Q: How much energy is required to raise the temperature of 11.7 grams of solid tin from 21.3 °C to 35.6...
A:
Q: What is the number of moles in Serotonin if the mass in grams is 21.29
A: Given, mass of serotonin= 21.29 g molar mass of serotonin =176.215 g mol-1 moles of serotonin = ?
Q: 4. Answer the following: a) When ammonia is added to solid silver chloride, what is the metal comple...
A: The salt silver chloride(AgCl) when reacts with ammonia(NH3) ;produces diammine silver(I) chloride. ...
Q: True or False: In the Ellingham Diagram, Cu oxidation is spontaneous at all given temperatures.
A: Given statement is : In the Ellingham Diagram, Cu oxidation is spontaneous at all given temperatures...
Q: Vine goes bad soon after opening because the ethanol (CH3CH,OH) in it reacts with oxygen gas (02) fr...
A: Given : Mass of oxygen = 5.65 g Mass of ethanol consumed = ? The given reaction is shown below : CH3...
Q: An unknown compound has the following chemical formula: Al,CI where x stands for a whole number. Mea...
A:
Q: For items 11 to 15 Predict the direction of the reaction that will be favored by the system below wh...
A: Answer is as follows
Q: The molar heat of combustion, A H, of methane is -890.7 kJ mol1. If 0.0500 moles of methane is combu...
A: Molar heat of combustion of methane = -890.7kJ/mol We have 0.05moles of methane. We have to calculat...
Q: Ammonium phosphate ((NH4),PO4) PO, is an important ingredient in many fertilizers. It can be made by...
A: Given that, ammonium phosphate [NH43PO4] is an important ingredient in many fertilizers. It is prepa...
Q: Üse the References t Hint: No calculations are required. For the reaction 6CO2(9) + 6H20(1)C6H1206 +...
A: 6CO2 + 6H2O -----> C6H12O6 + 6O2 delta H° = 2.8×103 kJ delta S° = -259J/K We have to predict the ...
Q: The equilibrium constant for the reaction Na2(g) → 2Na(g) is 2.47 at 1000. K. Calculate the value of...
A: Equilibrium is the state at which the rate of forward and backward reaction are equal in a reversibl...
Q: The two molecules below are separately exposed under the given conditions. What will be the resultin...
A: Concept : Electrocyclic reaction 1) it allowed in both heat & light. 2) it is known to occur fo...
Q: Acetylene (C,H,) gas and oxygen (0,) gas react to form carbon dioxide (CO,) gas and water (H,O) vapo...
A:
Q: Plot the data above on the graph on below.
A: This is a multiple questions. According to guidelines in multiple questions we can solve only the fi...
Q: 1. The inner wall of a thermos bottle is at -10°C while the outer at 99°F. The space between the wal...
A: Since, you have posted multiple questions I will answer the first question for you. For the rest to ...
Q: Green plants use light from the Sun to drive photosynthesis. Photosynthesis is a chemical reaction i...
A: Given, Photosynthesis is a chemical reaction in which water (H2O) and carbon dioxide (CO2) chemicall...
Q: The standard enthalpy of combustion, A H; ,of graphite is -393.51 kJ moli. Calculate the change in s...
A:
Q: Identify the functional groups of the encircled item: STRUCTURE A1 #2 OH N. #1 CI STRUCTURE A2 3 HO ...
A: Given structures are : Identify the circle functional groups = ?
Q: Determine the percent composition by mass of all elements in NCl5.
A: Mass percent can be defined as the mass of an element divided by the mass of compound which is then ...
Q: . Which condition favors the forward direction of a reversible reaction? A. Q = K B. Q K D. Q = 0
A: Which of the following is correct
Q: Ammonia (NH3) chemically reacts with oxygen gas (0,) to produce nitric oxide (NO) and water (H,0). W...
A: Here we are required to find the mass of oxygen gas consumed if it would react with 2 gm of ammonia ...
Q: Which of the following is an example of a nonspontaneous process? OSolid water (ice) subliming in a ...
A: A multiple choice question based on chemical thermodynamics that is to be accomplished.
Q: 1. Draw the Lewis structure for XeF4 and CF, Where valence electrons of Xe, C and F are 8, 4, 7 resp...
A: Since you have posted more than one question, we can solve only first question for you. If you want ...
Q: c. Balanced formula equation: Ba(OH), (aq) |HCl(aq) Complete ionic equation: Net ionic equation: +
A: Write the balanced formula, complete ionic, and net ionic equations for each of the following acid-b...
Q: The mean particle size by number is? and The mean particle size by mass is? Use the given data on th...
A: Given:
Q: Question 13. For the Zn-Pb voltaic cell, what is the theoretical voltage in volts? Assume both solut...
A:
Q: Determine the maximum allowable spacing s of the nails used to hold the top flange to the web. e bea...
A: Question is solved below
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: What is the systematic name of the ff. organic compounds: STRUCTURE B1 CH3-CH-CEC-CH2-CH-CH3 CH3-CH2...
A:
Q: In order to keep as much HCHO dissolved in the water in the embalming machine reservoir as possible,...
A: For gases At low temperature solubility is more. And HCHO is a gas so to have a great solubility ,...
Q: Use the References to access important values if needed for this question. Arrange the following fro...
A:
Q: Calculate the work, in joules, done by a gas as it expands at constant temperature from a volume of ...
A:
Q: Please provide neat solution
A: Given: G°f (CO(g)) = -137.168 KJ/mol G°f (H2(g)) = 0.0 KJ/mol G°f(CH4(g)) = -50.72 KJ/mol G°f (H2O(...
Q: (f) Potassium permanganate (g) Sodium hypochlorite (h) Beryllium bicarbonate (1) Ammonium dichromate...
A:
Q: With the help of the Lewis structure of XeF4, answer to the following questions. i) Calculate the fo...
A:
Q: In the figure here, a ball is thrown up onto a roof, landing 4.90 s later at height h = 18.0 m above...
A:
Q: Gaseous ethane (CH,CH) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (Co.) a...
A:
Q: 7 5 10 20 30 40 50 60 70 80 90 100 Temperature ("C) (b) Calculate the slope of the line. (c) Determi...
A: In multipart questions, we solve only first three subparts according to the Bartleby guidelines. Fir...
Q: ve IUPAC names
A:
Q: Standard Cell Potential Pair of Half-cell Anode Cathode Cu/Cu2 and Ni/Ni2 Pb/Pb? and Mg/Mg2 Cd/Cd2 a...
A: For the cell formation the E° of the cell must be positive . So that the reaction will be become spo...
Q: Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its r...
A: Consider the given information is as follows; Initial moles of ammonia gas = 10. molInitial moles of...
Q: Ammonia (NH3) chemically reacts with oxygen gas (02) to produce nitric oxide (NO) and water (H,0). W...
A: The balanced chemical equation for the reaction of Ammonia ( NH3 ) and Oxygen gas ( O2 ) is, 4 N...
Q: 6. List the following aryl halides from most to least reactive (in suzuki coupling): 2-bromotoluen...
A:
Q: 2. Below are reactions showing bases dissolving in water. Draw a particle diagram? representing each...
A: NaOH is a strong base and NH3 is a weak base. So, NaOH will dissociate completely and NH3 will not. ...
Q: When the entropy change of the surroundings is negative, the enthalpy change for system cannot be de...
A: ∆S universe= ∆S system+ ∆S surrounding. Given that ∆S uni.= -ve. The reaction is non spontaneous. Bo...
Q: Please identify the asymmetric carborn in the following molecules and mark them with an X. There may...
A: Assymetric carbon is the one that contains four different type of atoms attached to it and that is k...
Q: 5-57 You know the mechanism of HBr addition to alkenes, and you know the effects of various substitu...
A:
Q: Write the structural formula of the followin, 1.) 3 – Fluorostyrene 2.) p- Ethyltoluene 3.) 3,5 – Di...
A:
Q: List down the intermolecular forces present in each species. HBr CS₂ C₂H6 CH3COOH Br2
A: 1) HBr is a polar molecule and having dipole. The intermolecular forces present in HBr is dipole-dip...
Q: Which of the following is an example of a nonspontaneous process? O Ammonium nitrate dissolving in w...
A: Ammonium nitrate dissolving in water Answer : it is a spontaneous process, dissolution is spontaneo...
Step by step
Solved in 2 steps with 2 images

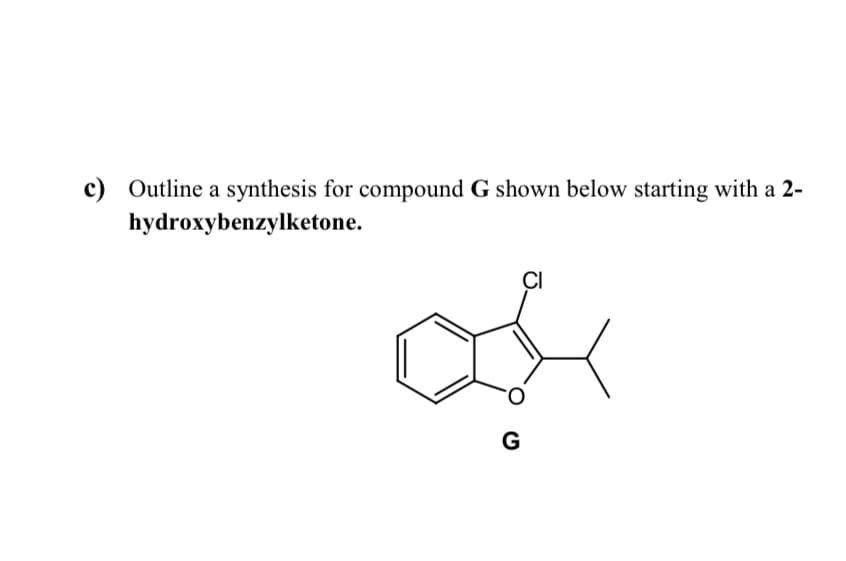
- Outline a multi-step synthesis of the compound shown below starting from benzene.(please give clear handwritten answer) Draw the structure of compound P and discuss the mechanism of the reaction of compounds N and O to give compound P, Figure 8. What is the name of this reaction?Two possibilities for a Wittig synthesis of the compound below involve using: 1)cyclohexanone and ethyl bromide or 2) bromocyclohexane and ethanal. Is either routebetter, and if so, why is one superior? Be specific and explain where the difficulty lies forthe inferior route
- Please give the appropriate reagents FOR 1, 2 AND 3 to complete the following synthesis.Propose a synthesis of molecule B from molecule A. For each step, indicate the necessary reagents.Provide a detailed mechanism for the following (Hint:halohydrin reaction). Use arrows to show electron flow and show intermediates.



