Q: Show a detailed arrow pushing mechanism for the reaction of the Aryl Diazonium Salt with an electron…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 2. Propose starting materials and reagents to achieve the following synthesis. Assume the circled…
A: Step 1: Choosing the Starting MaterialThe desired product,…
Q: Show work, thank you!
A:
Q: 3 PHASE DIAGRAMS. Sketch the temperature-composition (“T-x") phase diagram for acetone, (CH3)2 CO),…
A: To sketch the temperature-composition (T-x) phase diagram for the acetone-chloroform mixture, we…
Q: For thefollowing substances, tell whether each is a nonelectrolyte, weak electrolyte or a strong…
A: Magnesium hydroxide in water: Slightly solubleMagnesium hydroxide is a sparingly soluble ionic…
Q: A sample of neon gas at a pressure of 0.561 atm and a temperature of 24.8 °C, occupies a volume of…
A: The objective of the question is to find the pressure of a sample of neon gas after it is compressed…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: • A substance has a vapor pressure of 55.1 mmHg at 35 °C and a nor- mal boiling point of 116.9 °C.…
A: To find the enthalpy of vaporization (∆Hvap) for the substance described, we can use the…
Q: Question 17
A:
Q: Give a balanced reaction equation for the reaction between Ni(NH3)62+ and HCl
A: The reaction between Ni(NH3)62+ (hexaamminenickel(II) ion) and HCl (hydrochloric acid) involves…
Q: For 1 and 2, show the products of the following reaction:
A: Step 1:Step 2:
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients…
A: To balance the given equation under basic conditions:1. Balance the chromium atoms: Cr(OH)₃ + Ag₂O…
Q: Use this information to answer each question: A student constructs an electrochemical cell with a…
A: To calculate the experimental standard state free energy change, ΔGo, we can use the following…
Q: PHASE DIAGRAMS The vapour pressure of pure liquid CCl4 is 23 kPa and its Henry's law constant in…
A: Step 1: Vapour pressure of pure liquid CCl4 is,pA=23kPa(at290K)Vapour pressure of pure liquid Br2…
Q: Br 1. N3 2. LiAlH4 3. H₂O NH₂ 26 Preparation of primary amines using azide ion avoids the problem of…
A: Step 1:Step 2:Step 3:Step 4:
Q: Enter electrons as e". Use smallest possible integer coefficients. If a box is not needed, leave it…
A:
Q: Indicate how to synthesize 5-Methyl-3-hexanone from 1,3-dithiane.
A: The objective of this question is to outline a method for synthesizing 5-Methyl-3-hexanone from…
Q: dont provide handwritinhg solutiin...............
A: Step 1: An organic substance called methyl cyano acrylate has three different functional groups: an…
Q: Please don't upload any image just give me the answer with proper explanation
A: Approach to solving the question:Read alot of related books or research onlineDetailed…
Q: The pressure in Denver, Colorado (5280 ft elevation), averages about 24.5 in. Hg. Convert this…
A: The objective of this question is to convert the pressure from inches of mercury (in. Hg) to…
Q: Question 14
A: The balanced equation for the reaction of zinc with hydrochloric acid is:Zn(s) + 2HCl(aq) →…
Q: A nuclear power plant operates at 40.0 % efficiency with a continuous production of 1182 MW of…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: Which of the following is the correct expression Qrxn for the reaction occurring within this cell?…
A: The objective of the question is to determine the correct expression for the reaction quotient…
Q: 4) i) Fill in the reagents to complete the synthetic sequence below, and draw the final product in…
A:
Q: None
A: Step 1: an electrocyclic reaction can either be classified as conrotatory or disrotatory , based on…
Q: CH3 CH3 CH3 O-H NaOH heat Drawing >
A:
Q: Label the following as: SN1, SN2, E1, E2 reactions. Create examples with drawn mechanisms.
A: Step 1: Step 2: Step 3: Step 4:
Q: A 'H NMR spectrum is shown for a molecule with the molecular formula of C5H10O2. Draw the structure…
A:
Q: According to the HONC rule, oxygen atom has a neutral formal charge , when it forms 2 bonds around…
A: The HONC rule is a simple way to remember how many bonds are generally formed by the atoms of…
Q: What is the chemistry of how spf blocks uv rays from the sun Please please please answer as fast as…
A: Sunscreen works by shielding the skin from the harmful effects of ultraviolet (UV) radiation, which…
Q: match the correspoding molecule to IR spectrum. ( only on molecule per spectrum)
A: Spectrum E:shows band at 1725 cm-1 which is for C=O stretching, hence compound should have carbonyl…
Q: A sample of krypton gas at a pressure of 794 mm Hg and a temperature of 71 °C, occupies a volume of…
A: The objective of the question is to find the volume of a sample of krypton gas when it is cooled…
Q: Give a full curved arrow pushing mechanism for the following reaction. You can use the abbreviated…
A: Step 1: This is imine formation reaction. In this reaction cyclohexanone reacts with ammonia in the…
Q: QUESTION 9 How many grams of solute are needed to make 1300 mL of a 4.5% (m/v) NH4Cl solution? Do…
A: 9). - Calculate the mass of solute using the formula for mass/volume percent; % (m/v) = [mass of…
Q: 1) Using any reagents necessary (Cn, n<5 incorporated into final product), develop a synthesis for…
A: Step 1: Step 2:Hope you understand Please do rate ..... Thankyou
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: A 250.0-mg sample of an organic weak acid is dissolved in an appropriate solvent and titrated with…
A: The objective of this question is to determine the equivalent weight of an organic weak acid that…
Q: Question 14 Propose best Williamson ether syntheses for the following compounds. Select all…
A:
Q: From the following reaction and thermodynamic data, find S° of SOCI, and the T at which the reaction…
A: The objective of the question is to find the entropy (S°) of SOCl2 and the temperature (T) at which…
Q: None
A: Step 1:
Q: N HO-CH2 OH P-O-CH2 O_ N H H H H OH OH NH NH O nucleoside O nucleotide O nucleic acid O nucleic base…
A: Step :
Q: 2
A: For a molecule to be anti-aromatic, it must:be cyclicbe planarbe fully conjugatedhave 4n π electrons…
Q: Chemistry
A: The freezing points of the given substances are as follows: H₂O (Water): The freezing point is…
Q: Question 16
A: Step 1:Limiting reagent is the reactant that exhaust first in the chemical reaction and decides the…
Q: Predict the major products of this reaction. Cl₂ hv ? Draw only the major product or products in the…
A:
Q: 4) An undergraduate researcher, James, was given the task of removing a t-butyl ether from the…
A: The proposed reaction mechanism explains why James obtained the ortho product (C) instead of the…
Q: Most common amines (RNH2) exhibit pKa values between 35 and 45. R represents the rest of the…
A: The presence of a cyano group (-CN) introduces significant electron withdrawal due to its strong…
Q: None
A:
Q: Incorrect Your answer is incorrect. Tag the carbons in this molecule that are bonded to the H atoms…
A: Thank you.
Q: Zn + HgO → ZnO + Hg How many g of Hg (200.59 g/mol) will be produced in 30 days if the current…
A: Step 1: Calculating the amount of charge generated in 30days…
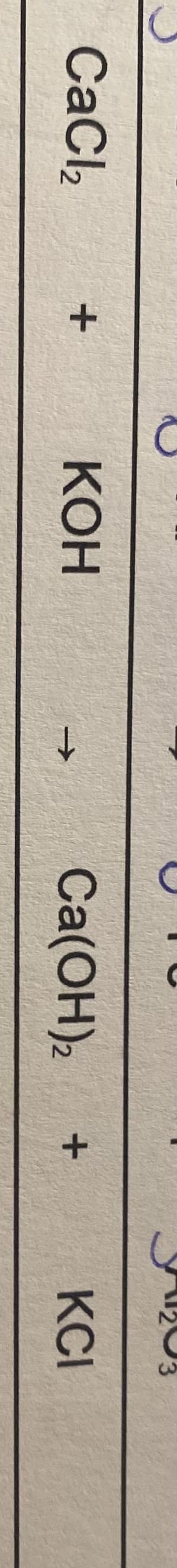
Balance the following equation
CaCl2 + KOH = Ca(OH)2 + KCI
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution

