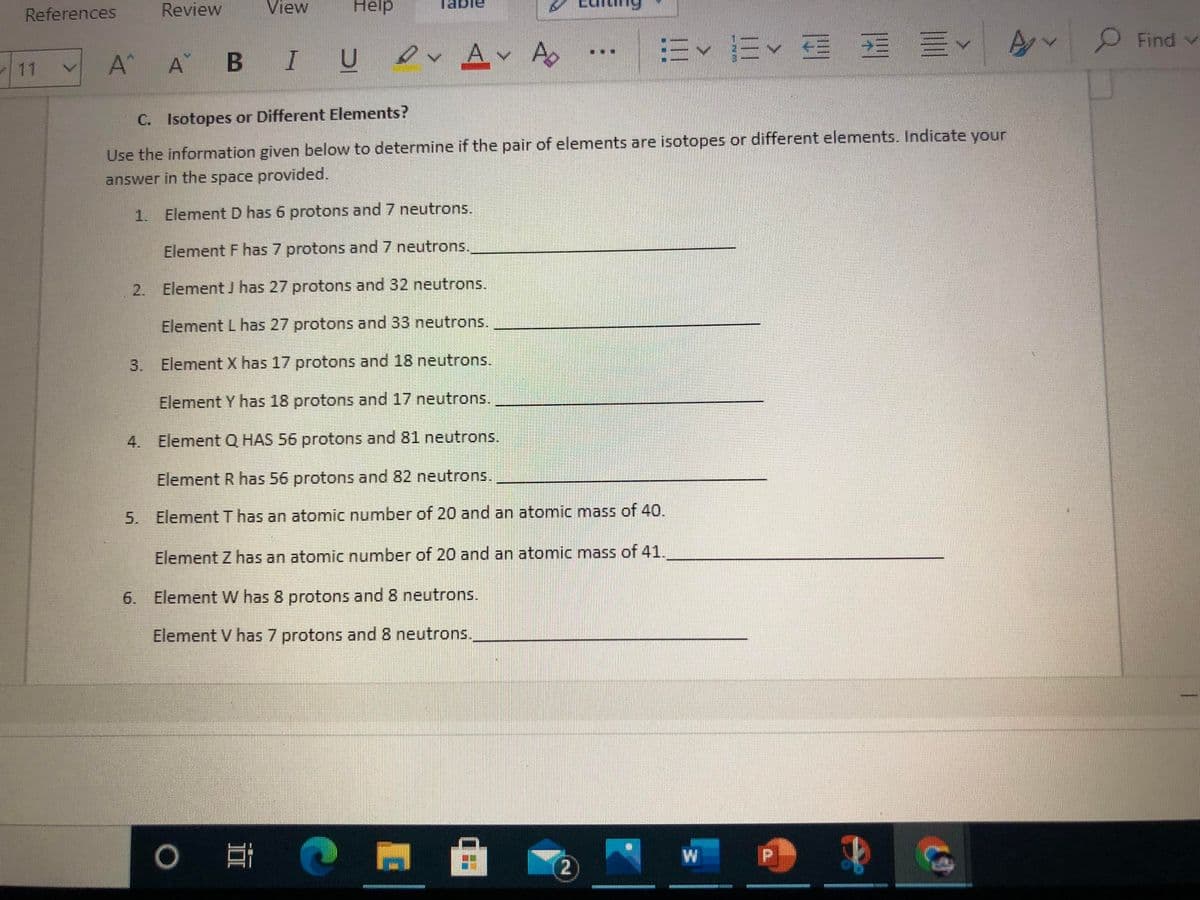
C. Isotopes or Different Elements? Use the information given below to determine if the pair of elements are isotopes or different elements. Indicate your answer in the space provided. 1. Element D has 6 protons and 7 neutrons. Element F has 7 protons and 7 neutrons. 2. Element J has 27 protons and 32 neutrons. Element L has 27 protons and 33 neutrons. 3. Element X has 17 protons and 18 neutrons. Element Y has 18 protons and 17 neutrons. 4. Element Q HAS 56 protons and 81 neutrons. Element R has 56 protons and 82 neutrons. 5. Element T has an atomic number of 20 and an atomic mass of 40. Element Z has an atomic number of 20 and an atomic mass of 41. 6. Element W has 8 protons and 8 neutrons. Element V has 7 protons and 8 neutrons.
C. Isotopes or Different Elements? Use the information given below to determine if the pair of elements are isotopes or different elements. Indicate your answer in the space provided. 1. Element D has 6 protons and 7 neutrons. Element F has 7 protons and 7 neutrons. 2. Element J has 27 protons and 32 neutrons. Element L has 27 protons and 33 neutrons. 3. Element X has 17 protons and 18 neutrons. Element Y has 18 protons and 17 neutrons. 4. Element Q HAS 56 protons and 81 neutrons. Element R has 56 protons and 82 neutrons. 5. Element T has an atomic number of 20 and an atomic mass of 40. Element Z has an atomic number of 20 and an atomic mass of 41. 6. Element W has 8 protons and 8 neutrons. Element V has 7 protons and 8 neutrons.
Chapter3: Atoms And Elements
Section: Chapter Questions
Problem 3SC
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
Transcribed Image Text:References
Review
View
Hélp
O Find v
A^
A B IU
evAv A
11
C. Isotopes or Different Elements?
Use the information given below to determine if the pair of elements are isotopes or different elements. Indicate your
answer in the space provided.
1. ElementD has 6 protons and 7 neutrons.
Element F has 7 protons and 7 neutrons.
2. Element J has 27 protons and 32 neutrons.
Element L has 27 protons and 33 neutrons.
3. Element X has 17 protons and 18 neutrons.
Element Y has 18 protons and 17 neutrons.
4. Element Q HAS 56 protons and 81 neutrons.
Element R has 56 protons and 82 neutrons.
5. ElementT has an atomic number of 20 and an atomic mass of 40.
Element Z has an atomic number of 20 and an atomic mass of 41.
6. Element W has 8 protons and 8 neutrons.
Element V has 7 protons and 8 neutrons.
W
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co