Calculate the heat of reaction deltaH for the following reaction: H2(g)+Cl2(g)——2HCl(g) Round your answer to the nearest kJ/mol.(hint: answer is not -185kj/mol)
Q: In which of the following combinations would you expect to have the greatest amount of heat…
A: Option B is weak acid + Strong Base.
Q: Calculate the heat of reaction AH for the following reaction: CH,(9) + 20,(9)–→CO,(9) + 2 H,O(g) You…
A:
Q: Use your table of Heats of Formation to calculate the heat of reaction for the following reaction:…
A:
Q: 2 H2 (g)-CHOH (g) AH 907 ko How many kilojoules of heat are released when 330 mg of CH;OH (g)…
A: The amount of energy released is as follows.
Q: Liquid hydrogen peroxide, an oxidizing agent in many rocket fuel mixtures, releases oxygen gas on…
A:
Q: Calculate the heat of reaction AH for the following reaction: 2 HCl(g) + Br,(g)→2 HBr(g) + Cl,(g)…
A:
Q: the given data, calculate the heat of reaction for the following reaction: (NH4)2Cr207(5) – Cr20a(s)…
A:
Q: What is the enthalpy of the reaction P406 (s) + 202(g)→P4010(s), given a) P4(s) + 302(g)→P406 AH =…
A:
Q: What is the enthalpy change for the reaction below 8 CO2(g) + 10 H2O(l) → 2 C4H10(g) + 13 O2(g) If…
A:
Q: calculate
A: Heat of formation for pure elements at room temperature value is Zero. Here Na, H2 and O2 these…
Q: How much more heat is released from the combustion of one mole of octane (C8H18) than from the…
A: Combustion of hydrocarbon produces CO2 and H2O Heat released from the combustion is calculated on…
Q: Calculate the heat of reaction AH for the following reaction: 2H,(9) + O,(9)→2 H,O(9) Round your…
A:
Q: Given the following enthalpy of formation: AH®[CO2] = -393.5 kJ/mol; AH°:[C¿H2] = 227.4 kJ/mol, and…
A: a. First, calculate the heat of reaction using standard enthalpy of formation data as follows:
Q: what is the enthalpy change when 45.7 grams of dinitrogen tetraoxide form? 2 NO2 (g) → N2O4…
A: Enthalpy change is the change in energy of the reaction.
Q: Calculate the heat of reaction AH for the following reaction: 2 HBr(g)→H,(g) + Br,(g) You can find a…
A:
Q: Calculate the heat of reaction AH for the following reaction: 2 HBr(g)→H,(g) + Br,(g) You can find a…
A: The heat of Reaction is the difference between the sum of bond energies of products and Reactants.…
Q: Calculate the enthalpy of formation of propane from: a. Combustion data (ΔHc = –2217 kJ mol–1; ΔHf…
A: Given data,∆Hcombustion=-2217kJ∆HCO2=-393.4kJ/mol∆HH2O=-285.2kJ/mol
Q: Consider the following heat of formation data: NH4(g) AH, = 95.4 kJ/mol %3D NH3(g) AH,° = -46.19…
A: ∆Hrxn is given by the ∆Hreactantsand ∆Hproducts.
Q: Calculate the AH for the reaction of the compound below with HBr. Show all work. Is the reaction…
A: Given an unsaturated alkene. We have to calculate the heat of the reaction, (∆H) when this given…
Q: Calculate the heat of reaction AH for the following reaction: CH,(9) + 20,(9)→CO,(g) + 2H,O(9) You…
A: Given reaction- CH4(g)+2O2(g) -> CO2(g)+H2O(g)
Q: Calculate the enthalpy of the following reaction: HCI + NaOH Nacl + H,0 AH HCI- -92.3 kJ/mol NAOH=…
A: The given reaction is as follows: HCl+NaOH⟶NaCl+H2O The expression for the enthalpy change for the…
Q: Calculate the heat of reaction delta H for the following reaction: 2HBr(g)——> H2(g)+ Br2(g) Round…
A: Bond enthalpy of H—H = 435 kJ.mol-1 Bond enthalpy of Br—Br = 192 kJ.mol-1 Bond enthalpy of H—Br =…
Q: Explain the cooling effect experienced when etha- nol is rubbed on your skin, given that 6.97…
A: When ethanol is rubbed on skin, cooling experience is observed. ∆H° for the conversion of liquid…
Q: 3. In the following reaction, the products are not favoured at room temperature, but are favoured at…
A: Given-> CO2(g) + H2(g) <------> CO(g) + H2O(g)
Q: Heat of formation values for four compounds are given in the table above. Compound ΔHf (kJ/mol)…
A: The reaction given is 2NH3g+3O2g+2CH4g→2HCNg+6H2Og The value of change in enthalpy can be given as…
Q: Calculate the heat of reaction AH for the following reaction. 2 HBr(g)-H,(9) + Br (g) You can find a…
A:
Q: The heat of combustion of 1-pentanol is -3331 kJ/mol. Given the following information, calculate the…
A:
Q: Given the heat of formation of the following compounds from their elemer AH°F = -393.5 kJ/mol CO:(g)…
A: The question is based on concept of chemical thermodynamics. We have to calculate enthalpy change…
Q: 17. Calculate the heat of combustion of propane C,H. CH, + 50, - 3CO, „ + 4H,0, Using the following…
A: Given: The thermochemical equation is as follows: 3C(s) + 4H2(g) -------> C3H8(g)…
Q: Use your table of Heats of Formation to calculate the heat of reaction for the following reaction:…
A: Thermodynamics is the branch of chemistry that mainly deals with the heat transfer between system…
Q: 13. From the table of standard heats of formation, ΔHf°, values, calculate ΔH° for the combustion of…
A: The balanced equation for the combustion of ethanol is
Q: Use the following reactions to answer the next two questions. 4PbO (s) → 4Pb (s) + 2O2 (g)…
A: The given three reactions are, (1) 4PbO (s) → 4Pb (s) + 2O2 (g) ∆H° = 876…
Q: calculate DeltasolutionH
A:
Q: Calcutlate the heat of reaction for: 2NaHCO3(s)->NaCO3+CO2(g)+2H2O(1)
A:
Q: Calculate the heat of reaction AH for the following reaction: 2 HCl(g) + Br,(g)→2 HBr(g) + Cl,(g)…
A: hhjj
Q: Use your table of Heats of Formation to calculate the heat of reaction for the following reaction:…
A: Heat of reaction or enthalpy change of reaction is used to measure the amount of heat energy…
Q: Calculate the heat of reaction AH for the following reaction: 2 H,(9) + 0,(9)→2 H,O(g) You can find…
A: Bond energy of H2 = 436 KJ/mole Bond energy of O2 = 495 KJ/mole Bond energy of O-H bond =…
Q: B2H6(g) + 3O2(g) --> B2O3(s) + 3H2O(g) ΔH = -2035 kJ Calculate the amount of heat…
A: B2H6(g) + 3O2(g) --> B2O3(s) + 3H2O(g) ΔH = -2035 kJThe amount of heat released when…
Q: Calculate the fuel value of CSH12, given that AH comb=-3535 kJ/mol. kJ/g Part 2 How much heat is…
A: Fuel value/calorific value is defined as the amount of heat released when a specified amount of…
Q: SnCl2 (k) → Sn (k) + Cl2 (g) 2 SnCl2 (k) + 2 Cl2 (g) → 2 SnCl4 (s) +325.1 kJ -372.4 kJ Using the…
A: The enthalpy of a reaction provides us with the amount of energy that is used up (absorbed) or given…
Q: Use your table of Heats of Formation to calculate the heat of reaction for the following reaction:…
A: Heat of reaction is given by ∆H=∆Hproduct-∆Hreactant where ∆H= Heat of reaction ∆Hproduct=Heat of…
Q: Calculate the heat of reaction deltaH for the following reaction: 2H2(g)+O2(g)——>2H2O (g) Round your…
A: The reaction given is, => 2 H2 (g) + O2 (g) → 2 H2O (g)
Q: Calculate the heat of reaction for the conversion of 1 mole of glucose into formaldehyde, given the…
A:
Q: Determine the enthalpy of formation of acetone, C3H6O(l) C3H6O(l) + 4O2(g) -> 3CO2(g) +3H2O(l)…
A: The enthalpy determines the heat associated with a chemical reaction. The change in enthalpy is the…
Q: Calculate AH°rxn for the reaction below, using the bond enthalpies provided (you may not need to use…
A: Whenever reactant to form product than during this process, old bonds of reactant breaks while new…
Q: 2. Calculate the heat of reaction, AH°, for the following: 3FECI:(s) + 4H0(g) → Fe,O.(s) + 6HCI(g) +…
A: Standard enthalpy of formation values Hf (FeCl2(s) ) = -341.79 kJ/mol Hf (H2O(g) ) = -241.818 kJ/mol…
Q: From the following enthalpies of reaction: 2 H2(g) + O2(g) → 2 H2O(l)…
A:
Q: Liquid hydrogen peroxide, an oxidizing agent in many rocket fuel mixtures, releases oxygen gas on…
A: The enthalpy change or heat involved during a decomposition reaction is known as heat of…
Q: 3. Compound Y has a molar mass of 85.0 g/mol. The combustion of 510.0 g of compound Y causes the…
A:
Q: Calculate the heat of reaction deltaH for the following reaction: 2NH3(g)——> N2(g) + 3H2(g) Round…
A:
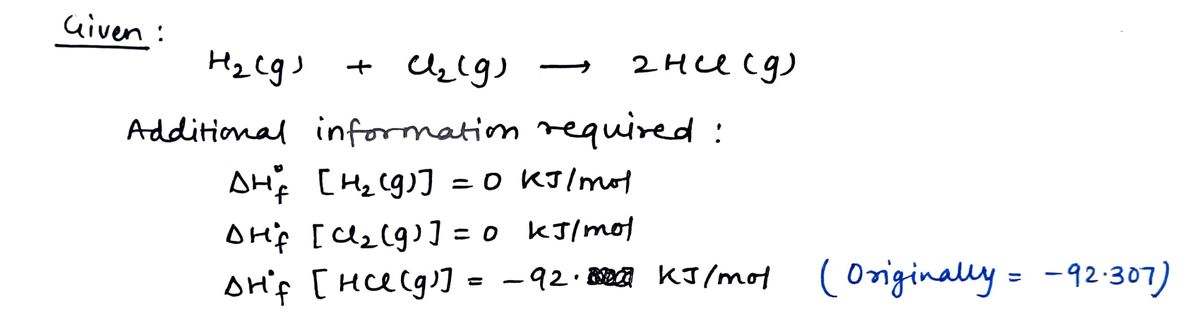
Step by step
Solved in 2 steps with 2 images

- The heat of neutralization, Hneut, can be defined as the amount of heat released (or absorbed), q, per mole of acid (or base) neutralized. Hneut for nitric acid is -52 kJ/mol HNO3. At 27.3C, 50.00 mL of 0.743M HNO3 is neutralized by 1.00 M Sr(OH)2 in a coffee-cup calorimeter. (a) How many mL of Sr(OH)2 were used in the neutralization? (b) What is the final temperature of the resulting solution? (Use the assumptions in Question 11.)Calculate the heat of reaction deltaH for the following reaction: 2H2(g)+O2(g)——>2H2O(g) Round your answer to the nearest kJ/mol.(hint: answer is not -484kj/mol)Calculate the heat of reaction deltaH for the following reaction: CH4(g)+2O2(g)——> CO2(g) + 2H2O(g) Round your answer to the nearest kJ/mol. (Hint: answer is not -794 kJ/mol)
- Calculate the heat of reaction deltaH for the following reaction: CH4(g) + 2O2(g)——>CO2(g)+ 2H2O(g) Round your answer to the nearest kJ/mol.Calculate the heat of reaction deltaH for the following reaction: 2H2(g)+O2(g)——>2H2O (g) Round your answer to the nearest kJ/mol.Find the heat of reaction and explain what happen to it. Calculate the ∆H of reaction for: C3H8 (g) + 5O2 (g) => 3CO2 (g) + 4H2O (l) The values of ∆H of reaction are as follows:C3H8 (g) = -103.95 kJ/molCO2 (g) = -393.5 kJ/molH2O (g) = -285.8 kJ/mol Topic: Enthalpy of Reaction
- Calculate the heat of reaction delta H for the following reaction: 2HBr(g)——> H2(g)+ Br2(g) Round your answer to the nearest kJ/mol.Use your table of Heats of Formation to calculate the heat of reaction for the following reaction: C2H4(g) + 3O2(g) ⟶ 2CO2(g) + 2H2O(g)How do I go about calcualting the estimated heat of reaction of the equation shown in the image. Could you explain in detail please. bond energies:H-H 436 kJ/molH-X 431 kJ/molH-C 414 kJ/molC-X 339 kJ/molX-X 243 kJ/mo
- what is the enthalpy change when 45.7 grams of dinitrogen tetraoxide form? 2 NO2 (g) → N2O4 (g) Delta H = – 57.2 kJ a. +2.61 x 10^3 kJ b. – 28.4 kJ c. + 57.2 kJ d. – 49.7 kJ e. – 57.2 kJ f. +28.4 kJ g. None of these h. +49.7 kJ i. – 2.61 x 10^3 kJCalculate the heat of reaction for: 2 C + 4 H2 + O2 ----> 2 CH3OH given: C + O2 ----> CO2 ΔH = -393.5 kJ 2 H2 + O2 ----> 2 H2O ΔH = -571.6 kJ 2 CH3OH + 3 O2 ----> 2 CO2 + 4 H2O ΔH = -1452.8 kJ a) -3383 kJ b) -477.4 kJ c) 3383 kJ d) 94.2 kJ e) -94.2 kJ13. From the table of standard heats of formation, ΔHf°, values, calculate ΔH° for the combustion of ethanol according to the reaction:C2H5OH(l) + 3 O2(g) -> 2 CO2(g) + 3 H2O(l)



