Q: By referring to the Appendix I, calculate and arrange the percentage of ionic character for the foll...
A:
Q: (b) Describe in brief the origin of isomer shift in Mössbauer spectroscopy. Fe2+ possesses a more po...
A: Isomer shift arrises due to diffarence in electron density around the nucleus. It can be determined...
Q: The element aluminum forms a a with the charge
A: The element aluminium forms a cation with the charge +3. The symbol for this ion is Al+3 with the na...
Q: (c) How many signals would you expect in 13ć-NMR of the following compounds? OMe OMe (A) (B}
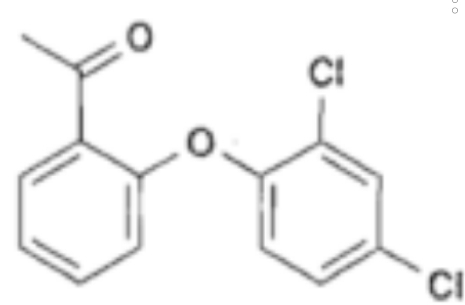
A: Chemically different carbon atoms show different peaks in 13c -NMR spectrum
Q: In a sterilizing retort, cans of food products were heated. The average temperature in the center of...
A: Sterilization is process used in destroy or inactivate the microorganism by heating the food product...
Q: 2. Aurumn (III) chloride, (AuCl3), has a solubility of 54 g/100 mL of water. Determine the solubilit...
A: Mol. Mass of AuCl3 = 303.5g/mol Solubility of AuCl3 is 54g/100ml We have been asked to find the solu...
Q: (3 points) Draw the products of the following acid-base reaction. You can just modify the starting a...
A:
Q: 1.) Is acetone a london disperse force, dipole dipole, or a hydrogen bonds? Explain. 2.) Is vinegar...
A: There are different types of forces act between the molecules:- Hydrogen bonding Dipole-dipole attr...
Q: Calculate the amount of heat needed to melt 2.00x10 g (35.81 mol) of iron at its melting point (1,80...
A: Given , Moles of iron =35.81 mol Melting point of iron =1809 k ∆H(fus) =13.80 kj/mol We have to c...
Q: Identify the conversion factors needed to convert between the number of moles and the mass of an ele...
A: To identify: the conversion factors needed to convert between the number of moles and the mass of an...
Q: Reagent Classifications: (USP, CP, AR, TG, and others) Kindly describe briefly EACH of the items ab...
A:
Q: 11) Drawing (1) shows a nonequilibrium system comprised of pure water separated from an aqueous solu...
A: The representation of system given initially is,
Q: A sample of a chemical compound contains 8.56 g of carbon and 1.44 g of hydrogen. What is the empiri...
A: The formal charge of an atom in a molecule can be defined as the charge residing on the atom if all ...
Q: . To what temperature must 15 L of oxygen gas at 0oC be heated at 1 atm pressure in order to occupy ...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: Trial 1 Tria AVERAGE DENSITY OF METAL g/cm? STANDARD DEVIATION g/cm Sample number nono
A: The densities of metals which are given in three trials are 9.420, 9.949 and 9.924 g/cm3. So, t...
Q: The mass of a 200Hg atom, as determined by experimental means, is 199.968316 u. Find the binding ene...
A:
Q: Utilizing skeletal structures, draw as many aldehydes as possible using the formula: C5H10O.
A: In the aldehyde functional group, a carbon atom is attached with one oxygen atom by a double bond, a...
Q: Be sure to answer all parts. Rank the following groups in order of decreasing priority: A. CH CH2 В....
A: According to IUPAC the priority order should be: Hydrogen<Alkane<Alkyne<Alkene
Q: tBu water tBu PdCl, methanol OH CHCI, NaOH, water MeN LDA, THE, 0°C
A:
Q: Provide the proper number of 'H NMR signals (peaks) for the following compound. HO
A: We have to predict the number of peaks for given compound.
Q: For the following reaction, if the rate of disappearance of O2 is 0.0450 molL1s1, what is the rate o...
A: The average rate of a reaction is defined as the rate of change of concentration of a reactant or a ...
Q: 2 trans-[Co(en)2C12]Cl is a chiral compound
A: First two statements is true but last statement is false.
Q: 1. Consider the 'H NMR spectrum shown below. - The chemical shifts in combination with the integrati...
A:
Q: is Is, 22p", 3 3p . The electron configuration of Select one OA Ga OB Ca OCK OD Ge OE A The name of ...
A: Electronic configuration of an atom can be defined as the distribution of electrons in orbitals of a...
Q: 1. Consider the equilibrium N;O4 (g) = 2NO, (g) AH° = 58.0 kJ In which direction will the equilibriu...
A: Since you have posted multiple questions, we will solve first question for you. To get remaining que...
Q: Which molecule would produce the spectra below? 11 10 1 200 180 160 140 120 80 60 40 20 ppm -CH3 o-C...
A: Given are NMR spectra
Q: What is the correct IUPAC name for the following A) Agl B) Al2S3 C) SnS2 D) Fe3P2 E) Al2(SO3)3 ...
A: IUPAC naming is done according to rules set by IUPAC
Q: a) Draw the mechanism for the following reaction. HN H3C H;C. NaBH;CN CH3 CH3 b) draw the mechanism ...
A: Mechanism of following nucleophilic attack on carbonyl carbon electrophile is shown below.
Q: Complete the following for the compound copper(I) sulfite . formula = Cu2s03 atom number in formula ...
A: The number of atoms in the given compound Copper (I) sulfite.
Q: b) Consider (Co(CI)6 and (Co(NH3)e]* ions. i. Illustrate the energy level splitting diagram for both...
A: Solution is given in next step
Q: 2) a) A 2.60 L sample of water contains 195.0 ug of lead. Calculate the concentration of lead in ppm...
A:
Q: Example: Using activities, find the concentration of OH in a solution of 0.075M NaCIO, saturated wit...
A: Given in question: Ion size (a) of OH- = 350 pm Ion size(a) of Mn2+ = 600 pm Concentration of soluti...
Q: The number of moles of water in a hydrated compound is calculated to be 0.04 moles. After heating th...
A:
Q: K3[Cr(OH)6] + H2O2 K2Cr0, + H2O + KOH
A: The reaction takes place in basic medium
Q: important is it to embed the 'emitter recoilness emission of a Y-ray. How The Mossbauer effect depen...
A: Emitter nucleus is embedded in a bulky, rigid crystal lattice to ensure that energy don't get lost i...
Q: How many signals would you expect to see in the proton NMR spectrum of the following compound? Pleas...
A:
Q: The Newman of 2,2,3-trimethylpentane is shown in the box. Predict the number of signals in 1°C NMR. ...
A: Number of signals in 13C NMR :- Chemically equivalent carbon will give same signal and chemically no...
Q: lowest freezing point
A:
Q: 26-30. About how old is a piece of wood from an aboriginal burial site that has a carbon-14 (half-li...
A: The carbon-14 initial activity rate, Ao = 15.6 counts per minute per gram The carbon-14 activity rat...
Q: Which of the following compounds is responsible for this 'H NMR spectra? [Hint: use the grid to dete...
A:
Q: b) Consider (Co(CI).F and (Co(NH3)e]* ions. i. Illustrate the energy level splitting diagram for bot...
A: Since Cl is weak field ligand then the complex shows high spin And NH3 is strong field ligand shows...
Q: 21. What is a major product of the reaction shown in the box? Br Pd-cat. ? Base H NMR of the major p...
A:
Q: draw 2 potential mechanisms for the etherification of ethyl vanillyl alcohol (3-ethoxy-4-hydroxybenz...
A: Etherification is the reaction between two alcohols in the presence of an acid which acts as a catal...
Q: d ! of the following isotopes is expected to be NMR inactive? b) 170 a) 2C viup c) 31p d) "F Q4: Con...
A:
Q: When salt dissolves, it can form either acidic or basic salt solutions. Calcium chloride, CaCl, is a...
A: CaCl2 i.e. calcium chloride which is a salt of strong acid ( HCl) and the Strong base Ca(OH)2 . Th...
Q: Consider mixing an aqueous solution of iron(III) chloride with an aquoues solution of potassium hydr...
A: Given, Mixing of an aqueous solution of iron(III) chloride with an aquoues solution of potassium hyd...
Q: The following are the recommended doses for a certain nicotine patch. Determine the overall grams of...
A: The doses given are, • One 21 mg dose per day for 6 wks• One 14 mg dose per day for 2 wks• ...
Q: ) trans-[Co(en)2Cl2]Cl is a chiral compound ) The coordination number for the anionic complex in K3[...
A: Chiral compound is defined as a compound which can not be superimposed on its mirror image.
Q: 1.) How long will it take for one drop of water to evaporate? 2.) How long will it take for one d...
A: 1. Answer - It will take 1-2 minute for one drop of water to evaporate . 2. Answer - It take 30 s...
Q: chemical formula
A:
Step by step
Solved in 2 steps with 2 images


