Q: ▼ Part A The following diagrams show the chemical reaction over a four-hour time period. If C is…
A: Equilibrium of reaction can be defined based on the concentration change of reactants and products.
Q: Tritium, which is , has a half-life of 12.3 years. Calculate the amount of time it would take for…
A:
Q: Why is this a cyclohexane ring when there is only one double bond?
A: The product is an enamine and is correctly drawn.
Q: Methylamine, CH3NH2, is a weak base. A 0.72 M aqueous solution of methylamine has a pH of 12.20.…
A:
Q: Nickel can be plated from aqueous solution according to the following half reaction. How long would…
A:
Q: Calculate the heat of reaction for the reaction 25+ 2 OF 2(g) → SO₂ + SF (g) 2(g) using the…
A: Data given: OF2 (g) + H2O (l) → O2 (g) + 2 HF (g) ΔH° = −277 kJ ------------ (1) SF4(g) + 2…
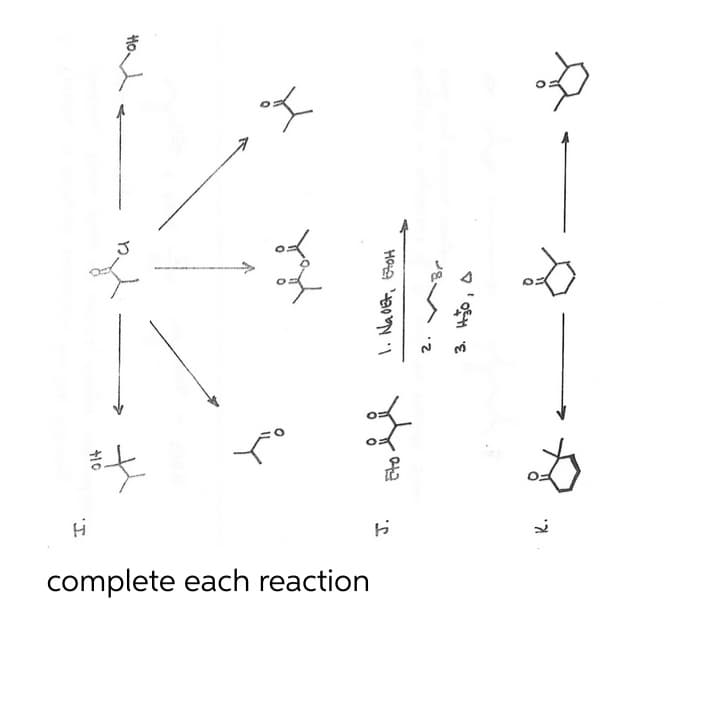
Q: Outline the synthetic steps necessary to carry out the conversion below. You may use any organic or…
A: Given transformation: We have to outline the synthetic steps that are necessary to carry out this…
Q: types of Electrodes
A: (1) Metallic – Metallic means consisting of partly metal or consisting entirely of metal. The…
Q: OH НО
A:
Q: 49. Identify structures A and B in the following reaction sequence. D 1) 0₂ 2) DMS A NaOH H₂O B…
A:
Q: 1equiv. 1. SH SH ZnCl₂ 2. CH₂MgBr || 3. HgCl₂, H₂O = III
A:
Q: Question #1 ● ● ● # of moles of p-ethyl phenol used: (Show calculations) Amount (moles) of unknown…
A:
Q: A 1 liter solution contains 0.339 M hydrofluoric acid and 0.452 M sodium fluoride. Addition of 0.085…
A: Given, [HF] = 0.339 M [NaF] = 0.452 M Volume of solution in L = 1 L Moles of Ba(OH)2 added =…
Q: as content is projected and Provide the reagents necessary to complete the following reactions. More…
A:
Q: Please draw the stepwise mechanism for this reaction !!
A: Concept based on organic chemistry reaction mechanism.
Q: 13. Draw structures to show how a triacylglycerol is formed using the following fatty acids: 16:1…
A: we have to draw the structure of a triacylglycerol identify functional group present identify it…
Q: Calculate AGxn for the given reaction at 700 K under the given conditions + 3 H₂(g) → 2 NH3(g) AGO =…
A:
Q: Write the thermochemical equation for the given reaction: (provide required to find in the problem)…
A: Burning of hydrocarbon in presence of atmospheric oxygen is called combustion process.
Q: Can you show the mechanism for those reactions
A: I have solved 4 . Pls repost your remaining questions seperately. Thanks
Q: Propose reaction mechanisms between benzoyl chloride with the following reagents: a) H₂O, pyridine…
A:
Q: Given the following equation: 2C4H10(g)+13O2(g)→8CO2(g)+10H2O(g)2C4H10(g)+13O2(g)→8CO2(g)+10H2O(g)…
A: The balanced chemical equation for the combustion of C4H10(g) is: 2C4H10(g) + 13O2(g) → 8CO2(g) +…
Q: C. d. HO 공연 그 I HO HO OH
A:
Q: Consider a solution of 235 mM acetic acid (pKa = 4.76). What would be the pH of this solution? And…
A:
Q: ▼ ▼ Part A NaOH with [OH-] = 1.4 x 10-²M Express your answer using two significant figures. 15. ΑΣΦ…
A: we have to calculate the hydronium ion concentration
Q: 9. Identify the chemical compound with the following Mass, IR, and H NMR spectra. Relative abundance…
A: By taking the help of Mass, IR and NMR spectroscopy we can determine the structure of the unknown…
Q: 1. a. Using retrosynthetic analysis, propose starting materials. Your answer should include two…
A:
Q: A buffer of acetic acid and sodium acetate is prepared by mixing 10.0 mL of 0.50 M acetic acid with…
A:
Q: . For the reaction N +0. → 2NO 2(g) 2(g) needed to generate 40 moles of NO? 0 ‚ÄHº : = 180.5 kJ, how…
A:
Q: Balance the following equations using lowest whole number coefficients. A number 1 must be placed…
A: Since, Balanced reaction represent the number of each atom present in both side are present in equal…
Q: Predict the major product of the following reaction. NH₂ 1a. OLI O 1b. MeCl 1a. CH3I (excess) K₂CO3…
A: -> Nucleophile can give Michael addition reaction to the alpha,beta - unsaturated carbonyl…
Q: 8. For each transformation below, (a) Label the newly formed functional group in the product. (b)…
A: #8: #(a): A substituted product is formed through the inversion of configuration. The substituted…
Q: rrange the following in order of DECREASING basicity. Type letter codes only of your answer in…
A: Decreasing order of Basicity: N > L > K > M > J Alkoxy group at para-position of…
Q: 4. Write out the 10 structure for the following polypeptide: Ο Η OH 요 H₂N-CH-C-N-CH-C-N-CH-COO + T…
A: The primary structure is the linear sequence of the amino acids of the peptides. When the peptide…
Q: Use the following information to answer the next question.
A: Given Chemical Reaction is Fe (s) + H2SO4 (aq) → Fe2(SO4)3 (aq) + H2 (g)
Q: Indicators can be used to help approximate the pH of a solution based on the indicator colour. Here…
A:
Q: 3. Calculate AG for the dissolution of Ca(OH)2 at room temperature and 100 °C.
A: Given - Volume of Ca(OH)2 at 25°C = 5.0 mL Volume of Ca(OH)2 at 100°C = 5.0 mL…
Q: Part E Calculate the pH of soy milk.
A:
Q: What is the major product from the following series of reactions? Problem viewing the image. Click…
A: The synthesis of the amine from alkyl halide by using some reagents which are given below
Q: QUESTION 27 The oxidation of hydrogen by oxygen is one of the most-used reactions in fuel-cell…
A:
Q: i) 1) -B-H 2 2) H₂O₂, H₂O
A: Alkynes undergo hydroboration reaction to form an enol which tautomerizes to form a Carbonyl…
Q: Draw the organic product(s) of the following reaction. + 0- HN(CH3)2 • You do not have to consider…
A: Here we have to write the major product formed in the following given reactions.
Q: Chemistry CI Starting Material HO Starting Material a Eto Target Molecule HO + enantiomer Target…
A:
Q: 3. Write out the 1° structure for the following polypeptide: CH, 11 OH 0…
A:
Q: The saturated calomel electrode, abbreviated SCE, is often used as a reference electrode in making…
A: For the galvanic cell we know that, the electrode having greater standard reduction potential will…
Q: 6. Find the amount of heat given off from combustion of 330 g of CH₂OH, according to the equation…
A: Given, Note: the equation is not balanced: CH3OH(l) + 3O2(g) ---> 2CO2(g) + 4H2O(l) Balancing…
Q: How does the structure of a Polymer affect its function.
A: Here we have to explain how does the structure of a polymer affect its function? We can explain it…
Q: Name: DESPR www.ged C1 4 HNA EX
A:
Q: Propose a synthesis for the following compound using toluene and any other reagents necessary. Show…
A:
Q: • A compound with molecular formula C3H6O2 displays the following IR, HNMR and. Propose a structure…
A: Given that, the molecular formula of an unknown compound is C3H6O2. Also, the IR spectra and the 1H…
Q: Rank from most to least acidic. To rank Items as equivalent, overlap them. View Available Hint(s) pH…
A:
5
Step by step
Solved in 3 steps with 3 images

- 2. ^ OH OHI. Name the two organic compounds. Br OH B. CH,O OH GH,0.- ¢ =ċ- c -ċ-ċ-B. is O =¢ si Br Catla Ç =03 AGNO, + K,PO, --> Ag.PO, + 3KNO, Describe what is happening Reactants combine into a large one Reactant breaks apart into smaller molecules O A pure element swaps places with an element in a compound elements from two different compounds switch place O a hydrocarbon reacts with oxygen to form carbon dioxide and water
- One millimeter of water contains about 20 drops. One gram of water contains 3.34x10^22 molecules of water. The density of water is 1.00 g/mL. How many molecules of water are there in 1 drop?A certain alloy with a density of 11.5 g/cm3 contains 55% iron by mass. a) How many pg of iron would be present in a 4.5"x5.0"x11" piece of alloy?A molecule has a mass of 4.65 x 10^-23 g. Provide twopossible chemical formulas for such a molecule.
- 3a. Another student performed the same percent composition of a hydrate experiment, but kent the crucible completely covered throughout the entire experiment. What effect would this have on the experiment? Cor 3b. Would the student's calculated percent water in the hydrate be high, low, or unaffected? Explain why or why not.Assuming gasoline to be pure 2-methylhexane, C7 H16 (actually, it is a mixture of many substances), an automobile getting 26.5 miles per gallon would consume 5.96 x 1023 molecules per mile. Calculate the mass of this amount of fuel. Mass = gAt 25°C methanol (CH4O) has a density of ~0.792 g/mL. If a reaction requires the chemist to use 644.50 moles of methanol, what volume will be required (in liters)? Use these atomic masses: C = 12.011 amu; H = 1.008 amu; O = 15.999 amu. Not these answers 26 0.004 0.009 26.07

