Calculate the wavelength of the photon omitted when an electron makes a transition from n =4 to n 3. You can make use of the following constants: 6.626 x 10-34 J.s c- 2.998 x 10 m/s 1m = 10° nm Express your answer to four significant figures and include the appropriate units. > View Available Hint(s) %3D Value Units Submit Provide Feedback Next >
Calculate the wavelength of the photon omitted when an electron makes a transition from n =4 to n 3. You can make use of the following constants: 6.626 x 10-34 J.s c- 2.998 x 10 m/s 1m = 10° nm Express your answer to four significant figures and include the appropriate units. > View Available Hint(s) %3D Value Units Submit Provide Feedback Next >
Chapter25: Instruments For Optical Spectrometry
Section: Chapter Questions
Problem 25.7QAP
Related questions
Question
100%
Transcribed Image Text:- of
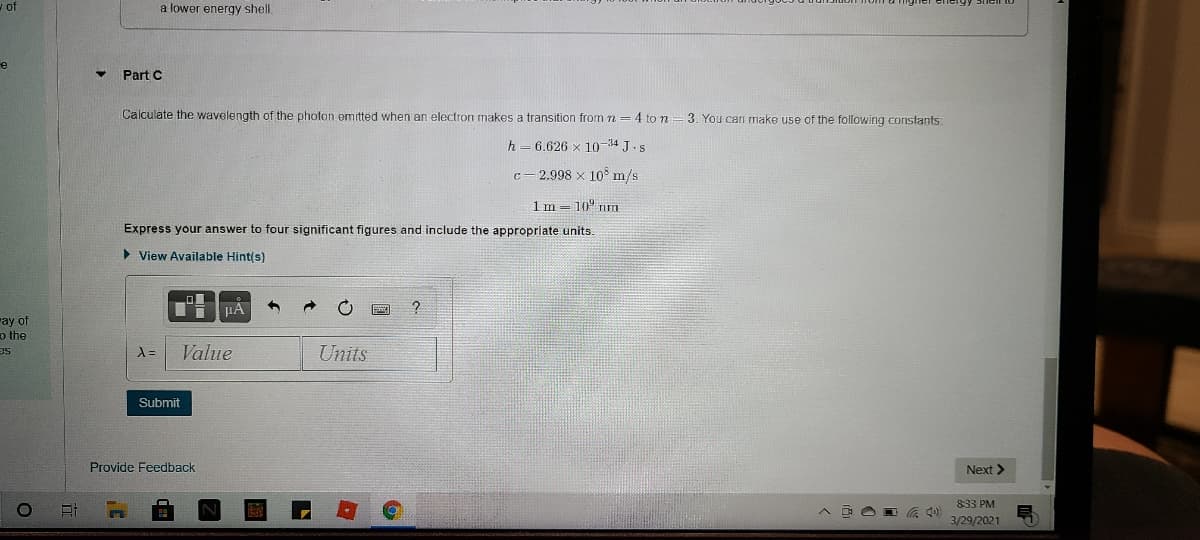
a lower energy shell.
e
Part C
Calculate the wavelength of the photon emitted when an electron makes a transition from n =4 to n
3. You can make use of the following constants:
h=6.626 x 10-34 J.s
c- 2.998 x 10° m/s
1m = 10° m
Express your answer to four significant figures and include the appropriate units.
> View Available Hint(s)
Ti HA
ray of
o the
Value
Units
se
Submit
Provide Feedback
Next >
833 PM
3/29/2021
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning