Q: p) Explain why the following reaction is endothermic based on pur knowledge of both bond enthalpy an...
A:
Q: Lewis structure name of highlighted group H :0 С — н H :0 — Н H :0: H -C- N - H н н - -
A:
Q: The reaction 2 C4H10(s) + 13 O2s) - has AG° = -5407 kJ. Determine the value of AG°; for C4H10(8). 8 ...
A: Given reaction is : 2 C4H10 (g) + 13 O2 (g) → 8 CO2 (g) + 10 H2O (g)and ∆Go = -5407 KJDetermination...
Q: Aqueous sulfuric acid (H,SO,) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium su...
A: Sulfuric acid reacts with sodium hydroxide to form sodium sulfate. The equation for the balanced che...
Q: Rola... File Edit View Insert 100% Normal text Arial ARLES' LAW A balloon, originally at 6.28 L and ...
A: since you have posted more than one question, we can solve only first for you. If you want other tha...
Q: Please identify the asymmetric carborn in the following molecules and mark them with an X. There may...
A: Asymmetric carbon centre is the carbon center with sp3 hybridised and having four different valencie...
Q: 6. List the following aryl halides from most to least reactive (in suzuki coupling): 2-bromotoluen...
A:
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: The two molecules below are separately exposed under the given conditions. What will be the resultin...
A: Concept : Electrocyclic reaction 1) it allowed in both heat & light. 2) it is known to occur fo...
Q: While touring pressurized water reactors, visitors are often granted access to the steam turbine gen...
A: In pressurized water reactors the heat transfer from core to turbines takes place in two different s...
Q: How many moles are in 4.75 x 1024 molecules of HCl?
A:
Q: 7. What is the uncertainty of the momentum of an electron if we know its position is somewhere in a ...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A:
Q: RLES' LAW A balloon, originally at 6.28 L and 211K has its temperature increased to 419K. What will ...
A:
Q: Current Attempt in Progress The equilibrium constant for the reaction Na2(g) → 2Na(g) is 2.47 at 100...
A: the relation of ∆G0 with equilibrium constant (K) is ∆G0=-RTlnK Given K=2.42 temperature(T)=1000 k ...
Q: What is the volume (liters) of 23.5 g of I2 at STP?
A:
Q: An ideal gas in a cylinder fitted with a piston expands at constant temperature from a pressure of 4...
A:
Q: 2. Write suitable detailed mechanisms to account for the formation of the products in the following ...
A: We have to give the mechanism for the formation of the product.
Q: When the entropy change of the surroundings is negative, the enthalpy change for system O will be po...
A: Enthalpy, entropy, Gibbs free energy change are some thermodynamic parameters. They are related as b...
Q: thermometer. A 52.4 g sample of aluminum, which has a specific heat capacity of 0.897 J'g.°C, is put...
A: Given, Mass of Al = 52.4 g Specific heat capacity of Al = 0.897 J.g-1.0C-1 Mass of water = 250.0 ...
Q: 5:36 Rank the compounds in each group according to their reactivity toward electrophilic substitutio...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A: 1.14 g of sodium bromide is produced from the reaction of 3.2 g of hydrobromic acid and 2.7 g so...
Q: (f) H,SO, (g) HNO, (h) HC10, (i) H,SO;
A: Anion should be name with end -ate Cation should be named as such.
Q: 1) 2-octane has the mass spectrum shown. Propose structures for fragment ions of m/z 99, 85, 71, 57,...
A: 2-octane has the mass spectrum at fragment ions of m/z 99, 85, 71, 57, 43 and 29. We have to Propo...
Q: List down the intermolecular forces present in each species. HBr CS₂ C₂H6 CH3COOH Br2
A: 1) HBr is a polar molecule and having dipole. The intermolecular forces present in HBr is dipole-dip...
Q: 1. A 10 L balloon contains helium gas at a pressure of 655 torr. What is the new pressure when the v...
A: Boyle's law: According to Boyle’s law, the absolute pressure exerted by a given mass of an ideal ga...
Q: 4. Calculate the pressure exerted by 3 moles of oxygen gas, O2, confined to a volume of 510 mL at 10...
A: Consider the given information is as follows; Moles of oxygen gas (O2) = 3 mol Volume (V) = 510 ...
Q: 1. A 10 L balloon contains helium gas at a pressure of 655 torr. What is the new pressure when the v...
A: 1. Given that, a 10 L balloon contains helium gas at a pressure of 655 torrs. We have to calculate t...
Q: Hydrofluoric acid, HF, has a K, of 6.8 x 10. What are [H,O* ]. [F], and [OH]in 0.960 M HF? [H,O*]-0....
A:
Q: Which of the following is an example of a nonspontaneous process? O Ammonium nitrate dissolving in w...
A: Ammonium nitrate dissolving in water Answer : it is a spontaneous process, dissolution is spontaneo...
Q: the precipitate to be obtained will be more likely colloidal if the computed relative supersaturatio...
A: Precipitation can be defined as the process of conversion of a dissolved substance into an insoluble...
Q: Suppose a 250. mL flask is filled with 0.80 mol of NO and 2.0 mol of NO,. The following reaction bec...
A:
Q: Identify the product in each of the following reactions: Nal (1 mol) (a) Cl, C5H10CII acetone Cl SNa...
A: Given data :-
Q: methyl group.
A:
Q: 7. Consider the following mechanism and reaction coordinate diagram. Step 1: A(g) + B(g) → AB(g) 60T...
A:
Q: An ideal gas in a cylinder fitted with a piston expands at constant temperature from a pressure of 8...
A:
Q: Make a general statement on the effect of solids upon the freezing point of liquid in which they are...
A:
Q: Essential oils are concentrated liquid containing metabolites extracted from plant sources and are w...
A: A question based on properties of liquids that is to be accomplished.
Q: Which would likely increase the rate of a chemical reaction? A. Cooling the reaction mixture. B. Dec...
A:
Q: Nitrogen dioxide is one of the many oxides of nitrogen (often collectively called "NOx") that are of...
A:
Q: Calculate the pH of a 0.10 M HCOOH solution. Ka if HCOOH is 1.8x10-4 a. 5.78 b. 2.38 c. 5.42 d. 3...
A:
Q: View Policies Current Attempt in Progress Is the following reaction spontaneous at room temperature ...
A: For a spontaneous reaction, ∆S of a reaction should be positive.
Q: Lewis structure name of highlighted group H H н— о С — С — н H H H : 0— С — н | H :0-H H :0: H -C. N...
A:
Q: Suppose a 500. mL flask is filled with 1.3 mol of Cl,, 1.4 mol of HCl and 1.6 mol of CCl. The follow...
A: Given, Cl2(g) + CHCl3(g) ⇌ HCl(g) + CCl4(g) The equilibrium constant, Keq = 0.723 Volume of the fl...
Q: Which of the following equations is a mathematical definition of enthalpy? Ο ΔΗ- ΔΕ-ΡAV Ο ΔΗ-ΔΕ + ΡA...
A: enthalpy is the sum of the internal energy and the product of the pressure and volume of a thermodyn...
Q: What is the molality of a solution containing 85.3 g of silver perchlorate (A9CIO4) dissolved in 932...
A: Mass of silver perchlorate (AgClO4) = 85.3 gram Molar mass of silver perchlorate = 207.319 g/mol Mas...
Q: A gas has the initial conditions of: 21.2 Liters • 279 K 1.03 atmospheres If its pressure is increas...
A:
Q: The reaction 2N20(g) 2N2(3) + O2(g) has AH° = -163.14 kJ. What is the value of AE for the decomposit...
A:
Q: Use activity coefficients to calculate the pH after 10.0 mL of 0.110 M trimethylammonium bromide wer...
A: Solution Given that 10.0 mL of 0.110 M trimethylammonium bromide 5.0 mL of 0....
Q: The ratio of effusion of helium to another noble gas element is 3.16:1.00. What is the other element
A: Noble gases are the elements of group 18. Group 18 contain sevent elements helium, neon, argon, Kryp...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

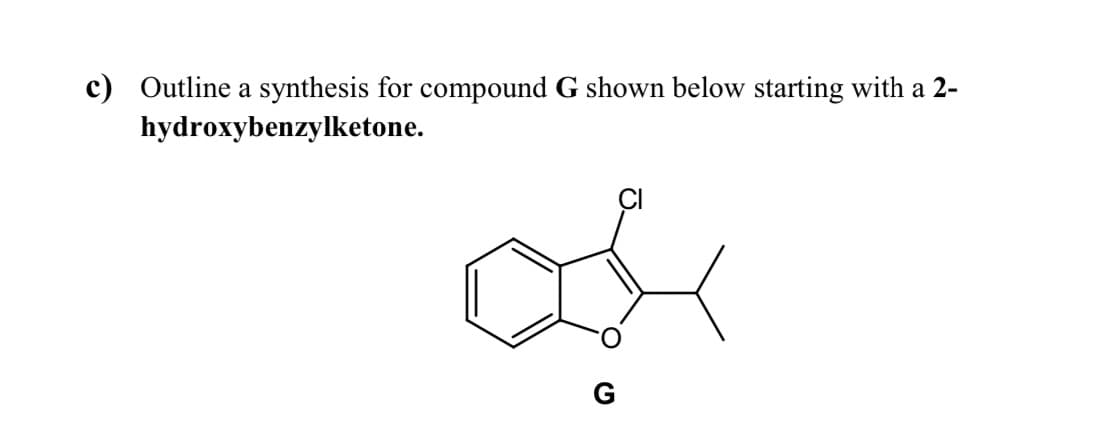
- Write a mechanism for the reaction below. Hint: think retro!Using only organic reagents of 4 carbons or fewer, as well as any necessary solvents/inorganic reagents, propose a synthesis of the molecule below. Assume all products can be separated easily. If more than one product is to be expected in any step, indicate which product will be taken further.Elaborate an efficient synthesis of the molecule below starting from benzene (your choice). Mechanisms are not required
- Outline a multi-step synthesis of the compound shown below starting from benzene.Propose a synthesis of the compound below using only ethyne, methyl bromide, and methanol as sources of carbon. Is the product optically active? ExplainAfter E2 elimination, will this product contain deuterium (D)? Justify your answer.
- Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each stepshobha Propose a plausible mechanism for each of the reactions below with clear arrow pushingwrite out a complete mechanism for the reaction below

