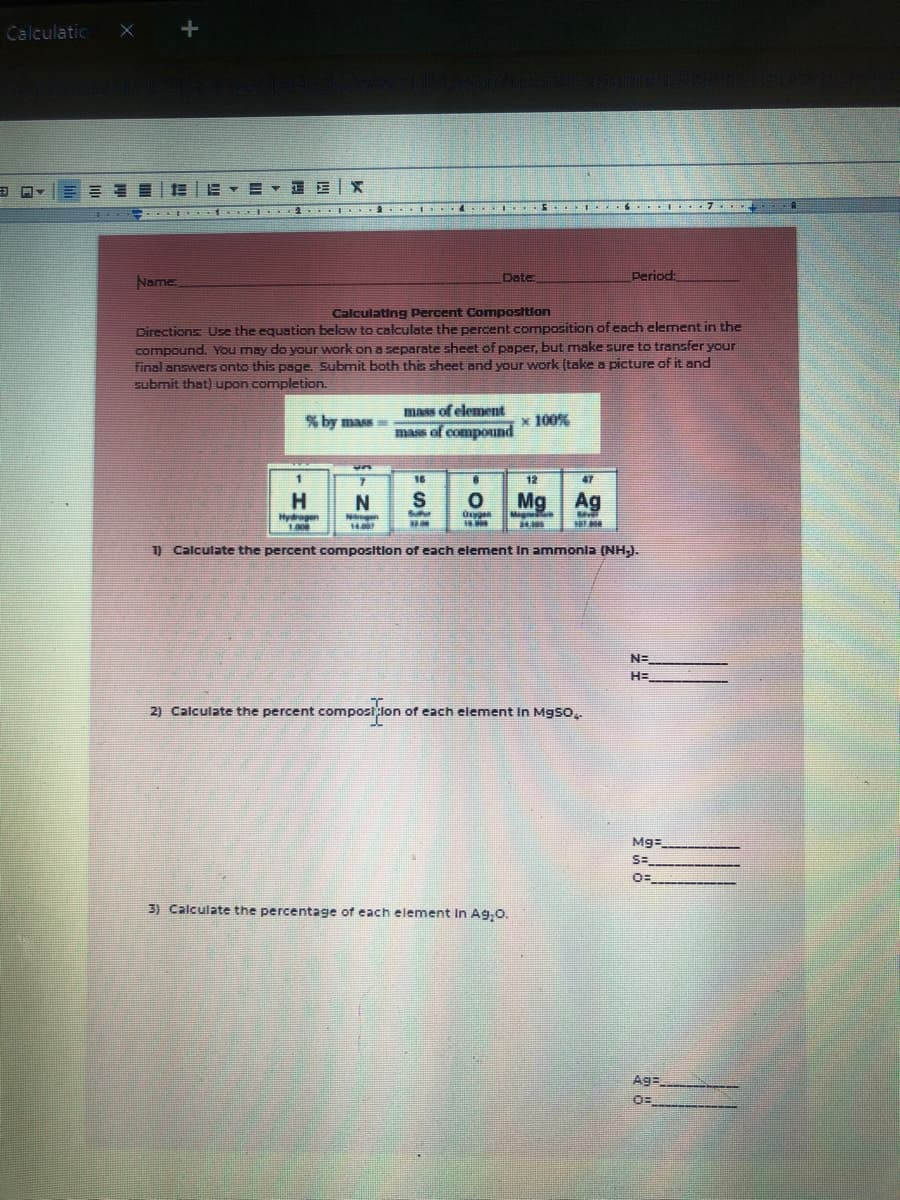
Calculating Percent Composition Directions Use the equation below to calculate the percent composition of each element i compound. You may do your work on a separate sheet of paper, but make sure to transfer your final answers onto this page. Submit both this sheet and your work (take a picture of it and submit that) upon completion. the mass of element mass of compound % by mass= x 100% 16 12 47 H. Mg Ag Hydregen 100 .00 1) Calculate the percent composition of each element In ammonla (NH). N= H= 2) Calculate the percent compost lone of each element In MgSo, Mg= S= 3) Calculate the percentage of each element In Ag,0. Ag=
Calculating Percent Composition Directions Use the equation below to calculate the percent composition of each element i compound. You may do your work on a separate sheet of paper, but make sure to transfer your final answers onto this page. Submit both this sheet and your work (take a picture of it and submit that) upon completion. the mass of element mass of compound % by mass= x 100% 16 12 47 H. Mg Ag Hydregen 100 .00 1) Calculate the percent composition of each element In ammonla (NH). N= H= 2) Calculate the percent compost lone of each element In MgSo, Mg= S= 3) Calculate the percentage of each element In Ag,0. Ag=
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter2: Atoms Molecules And Ions
Section: Chapter Questions
Problem 165SCQ
Related questions
Question
100%
Transcribed Image Text:Calculatic
E - E- I E|X
4 D S... E S . . 7
Name
Date
Period:
Calculating Percent Composition
Directions Use the equation below to calculate the percent composition of each element in the
compound. You may do your work on a separate sheet of paper, but make sure to transfer your
final answers onto this page. Submit both this sheet and your work (take a picture of it and
submit that) upon completion.
mass of element
% by mass
x 100%
mass of compound
16
12
47
H.
S
Mg
Ag
Hydregen
1) Calculate the percent composition of each element In ammonla (NHJ.
N=
H=
2) Calculate the percent compostlon of each element In Mgso..
Mg=
S=
3) Calculate the percentage of each element In Ag.0.
Ag=
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning