoic anhydride with excess of ethyl magnesium bromide gives 3-ethylpentan-3-ol.First…
Q: List two possible steps in the procedure that might've led to the gain and impurities to appear to…
A: The objective of this question is to identify two possible steps in a cycle of reactions involving…
Q: 2. Suggest a stepwise synthesis for the following reactions. CLEARLY showing the PRODUCTS AT EACH…
A: SYNTHESIS :EXPLANATION:In the first step phenoxide ion was prepared from phenol by using base NaOH.…
Q: :):):));;$:&:&$:
A:
Q: Aqueous sulfuric acid (H_{2}*S*O_{4}) reacts with solid sodium hydroxide (NaOH) to produce aqueous…
A: The objective of this question is to calculate the percent yield of sodium sulfate in a chemical…
Q: Calculate the volume in of a 0.606M copper(II) sulfate solution that contains 500. mmol of copper 2…
A: The objective of this question is to calculate the volume of a 0.606M copper(II) sulfate solution…
Q: 9. (10 marks) The equilibrium constant (KP) at 25 °C is 3.2 x 10-10 for the reaction: 2 Cr³+ (aq) +…
A: The objective of this question is to find the equilibrium concentrations of Cr3+, Cr2+, and Pb2+ in…
Q: I need help with part b
A: 2. After adding 15 mL of KOH: At this point, 15 mL of 0.100 M KOH reacts with the barbituric acid.…
Q: The reaction order does not have to take only integer values. In order to decompose substance A, the…
A: Step 1: Step 2: Step 3: Step 4:
Q: Assume that each compound in the drug mixture would make a spot on the paper the same size as those…
A: Step 1: No, it's unlikely that paper chromatography would be sufficient to separate all the…
Q: Organic Chemistry Please help with solving problem. What is the product of this reaction? Thank you
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Which of the following is expected to be less soluble in water: ZnS or HgS? Explain the answer in…
A: Here is a list of Solubility Rules for Ionic compounds: Zinc sulfide (ZnS) is an ionic compound…
Q: I need answer expert solutions and step by step
A: Step 1: Understanding the ProductProduct Name: The product is a single-dose vial containing 10 mL of…
Q: Propose an efficient synthesis for the following transformation: Br The transformation above can be…
A: Step 1: Step 2: Step 3: Step 4:
Q: A student performed titration to determine the concentration of potassium permanganate solution with…
A: The objective of this question is to determine the concentration of the permanganate solution used…
Q: None
A: Step 1:C3H8 + O2 → CO2 +H2OWe have to balance this reactionStep 2:Balance Carbon Atoms: Start by…
Q: need help with the major product
A: Step 1: Step 2: Step 3: Step 4:
Q: P
A:
Q: Write a charge balance equation and one or more mass balance equations for 0.10 M HCl and 0.050 M…
A: The objective of the question is to write a charge balance equation and one or more mass balance…
Q: Part 1 of 2 H2 in the presence of a Pd catalyst. Modify the Fischer projection of the monosaccharide…
A: let's break down the hydrogenation process step by step:Starting Fischer Projection:The given…
Q: H Convert the aldohexose to the a anomer using a Haworth projection. C=0 HO -H HO H H .OH H OH CH₂OH…
A: If we convert it into the α anomer using the Haworth projection, we need to represent it in a cyclic…
Q: Provide the necessary sequence of reactions to generate the products below from their respective…
A: Step 1: In the first step, the methyl cyclopentane undergoes monochlorination in the presence of…
Q: Check the box under each molecule in the table below that is an isomer of this molecule: HO If there…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,DA. OsO4 and NMO B. Br2 and H20 C. Hg(OAc)2, H2O and NaBH4, NaOH D. RCO3H E. BH3-THF and H2O2, NaOH Which reagent will complete this reaction?Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):
- 1 Consider the reaction of (R)-2-chloro-3-methylbutane with sodium iodide to form aproduct.(a) Draw the reaction scheme with the correct stereochemistry (reactant + NaI → product+ NaCl). Circle the nucleophile and draw a rectangle around the electrophile.(b) What is the symbol used for mechanism shown in 1(a)(c) If the sodium iodide was replaced with sodium hydroxide, the product is anALKENE. Draw a reaction MECHANISM to show how this happens.(d) Draw the reaction energy diagram for the reaction in 1(c) and label the activationenergy. (e) Using any alcohol with five carbons, and any carboxylic acid with six carbons, draw areaction to show how we would make an ester.(f) Describe the practical on esters. Please answer (d) to (f)2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 31a) What’s the product in the following reaction? a. 4 b. 1 c. 2 d. 5 e. 3 1b) What reagents would you use to prepare product A with good yield? a. CH3CH2CH2CH2I; 2) Li, NH3 (liq), -33 °C b. KNH2; 2) CH3I; 3) H2, Pd/C c. HBr; 2) (CH3CH2CH2CH2)2CuLi d. NaNH2; 2) CH3CH2CH2CH2Br; 3) H2, Lindlar catalyst e. (CH3CH2CH2CH2)2CuLi; 2) H2, Lindlar catalyst
- What alkene is needed to synthesize each 1,2-diol using [1] OsO4 followed by NaHSO3 in H2O; or [2] CH3CO3H followed by −OH in H2O?What is the rate law implied by the mechanism given below? CH3COCH3(aq) + H+(aq) ←→ CH3C(OH)CH3+(aq) (fast, reversible) CH3C(OH)CH3+(aq) → CH3C(OH)=CH2(aq) + H+(aq) (slow) CH3C(OH)=CH2(aq) + Br2(aq) → CH3C(OH)CH2Br+(aq) + Br-(aq) (fast) CH3C(OH)CH2Br+(aq) → CH3COCH2Br(aq) + H+(aq) (fast) A. Rate = k[CH3COCH3][H+] B. Rate = k[CH3COCH3] C. Rate = k[CH3COCH3][Br2] D. Rate = k[CH3COCH3]2 E. Rate = k[CH3COCH3][Br2]/[H+] (Answer is A, looking for explanation why!).What is a possible formula for a carbocation with m/z=97
- What is the major product of the reaction below? What is the mechanism? (See the image) a. 2-bromo-3-methylpentane, SN2 b. 3-methoxy-2-methylpentane, SN2 c. 3-bromo-2-methylpentane, SN2 d. 2-methoxy-3-methylpentane, SN2Write reactions of propan-1-ol with the following reagents: a. Na b. PCl54 Identify the required reagent(s) for step 1. Select the single best answer A. Br2 , CCl4 B.Br2,H2O C. HBr D. mCPBA E.1BH3,THF;2.H2O2,NaOH F. H2O, H2SO4 Step 2: Draw the structure for compound A. Step 3: CH3CO2Na NaOCH3 CH3OH, heat H2SO4, heat t-BuOK, heat NaCN

