Calculate the each initial CONCENTRATION for Pb(NO3)2 and KI before filling-out this chart. (Refer t Steps 1 and 2 of the procedure. Use the formula for Molarity to calculate the concentration of each salt after preparing the solution from the solid crystals dissolved in 100 mL of water. Do this for Lead (II) Nitrate and KI separately. Then use the dilution formula (starting with the initial concentration) for EACH beaker – each will have a the same total volume, but different volume of each solution added for each beaker. In short, FOR EACH BEAKER, multiple the concentration for Pb(NO3)2 with the concentration of KI.
Calculate the each initial CONCENTRATION for Pb(NO3)2 and KI before filling-out this chart. (Refer t Steps 1 and 2 of the procedure. Use the formula for Molarity to calculate the concentration of each salt after preparing the solution from the solid crystals dissolved in 100 mL of water. Do this for Lead (II) Nitrate and KI separately. Then use the dilution formula (starting with the initial concentration) for EACH beaker – each will have a the same total volume, but different volume of each solution added for each beaker. In short, FOR EACH BEAKER, multiple the concentration for Pb(NO3)2 with the concentration of KI.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 61E: The photograph below shows a beaker of water and a sugar cube, the combined mass of which is...
Related questions
Question
Calculate the each initial CONCENTRATION for Pb(NO3)2 and KI before filling-out this chart. (Refer t Steps 1 and 2 of the procedure.
Use the formula for Molarity to calculate the concentration of each salt after preparing the solution from the solid crystals dissolved in 100 mL of water. Do this for Lead (II) Nitrate and KI separately.
Then use the dilution formula (starting with the initial concentration) for EACH beaker – each will have a the same total volume, but different volume of each solution added for each beaker. In short, FOR EACH BEAKER, multiple the concentration for Pb(NO3)2 with the concentration of KI.
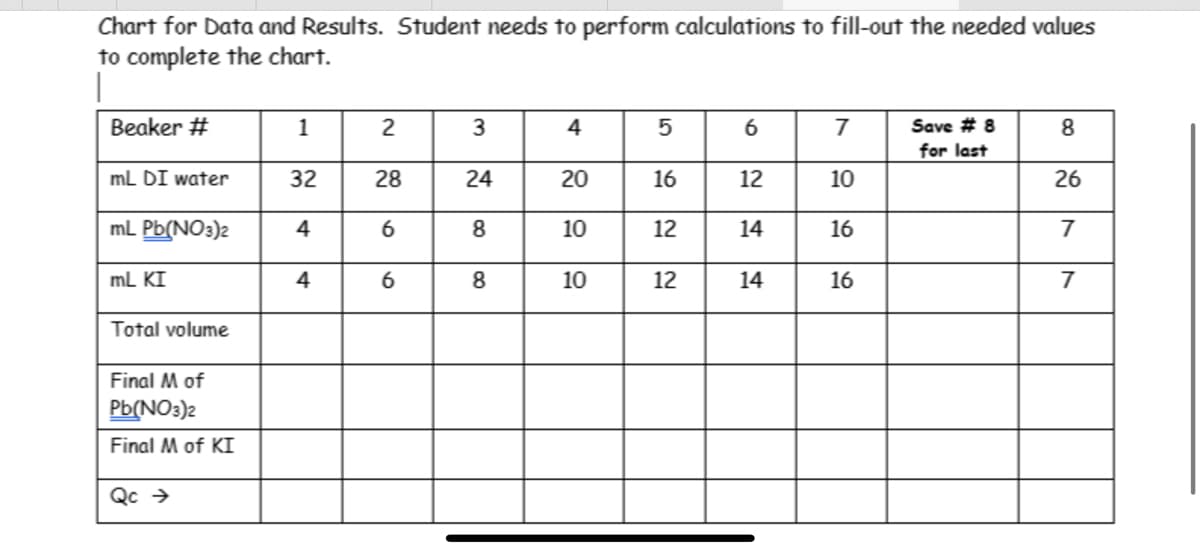
Transcribed Image Text:Chart for Data and Results. Student needs to perform calculations to fill-out the needed values
to complete the chart.
|
Beaker #
mL DI water
mL Pb(NO3)2
mL KI
Total volume
Final M of
Pb(NO3)2
Final M of KI
Qc →
1
32
4
4
2
28
6
6
3
24
8
8
4
20
10
10
5
16
12
12
6
12
14
14
7
10
16
16
Save #8
for last
8
26
7
7

Transcribed Image Text:Experiment #6: Ksp and Qc
After printing/copying or studying this guide/procedure, watch the Video (4.55 minutes)
by clicking on the link provided for Experiment #6 Module (under "Content").
You can watch the video however number of times you need to understand the experiment, within
the allotted time period of availability. The video opens at 8 AM on the day/date that the
experiment is schedules to be performed. You are given 48 hours to complete viewing the video.
The demo for this experiment has inserted some "fun parts", please don't get annoyed and just
ignore them.
For this video the precipitation of lead (II) iodide is demonstrated. Although the amounts
of compounds used are provided it will be the student's responsibility to calculate the actual
concentration for each mixture made.
The materials used in the experiment:
Analytical or top-loader balance
Eight 100-ml beakers
Erlenmeyer or volumetric flasks
Graduated cylinder
Lead Nitrate crystals and Potassium Iodide crystals
Summary of Procedure followed in the video: Take down notes and write down results as
necessary. Try to note down your results and observations in logical manner.
1) Assume that 0.331 gram of each given salt is weighed (separately)
2) In separate flasks, each salt is dissolved in 100 mL of DI water (calculate the initial
molarity of each solution assuming total volume of 100 mL). You will need a Periodic
Table. Reminder: Round all values of atomic masses to proper whole numbers.
3) The 8 clean beakers are arranged and numbered from #1 to #8 in proper order.
4) DI water is added in different volumes, Pay attention to the video and refer to chart below
(next page).
5) Lead Nitrate solution prepared in #2, is added to the beakers in varying volumes
accordingly (Refer to video and chart)
6) Potassium Iodide solution prepared in #2, is added to the beakers in varying volumes
accordingly (Refer to video and chart)
7) Beaker #8 is moved between Beakers #2 and #3.
8) Then, 26 mL of DI water is placed in Beaker #8, to which is added 7 mL of Lead nitrate and
7 mL of Potassium Iodide.
9) Note down your observations and perform the pertinent calculations. (see next page for
suggested table for results)
Online Lab for Ksp & QC
LLLCruz, Page 1 of 2
10) Take the quiz for Experiment #6 which takes the place of a lab report. You only have two
attempts to access the quiz.
Assume the Ksp of the precipitate to be 1.4 x 10-8. Use this
value to form conclusions.
(Note: there is a typo in the value of the Ksp provided in the demo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 7 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning