Q: Determine the concentration (M) of H3O+ in a 0.589 M solution of a triprotic weak acid, H3A, given…
A: Given, the concentration (M) of H3O+ in a 0.589 M solution of a triprotic weak acid, H3A is:
Q: how many milliliters of an aqueous solution of 0.124 M barium hydroxide is needed to obtain 12.7…
A: Given, Mass of barium hydroxide salt = 12.7 g Concentration of barium hydroxide solution = 0.124 M =…
Q: a) Acetic acid is a weak acid that dissociates into acetate ion and hydronium ions in solutions:…
A: 1.) To calculate ratio , we would use Henderson Hasselbalch equation. 2.) To make a buffer of given…
Q: What is the IUPAC name for the following molecule? CH3 H3C-C-CH,CH,F CH3
A: Rule:1 find the longest continuous chain of carbon atoms, and use the name of this chain as the base…
Q: Give some advantages of steam distillation over simple distillation.
A: Steam distillation is a process used for separating and purifying isolate temperature-sensitive…
Q: The pH of a saturated soloution of magnesium hydroxide, Mg(OH)2, (milk of magnesia) was found to be…
A: We have to predict the Ksp of salt.
Q: Outcome 11 Evaluation: Bridge Assignment #1 Predicting Products of EAS with Directing Groups 1) Just…
A: GIVEN:-
Q: What is the OH- concentration in a 0.106 M solution of ammonia? The pKb at the temperature of the…
A: Answer: 1.34×10^-3 M
Q: E'= 1.678Y Nida(s) Cu?9) -> Curs) Given unbalon ced areacrion: Eこ0.34 2 V Draw the galvanic cell (be…
A:
Q: 5. To determine whether a shiny silver-colored rock is actually silver, a chemistry student decides…
A: GIVEN:-
Q: Which type of distillation set-up is appropriate for the given condition? Stable component of…
A:
Q: An aqueous solution of NaCl is treated with an aqueous solution of Silver Nitrate. What would you…
A: Given that - Aqueous solution of sodium chloride, NaCl Aqueous solution of silver nitrate, AgNO3…
Q: mass m = 0.056 kg of benzene vapor at its boiling point of 80.1 °C is to be condensed by mixing the…
A:
Q: When aqueous solutions of potassium cyanide and hydroiodic acid are mixed, an aqueous solution of…
A:
Q: What forms after adding HCl (reacidification) to methyl salicylate? Draw and name the compound…
A: We have to tell what happened when HCl is added to methyl salicylate. And we also give the name of…
Q: how many milliliters of an aqueous solution of 0.124 M barium hydroxide is needed to obtain 12.7…
A:
Q: Provide the correct IUPAC name for the skeletal (line-bond) structure shown here. Br
A:
Q: PRE A Enough of a monoprotic acid is dissolved in water to produce a 1.55 M solution. The pH of the…
A:
Q: A system absorbs 1.66E2 kJ of heat and the surroundings do 1.032E2 kJ of work on the system. What is…
A: We have to predict the change in internal energy of system.
Q: 1)A patient with respiratory alkalosis has a blood plasma pH of 7.82. What is the [H3O + ] of the…
A: 1) Given, pH of blood plasma = 7.82 Concentration of H3O+ ion = [H3O+] = ? The formula used are,…
Q: Consider the coordination complex [Cr(OH2)3CI3]Br3 Identify the ligands by name (include the word…
A: Given complex--- [Cr(OH2)3Cl3]Br3
Q: discussion on cis and trans potassium dioxalatodiaquachromate
A: GIVEN:- discussion on cis and trans potassium dioxalatodiaquachromate
Q: 1.1 Fill in the missing (correct) words only: (a) In general, as and as increases. increases,…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: VII Write the name of organic compounds obtained from the following reactions. (1) reaction of…
A:
Q: What products are obtained when (R)-2-bromobutane reacts with CH 30 /CH 30H under conditions for…
A:
Q: What is the key feature of a dialysis bag needed for it to be useful for measuring the binding…
A: Answer - Equilibrium dialysis is useful for measuring small molecule binding to macromolecule . It…
Q: For the aqueous [Hgl, complex K,6.76 x 10 at 25 "C. Suppose equal volumes of 0.0092M Hg(NO,),…
A: Given : Kf = 6.76 x 1029 [Hg(NO3)2] = 0.0092 M [NaI] = 0.20 M
Q: A 1,2-methyl shift occurs when which of the following iodides is heated in ethanol? O…
A: In case of 1st compound 1,2 methyl shift take place. 3 degree carbocation is more stable than 2…
Q: It 4.30 L of CO, gas at 14 °C at 795 mmHg is used, what is the final volume, in liters, of the gas…
A: V1 = 4.30 L T1 = 14°C = 14 + 273 = 287 K P1= 795 mmHg T2 = 35°C = 35 + 273 = 308 K P2= 755 mmHg V2=?…
Q: malate + NAD+ oxaloacetate + NADH + H+ If the pH = 7 and [malate] = 0.0010 M, [oxaloacetate] =…
A: For the given reactions the standard reduction potentials are taken from electrochemical series data…
Q: Based on TLC, how can you tell if the reaction is complete and what is the relevvance of the…
A: Answer: TLC- TLC (This layer chromatography) is the separation technique used to determine the…
Q: At 22 ∘C, an excess amount of a generic metal hydroxide, M(OH, is mixed with pure water. The…
A:
Q: d. After subtracting 2 electrons per bond used in part (b) above, the number of ele is The number of…
A:
Q: Name the following compounds following IUPAC rules..
A:
Q: d) 250 mLof o-0750 M Ag NO3 from the solid reagert -
A: In multiple questions we solve only first question according to the Bartleby guidelines. Calculate…
Q: 3) Calculate the pH of each solution given the following [H3O'], and state whether the solution is…
A: 3) Given, Concentration of the H3O+ are: a) 5.1 × 10-7 M b) 6.75 × 10-8 M c) 4 × 10-2 M pH of each…
Q: Please fill in blanks as shown on problem!!
A:
Q: Compute for the total acidity of the food sample assuming that ten grams of sample was dissolved to…
A:
Q: Question 1 1.1 Fill in the missing (correct) words only: (a) In general, as and as increases,…
A: a) In general, as pressure increases, non-ideal behavior increases and as volume decreases,…
Q: 11-64 The tosylate of (2R,3S)-3-phenyl-2-butanol undergoes E2 elimination on treatment with sodium…
A: We must note here Z product is forming. Let us discuss the reaction.
Q: Use these findings to answer the question. Compound X is optically inactive and has the formulac 16H…
A: The dibromo compound gives a dehydrohalogenation reaction to form a double bond. The alkene gives…
Q: Cl2(g) 2HCI(g) AH = –184.6 kJ %3D | 2: O2(g) + 2 F2(g) AH = -49.4 kJ ΔΗ 3: 202(g) → 2NO2(g) AH…
A: Given, Reaction, N2 + O2 ---> 2NO □H = 66.4 kJ Volume of N2 = 167 L Moles = Volume (L)/22.4…
Q: Which of the solvents given below will boil first? ethanal = 120.26 KPa %3D ethanoic acid= 1.94 KPa
A: Given-> Ethanal = 120.26 KPa Ethanoic acid = 1.94 KPa
Q: Hello can this problem please be simplified, I would like to make sure I am getting each step down…
A: P1 = 193 psi V1 = 39 mL T1 = 331 K P2 = 795 psi V2 = ? T2 = 675 K
Q: AChange Reactions Prelab Qulestions Anniston Stoall Name Section CHM l01-217 1. Write complete…
A: Precipitation occurs if the solubility of the product formed during the reaction is less than that…
Q: A 10.00-g sample containing an analyte was transferred to a 250-mL volumetric flask and į diluted to…
A: Solution - According to the question - Given - In the spiked sample, the signal is due to 10 ml of…
Q: The solubility of magnesium oxalate, MgC2O4, in water is (7.70x10^0)x10-3 mol/L. What is the Ksp…
A:
Q: A generic salt, AB, has a molar mass of 229 g/mol and a solubility of 5.70 g/L at 25 °C.…
A: We have to predict the Ksp of this salt.
Q: addition of S2- ion to Fe(OH)2(s) results in the formation of FeS(s). Explain why the addition of…
A:
Q: For the aqueous AIF complex K= 6.92 x 10" at 25 °C. .3+ Suppose equal volumes of 0.0076M Al (NO,),…
A:
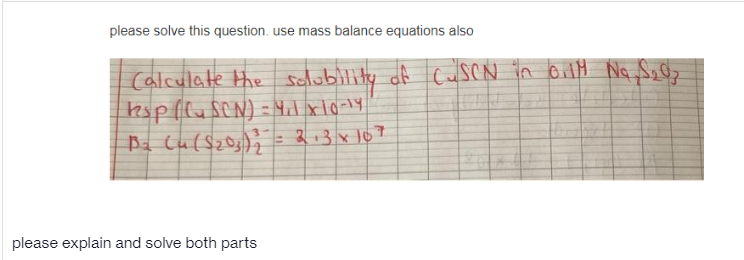
Step by step
Solved in 3 steps

- Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.Using the percent purity calculations, determine the percent yield of synthesis of aspirin. Part I Synthesis of Aspirin Mass of salicylic acid used (g) 2.029g Volume of acetic anhydride used (mL) 5ml Mass of acetic anhydride used (vol. × 1.08 g/mL) 5.4g Mass of aspirin synthesized (g) 3.256g Part II Melting Temperature Data Melting temperature (°C) 133°C Part III Salicylic Acid Standard Stock Solution Initial mass of salicylic acid (g) 0.210g Moles of salicylic acid (mol) 0.0147 mol Initial molarity of salicylic acid (M) 0.724 M Part III Beer’s Law Data for Salicylic Acid Standard Solutions Trial Concentration (M) Absorbance Water (mL) 1 10 0.301 0 2 7.5 0.219 2.5 3 5.0 0.163 5.0 4 2.5 0.074 7.5 Best-fit line equation for the salicylic acid standards Test of the Purity of the Synthesized Aspirin Initial mass of aliquot of product (g)…An impure sample of 0.1002 g benzoic acid underwent purification by recrystallization. A mass of 0.0542 g of white solid benzoic acid was obtained. Calculate the % purity of the initial sample. Show calculations and report the result with correct number of significant figures. Hi there please answer all parts clearly and concisely with legible hand writting, thank you!
- 5.00 mL of stock solution is diluted to 25.00 mL, producing solution ALPHA. 10.00 mL of solution ALPHA is diluted to 25.00 mL, resulting in solution BETA. 10.00 mL of solution BETA is then diluted to 25.00 mL, producing solution GAMMA. dilution factor for ALPHA from stock solution = 0.167 dilution factor for BETA from ALPHA solution = 0.0476 part c and d?Determine the percentage Fe in a sample of limonite from the following data:Sample : 0.5000g ; KMnO4 used = 50.00 ml ; 1.000ml of KMnO4 is equivalent 0.005317 g Fe,FeSO4 used = 6.00 ml; 1.000ml FeSO4 is equivalent 0.009200 g FeO ( ans 44.59 %)After heating 2.000 g of MgSO4.7H2O to constant mass, the remaining residue in the crucible weighs 0.978 g. Using the data above , calculate the mass of water lost. After heating 2.000 g of MgSO4.7H2O to constant mass, the remaining residue in the crucible weighs 0.978 g. Using the data above , calculate the mass percent of water in the hydrate. 16 AND 17 PLEASE
- Using a 25mL pipette, you prepare a standard solution by pipetting 25.00mL of the standard iron solution[0.25g/L] into a 500mL volumetric flask. Then, using a 10mL pipette you transfer a 4mL aliquot of this solution into a 50mL volumetric flask and dilute up to the mark. Calculate the % uncertainty.Given the following data, what is the molarity? Include the relative error Error is 1.0 mg or 1.0e-4using exactly 5.00 mL of 0.0400 M stock CuSO4 solution. Add 100 mL of water. Data for Part IMass of empty dish: 32.470 g empty dishVolume of 0.0400 M solution: 5.00 mL CuSO4 solution.Mass of dish and 0.0400 M solution: 37.497 g dish and solutionMass of dish and CuSO4 solid: 32.503 g dish and CuSO4 solidCalculations for Part I1. Calculate the mass of solution2. Calculate the mass of solid CuSO4 dissolved in the solution.3. Calculate the number of moles of solid CuSO4 dissolved in the solution.4. Calculate the mass of water evaporated from the solution.5. Calculate the density of solution, (g solution/mL solution).6. Calculate the % by mass, CuSO4 in solution (100 x g CuSO4/g solution).7. Calculate the molality of solution (moles CuSO4/kg solvent).8. Calculate the molarity of solution (moles CuSO4/L solution).9. Given that the true molarity is 0.0400 M, calculate the percent error of your result.
- Using a 10mL pipette, you prepare a standard solution by pipetting 10.00mL of the standard iron solution [0.25g/L] into a 100mL volumetric flask. Then, using a 25mL pipette you transfer a 25mL aliquot of this solution into a 250mL volumetric flask and dilute up to the mark. Calculate the % uncertainty7. Consider a1Msolution ofNa3AsO4. Write the charge and mass balance equations for this system. (please type answer not write by hend)Given the following:Feed weight = 1 kgFeed grade = 16 grams per tonConcentrate Weight = 126.03 gConcentrate Grade = 0.0158%Recovery = 23%Provide the formula on how the Recovery was calculated.







