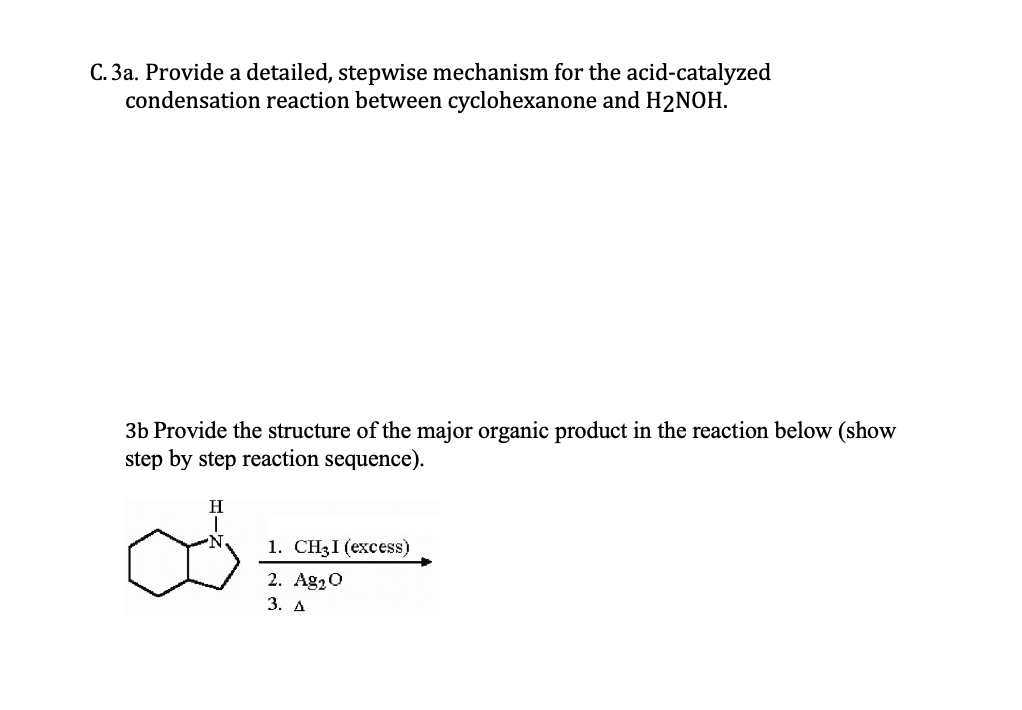
C. 3a. Provide a detailed, stepwise mechanism for the acid-catalyzed condensation reaction between cyclohexanone and H2NOH. 3b Provide the structure of the major organic product in the reaction below (show step by step reaction sequence). H 1. CH31 (еxсess) 2. Ag20 3. А
Q: Suggest short, efficient reaction sequences suitable to produce the compound from the given starting…
A: We have to carry out the required transformation.
Q: plete the synthetic sequences by drawing products/substrates/reagents in empty spaces in reactions…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: NaOCH3 + Naci CH;OH The nucleophilicity of the methoxide anion (CH30") is reduced in protic solvent…
A: For E2 elimination reaction strong base ,beta hydrogen with respect to good leaving is required and…
Q: PS#3 part 2- Synthesis: Given this group of simple compounds, devise a plan to synthesize each…
A: We have to snthesize the following given compounds from the given starting materials in multistep…
Q: 6. 2-methyl-1,1-diphenylpropene can be prepared by Wittig reaction. a) Suggest any suitable carbonyl…
A:
Q: a. NABH4 then H3o+ b. PBr3 c. Mg in dry ether, then CH2=O, then H30* d. Dess-Martin Periodinane e.…
A: The question is based on the concepts of Organic reactions. we have been given certain options. we…
Q: Using your reaction roadmap as a guide, show reagents and conditions to bring about these…
A:
Q: 1. HNO3, H,SO, 2. Fe, HCI 3. NANO,, HCI 4. HBF, 5. KMN04, A CH(CHS2
A:
Q: 3. A. Provide the major organic product of the reaction below and a detailed, stepwise mechanism…
A: The mechanism for the reaction between given molecule and methanol in the presence of heat is shown…
Q: Predict the two most likely mechanisms for the reaction of 2-lodohexane with sodium ethoxide. OA SN2…
A:
Q: Select the final product from the following reaction sequence: 1. NBS 2. KOTBU 3. H,O, catalytic…
A:
Q: 6) Consider the following reactions scheme below. NNH- H. HgCB Step II Step I HO Step II H HCN…
A:
Q: 3. Consider the following carboxylic acid (propanoic acid) and answer the question that follows:…
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: to CH;CH,OH Br Major Minor Nucleophilic substitution mechanism:
A:
Q: Propose structural formulas for compounds A, B, and C in the following conversion. Also show how to…
A: Concept: The study of the chemical processes occurring in living matter is called biochemistry. It…
Q: Suggest short, efficient reaction sequences suitable to produce the compound from the given starting…
A: Benzyl methyl ether can be prepared by the reaction of benzaldehyde dimethyl acetal and…
Q: 1. O3 2. HOẶC
A: The product of the above organic transformation is given below
Q: II (a) I= a, B-diketone, II = a, ß-diester, III = B-keto nitrile (b) I = B-diketone, II = B-diester,…
A: In this question, we will Identify the class of all three Compounds. You can see details Solution…
Q: In the reaction scheme below, the furan A is converted to the thiophene B via a diketone. Write a…
A: Formation of a Diketone:
Q: following
A:
Q: 6. In an organic chemistry lab, chemists were attempting to convert compound I into compound II via…
A:
Q: Answer ALL parts of the question (a) Predict the organic products formed when 3-methoxybenzaldehyde…
A: Applying concept of canonizarro reaction.
Q: POD 9: 02-10-21 Provide the Stepwise synthesis for the following transformation
A: In this transformation, from a saturated lower analog, saturated higher analog is formed. Thus it…
Q: Choose the best reagent(s) for carrying out the following conversion from the list provided below.
A: In the given problem primary alcohol is being converted to aldehyde . It oxidation of primary…
Q: 2. Selectivity. Show the reaction intermediates obtained after each step and the major product…
A: The details explanation of the organic reaction is given below
Q: The following reactions have been used to synthesize dieldrin and aldrin (named for Diels and…
A: This suggest that this reaction would be diels alder reaction and the molecule X is an alkene,…
Q: Provide a plausible arrow pushing mechanism for the reaction below. You may abbreviate the…
A: This is an example of Diels-Alder reaction followed by cyclization and aromatization.
Q: (a) A useful synthesis of some diols includes the reaction of "bis-Grignard" reagents (compounds…
A: Organic reactions:
Q: I. Write the name for the mechanisms separately through which product A and product B are formed.…
A: The answers are given step by step below
Q: Draw the structure of the triphenylphosphonium salt and Wittig reagent formed from each chloroether.
A:
Q: Using acetylene and 2-methylpropane as your only sources of carbon atoms, propose a plausible…
A: Here we are required to predict the reagent used for conversion of given starting material to…
Q: (a) Give starting materials (including molar ratios) for synthesis of compound Z. Ph EIO2C OEt F3C…
A: This is an example of Hantz pyridine synthesis.
Q: H2 Lindlar catalyst -) 12. 1. 1 eq HCI 2. 1 eq HBr 13. 1. disiamylborane 2. H2O2, NaOH 14. Use an…
A: Note: As per the bartleby rules, one has to only answer starting 3 part if there are more than 3…
Q: 6. Complete the following reaction scheme (а) hv Br2 (b) hv NBS ÇCL4, (a solvent) (c) H2 Pd/C
A:
Q: When anisole (methoxybenzene) is chlorinated the products are 65% 4-chloro anisole and 34.9% 2-…
A: D is the correct reason to explain this.
Q: 1.b. Acidity of Phenols – Reaction with NaHCO3 Test Sample Description Of Mixture Inference Slightly…
A: Reaction with NaHCO3 = 1) Phenol = Reaction of phenol with NaHCO3 is,
Q: OH so,H So, Na* O Na* NaOH NaOH H,0* H,0 350°
A: Given, Benzene Sulfonic Acid to Phenol
Q: 8. Organometallic reagents, such as organolithium (RLi), Gilman or Organo-cuprates (R2)CuLi) and…
A: LiCH3 is the organolithium compound that releases CH3 as a nucleophile which attacks the carbonyl…
Q: Provide a plausible arrow pushing mechanism for the reaction below. You may abbreviate the…
A: The reactions follow (2π+4π ) cyclo addition Diels elder reaction. The first step shows Diels -alder…
Q: CH3 CH3 CH3 CH3 CH3 CH3 (1) (2) (3) (4) (5) `NO2 `NO2 NO2 NO2 NH2 NHCOCH3 NHCOCH3 NH2 N2* From the…
A:
Q: Which structure is a reasonable intermediate for this reaction? (HA = generic acid) 1) CH3MGB…
A: The above reaction is the addition of grignard group to the ester. In the Reaction RMgX is used as…
Q: Provide the major organic product of the reactions shown below. Show the from each step shown below.…
A: ->Na2Cr2O7 is oxidizing agent which can oxidize primary alcohol to carboxylic acid.…
Q: . The following diagram represents a synthesis which begins with bromoethane. (i) (iii) C2H6O (C)…
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Choose the best reagent(s) from the list provided below for carrying out the following reaction…
A:
Q: The structural formulae of wWo compounds X and Y are given below: OCH(CH,)2 Y a) Write a reaction…
A: The question is based on the concept of organic reactions. we have to convert one compound into…
Q: Alkane A, shown below, reacts with bromine in a radical substitution reaction. CH3 f CH3 Alkane A…
A: Monosubstitution of alkane forms a mixture of organic products. Free-radical bromination of alkane…
Q: Show how you would prepare the following from the indicated starting materials. More than one step…
A:
Q: A key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve…
A: The above conversion can be carried forward by a famous cross-coupling reaction known as…
Q: Below is a selection of reactions; you may not need all of them. Write the letters of the needed…
A:
can i get help with this problems
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Acid Halide Preparation reaction mechanism: Please use HCl and SOCl2 as reagents(a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acidBenzene, C6H6 undergoes substitution reaction with concentrated nitricacid, HNO3 to produce compound L. The reaction of compound L withbromine, Br2 in the presence of iron tribromide, FeBr3 produced compoundM. Benzene also undergoes Fridel-crafts alkylation reaction withchloroethane, CH3CH2Cl using catalyst N to produce compound P (i) Draw the structural formula of L, M and P (ii) State catalyst N. (iii) Show the formation of electrophile that will be reacted with benzene for theformation of compound P.
- give the reagents for parts a-pIn this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?3 Give mechanisms for the acid-catalyzed and base-promoted alpha-halogenation ofketones. Explain why multiple halogenations are common with basic catalysis andgive a mechanism for the haloform reaction
- A key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo-6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used tocarry out this step.Complete the reaction schemes below providing the reagents required to achieve thetransformation. More than one step may be necessary for each scheme.1. Esters can be synthesized from the nucleophilic substitution reaction of anhydrides and alcohol in the presence of a base give a complete mechanism and the final product for the reaction below 2. using appropriate illustrations, explain what is meant by the tetrahedral transition state( also indicate the geometry for the starting material and product).

