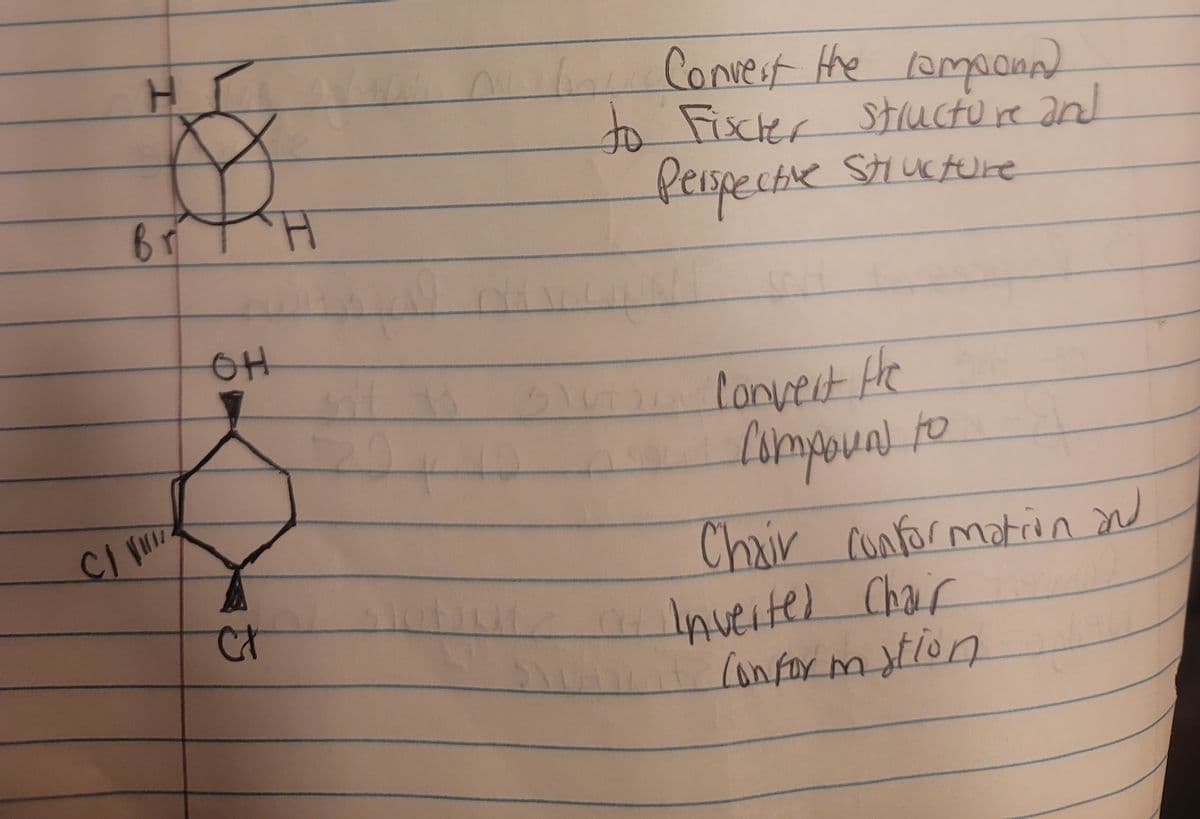
H Br H Convert the componn) to Fischer structure and Perspective Structure OH Ct Convert the Compound to Chair conformation and a Inverted Chair Conformation
Q: The solubility of AgCl in water at 25 °C is measured to be 0.0019 Round your answer to 2 significant…
A: Given solubility of AgCl is 0.0019 g/LMolar mass of AgCl is 143.5 g/molSolubility in mol/L can be…
Q: None
A: Rules in assigning priorities in Cahn-Ingold-Prelog system: (1) Atomic Number - The atom directly…
Q: Please draw out the whole mechanism for the following reaction.
A: The question involves the transformation of cyclohexa-1,3-diene into…
Q: help 6?
A:
Q: 7. Which of the following species are likely to be nucleophiles and which electrophiles? ( explain…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A:
Q: Explain why each of the following reactions will not proceed as written. a. + CO2 COOH b.…
A: Substitution reactions occur when an atom or a group of atoms is replaced by another atom or group.…
Q: 3)Which has the most liters? has the highe a) 1.22 moles N2 b) 3.22 moles CO2 c) 1.66 moles CO d)…
A: Step 1: SolutionWe know the ideal gas equation=> PV = nRT where, P is pressure in atm, V is…
Q: A 3.0 L container of neon gas has a pressure of 1.0 atm. What will the pressure be if the volume is…
A: The objective of this question is to find the new pressure of the neon gas when the volume is…
Q: Assessment 8.50 Predict the products you would get when the following alkenes react under the…
A: Step 1: Step 2: Step 3: Step 4:
Q: The van der Waals equation of state was designed to predict the relationship between pressure p,…
A: Step :
Q: 25 26 27 28 29 3 8 3.5 4 4.5 8 288 401 8 201 20 10 5 4. Molecular weight = 86; with 10 MICRONS 5.5 6…
A: Rule of thirteen is a simple mathematical calculation which can be used to determine the molecular…
Q: Provide synthetic strategies that will transform the unsaturated ketone in the middle to the two…
A: The objective of the question is to transform the unsaturated ketone to the lactones given.An…
Q: Compare and contrast the amount of caffeine you obtained from the single vs multipleextractions with…
A: Approach to solving the question:Understanding the Experiment Setup: Firstly, it's essential to…
Q: Draw the first compound on Newman structure and Fischer structure Draw the second compund in Wedge…
A:
Q: Part II: 1. Draw the mechanism and the energy diagram for the reaction shown below. Include any…
A: Here in this question H3O+ given which is a source of H+ and H2O.
Q: None
A:
Q: Need full mechanism
A: The objective of the question is to provide the mechanism.
Q: 2) For each of the carbocations shown below, include a mechanism that shows its rearrangement to the…
A: The objective of the question is to determine the rearrangement of given carbocations to their most…
Q: Homework: The pK values of glutamate (Glu, E) are: 2.19, 4.25 and 9.67. You have 10 mL of 1M…
A: The objective of the question is to calculate the .
Q: Decrease volume Add Heat Left Left END Left Remove HCl(aq) Right Right Left Right Decrease pressure…
A:
Q: Draw the missing organic structures or select the missing reagents in the following multistep…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: O Chemical Reactions = Calculating molarity using solute moles 3/5 Bisho A chemist prepares a…
A: Step 1:- To calculate the concentration of the barium acetate solution, we use the…
Q: The Kp for the reaction A(g) ⇌ 2B(g) = 0.0450. So, what is Kp for the reaction 2 A (g) ⇌ 4 B (g)
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: True or false, explain your answer.
A: The compound contains a stereocenter, specifically the carbon atom bonded to the oxygen atom (C∗).…
Q: please explain the answer
A: The objective of the question is to find the coefficient of Ni in the balanced chemical equation for…
Q: PV = nRT N2(g) + 3H2(g) --> 2NH3(g) A steel tank is filled at constant temperature with 428 torr of…
A: Given: PN2=428torr;PH2=515torrN2(g)+3H2(g)→2NH3(g)Step 1: Solve for the pressure of NH3…
Q: Determine the value of Ksp for Zn (PO4)2 by constructing an ICE table, writing the solubility…
A: Step 1: Recall that pure substances (solid and liquid) are not included in an ICE table. Let x be…
Q: Be sure to answer all parts. -1 The rate constant of a reaction is 7.7 × 10³ s−1 at 25°C, and the…
A:
Q: After creating her standard curve for absorption versus the concentration (M) of FD&C Red 40 and…
A: The objective of this question is to find the percent by mass of FD&C Red 40 in the Kool-Aid…
Q: Please re do over the work and check it it correct
A: Treatment of vicinal (two halogens on adjacent carbons) dihalides with a strong base like NaNH2…
Q: Please make sure answer is correct a lot of controversial answer on this I will trust you and take…
A:
Q: 8. Provide the product for the reaction below. Do not copy and paste from someone else's work; must…
A: Step 1:Step 2:
Q: The following results were obtained for an enzyme-catalyzed reaction Substrate concentration (mmol…
A: The objective of this question is to calculate the Michaelis-Menten constant (Km) and the maximum…
Q: ㅁ Question 9 1.5 pts A researcher wants to determine the concentration of beta-carotene in carrots.…
A: If we calculate all the Molarity of solution using M1V1=M2V2, then this will be the chart and the…
Q: Draw an alkyl halide that would undergo an SN2 reaction to yield this product under the conditions…
A: The reaction proceeds via SN2 mechanism. In this mechanism, there is always an inversion of…
Q: What is a good way to test for losses during your procedure? Change all parameters during the…
A: The objective of the question is to identify the best method to test for losses during a procedure…
Q: Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing 2 the solubility…
A: Step 1: Solubility product constant (Ksp)Measure of the solubility of a compound in a solvent at a…
Q: You purchase a bottle of Sprite from a vending machine. The label states that Sprite contains 6.4 ×…
A: Concentration of bicarbonate ion (HCO3-) = pH = 4.51Pressure of CO2 in the headspace = 1.33…
Q: Draw the major organic product(s) of the following reaction. Br + NaCN DMF • You do not have to…
A: Step 1: halide is removed forming a stable carbocation. Step 2: Attack of a nucleophile on the…
Q: 2. Explain with a chemical equation, why methyl benzoate dissolves in conc. H2SO4 but not in water.
A: The objective of the question is to understand why methyl benzoate, an organic compound, dissolves…
Q: None
A:
Q: The vapor pressures of CCl4 (A) and C₂HC 3 (B) between T = 350 and 360 K, can be determined…
A: The objective of the question is to calculate-The mole fraction of in Liquid PhaseThe mole fraction…
Q: please explain your answer
A: The objective of the question is to identify and draw the structural formulas of the two…
Q: 13. LiAlH(OtBu)3 14. .COOME 15. .CN 16. 17. 8. H H DIBAL 025 OH CHO NH2 H H OH
A:
Q: If 7.94 g of CuNO3 is dissolved in water to make a 0.860 M solution, what is the volume of the…
A: The objective of this question is to find the volume of the solution in milliliters when 7.94 g of…
Q: Predict the major product of the following reaction: OH Na2Cr2O7 H2SO4, H₂O ? ○ There is no…
A: Step 1:The reagent combination Na2Cr2O7, H2SO4, and H2O is commonly known as chromic acid. It is a…
Q: analyte minimum concentration maximum concentration slope (A/C) uncertainty Caffeine 0.153 0.5067…
A: Uncertainty is the measurement of error in the graph.It can be calculated by the formula:Given Data:…
Q: Draw and label the chemical structure of the following reaction (include the curly arrow reaction…
A: The chemical reaction, including the curly arrow mechanism: H H \ / C=C / \…
Q: Copper-gold binary system is an “ideal mixture”. Use the three criteria (electronegativity, radius…
A: The objective of the question is to verify the fact that the copper-gold binary system is an ideal…
Draw the first compund in Fischer structure and 3D perspective structure( as if it was a molecular model)
Draw the second compund in Chair conformation structure and Inverted chair conformation structure
Step by step
Solved in 2 steps with 1 images

- Draw and identify the structuresof glucose, its anomers, and itsepimers, both as Fischer projectionsand as chair conformationsIndicate wether the following pairs of compounds are constitutional isomers, diastereomers, enantiomers, or the same compoundhow to draw chair conformations as consituinal isomers?
- how would u draw the lowest energy chair conformation?Which structure below shows the chair conformation of β-D-glucopyranose? The Fischer projection of this compound is shown in the box. Click on a letter A through D to answer.Represent the process using the symbol (<-> ) to indicate that the two chair conformations arein dynamic equilibrium.
- Identify each pair of structures below as enantiomers (E), diasteromers (D), or conformers (C).For each pair of structures shown, indicate hether the two species are constitutional isomers, enantiomers, diastereomers of one another, or identical moleculesLocate with an asterisk the stereogenic centers (if any) in the following structure: C. CH3CHBrCF3

