Q: Provide proper IUPAC name for the following compound: НО H3C OH OH mevalonic acil
A: Given compound is : Give the IUPAC name of this compound ? Carboxylic acid (-COOH) act as the…
Q: Using the balanced equation and the data in the table below, calculate the theoretical enthalpy of…
A: To calculate enthalpy of combustion , we would use gives bond energies. For this we have to…
Q: What is the major product of the reaction below under the provided thermodynamic conditions? HBr (1…
A: Given reaction is : What is the major product of the reaction under thermodynamic conditions ?…
Q: Why is it important to assess factors that influence egg white foam stability?
A: Egg white foams are made from whipping egg whites very quickly to add air and create volume. Airy…
Q: 13) Write in the reactants needed to make the following transformations (only one reactant needed…
A: According to the bartleby guidelines for multiple questions, I can solve only first one question. If…
Q: Q2. The density of a metal is 13.6 g/cm³. The density of the metal is ------- kg/m³ B. 1.36 × 104…
A:
Q: Please assign the peaks with explanation. Thank you.
A:
Q: The pH of 0.01 m Potassium hydrogen phthalate is... O 2.05 O 3.55 O 4.00 O 5.00 O 6.00
A: Here we are required to find the pH of the solution of 0.01M of KHP.
Q: What are the different ways of separating mixtures? Explain the difference between chemical…
A: If a mixture is made by mixing water and sugar then the mixture maintains the properties of both…
Q: Predict the major product of the following reaction. NH₂ 1a. OLI O 1b. MeCl 1a. CH3I (excess) K₂CO3…
A: -> Nucleophile can give Michael addition reaction to the alpha,beta - unsaturated carbonyl…
Q: The sugar industry uses barium hydroxide solution to refine sugar beets. What is the amount…
A:
Q: Oxygen molecules dissociate to form oxygen atoms. spontaneous nonspontaneous A tray of water is…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Question #1 ● ● ● # of moles of p-ethyl phenol used: (Show calculations) Amount (moles) of unknown…
A:
Q: 10. Ethanol OH weighing 3.56 g is burned in a calorimeter. The temperature rises from 15.C to C. The…
A: During the burning of ethanol the heat released is absorbed by water and a calorimeter. First of…
Q: Solve the dH of reaction in kJ for N₂O4 --> 2 NO2 Given the following information. • A rigid gas…
A: The balanced chemical equation is: N2O4 → 2NO2 ; ∆Hrxn = ?? (To be calculated) Since all of the N2O4…
Q: Question 15 What is the molarity of a solution containing 80 g of H 2SO 4 in 345 mL of solution? O…
A: The question is to find the molarity of solution prepared by dissolving 80 gram of H2SO4 in 345 ml…
Q: According to the graph below, The ratio for One Metal is to how many LIGAND? ( Write a whole number)…
A: Here Given that, In the spectrometric titration curve, By using linear regression, the intersection…
Q: neon effuses 2.77 times faster than a hypothetical form of molecular phosphorus under the same…
A:
Q: Compare Cl₂ gas and F2 gas at 300K. Which one of the following statements is true? The F2 gas will…
A:
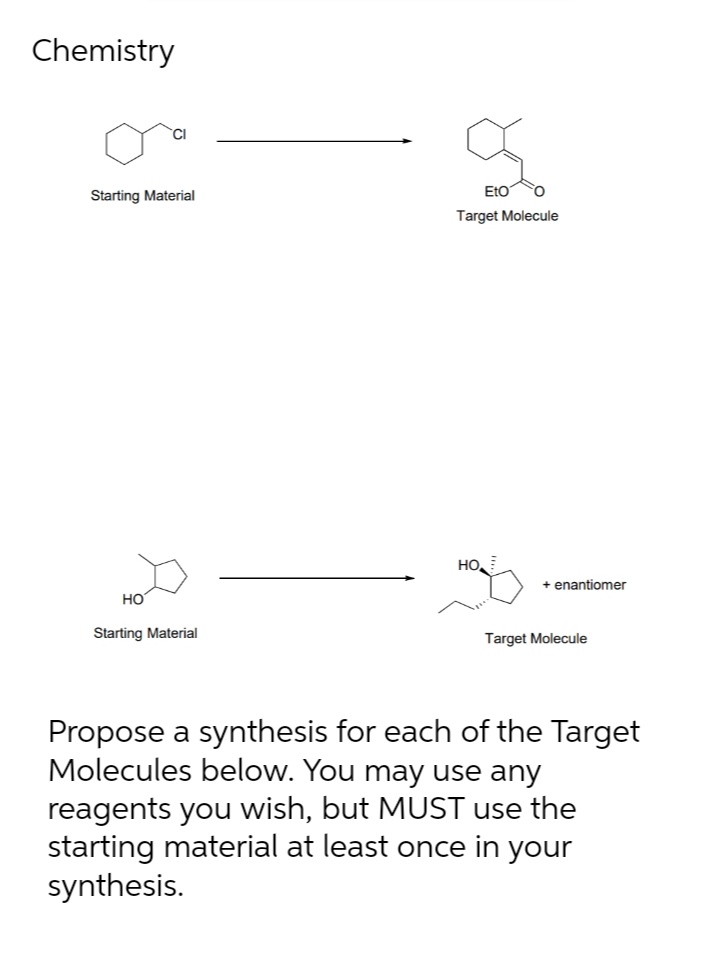
Q: 3. Propose a reasonable synthesis for the following transformation. H
A:
Q: For the reaction below, A2+B → A2B if ΔG°= -96.4 kJ•mol-1, what is the value of K at 298 K ?
A:
Q: 1. Determine the number of carbon atoms in the longest chain of each of the following, and name the…
A: According to guidelines we are supposed to answer only one question please ask rest of the question…
Q: Draw the structure(s) of the product(s) of the reactions below. Be sure to indicate any relevant…
A: We have to draw the products of the given reaction.
Q: The amount of work to compress a gas is 80J, 30 J of heat is given off to the surroundings.…
A: Given,Work to compress a gas = 80 JHeat given off by the system = 30 JRequired,…
Q: ed er A hot air balloon of 811m³ is made from 8.50kg of materials. The outside atmosphere is 1.00…
A: Given the mass of the materials of the balloon = 8.50 Kg Volume of the balloon, V = 811 m3 We have…
Q: Identify the most likely transition metal M: a. K3[M(CN)6], in which M is a first-row transition…
A:
Q: Question 2 Consider reaction A) Os (s) →20s (g) →OsO4(s) versus reaction B) Os (s) →20s (g) →…
A:
Q: 3. The equivalence point of the ethanoic acid titration occurs when pH-8.7. The ionic product of…
A: Given: The pH at the equivalence point = 8.7 Kw = 1.0 × 10-14 mol2 dm-6 We have to calculate the…
Q: A standard galvanic cell is constructed with H*|H₂ and Cu2+ |Cu half cell compartments connected by…
A:
Q: Use the Henderson-Hasselbalch equation to calculate the pH of a solution of HEPES buffer made from…
A: Given -> Volume of conjugate base = 150 ml Molarity of conjugate base = 0.4 mM = 0.4 × 10-3 M…
Q: (6) The pressure of the gas (mmHg) given by the following manometer is: (A) 793.0 (B) 751.2 (C)…
A:
Q: Write the chemical equations for the reaction of 2,4-Dinitrophenylhydrazine with acetone.
A: Here we are asked to write the chemical equation for the reaction between2,4-dinitrophenylhydrazine…
Q: Draw the structure(s) of the product(s) of the reactions below. Be sure to indicate any relevant…
A:
Q: ဝ a) H3NCHCNHCH₂CO₂ CH3 -N=C=S 1) NaOH 2) anhydrous CF3COOH 3) aq HCI HN- H CH3 CI + Hဒ္ဒNCH၇CO2H
A:
Q: Use the References to access important values if needed for this question. A student determines that…
A:
Q: If 150 mL of a 1.46 M solution is diluted to a volume of 200 mL, what is the final molar…
A:
Q: Ammonium oxalate dissociates when it dissolves in water. If the concentration of the oxalate…
A: Here we have to calculate the concentration of ammonium oxalate if the concentration of oxalate ion…
Q: 4. Write out the 1° structure for the following polypeptide: Ο Η ОН H₂N-CH-C-N-CH-C-N-CH-COO T CH-OH…
A:
Q: 3) Draw the product of the following reactions: a. H3CO- CH3 b. Draw the product of the following…
A:
Q: 1. Carbon monoxide is a toxic gas due to how it binds to the Fe in hemoglobin blocking the ability…
A:
Q: Question 42 Which is the major organic product of this reaction? A B. O AICI benzene C₁ D. ?
A: According to the bartleby guidelines for multiple questions in two different image, I can solve only…
Q: Consider the reaction PC15(g) PCl3(g) + Cl2(g). Calculate the equilibrium constant (Kp) for the…
A:
Q: 2. The student wants to conduct the same titration with chloric (1) acid, HOCI. The acid…
A: Given: The acid constant of the chloric acid is Ka=3.2×10-8 To find, Here when the sodium hydroxide…
Q: A pHmeter is calibrated using apH 4.00 buffer with 174.48 MV, PH 7.00 with OMV and PH 10.0 buffer…
A: We have given that A pH meter calibrated using p pH 4.00 buffer with 174.78mV , pH 7.00 buffer with…
Q: C. d. HO 공연 그 I HO HO OH
A:
Q: types of Electrodes
A: (1) Metallic – Metallic means consisting of partly metal or consisting entirely of metal. The…
Q: Provide the curved arrow mechanism of the reaction of 2-ethylcyclopentanone treated with pyrrolidine…
A:
Q: Given the following equation: 2 C4H10 + 13 O2 →→ 8 CO2 + 10 H2O How many grams of C4H10 are needed…
A:
Q: Provide proper IUPAC name for the following compound: es
A:
Q: Absorbance 14 1.2 1 0.8 0.6 0.4 0.2 Curve 1 Curve 2 Curve 3
A: From Beer- Lambert's Law: A= Є c l where, A= absorbance, Є= molar extinction coefficient…
Step by step
Solved in 3 steps with 3 images

- Unknown compound D,whose formula is C28H44O, is an alcohol that produces a compound whose formula is C28H50O upon catalytic hydrogenation with excess H2 over Pt.Indicate how many rings and pi bonds compound D contains based on the data given.Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Predict the mechanimsm and reactants to synthesize the picture below. Write legibly and ill give you a thumbs up.Chemistry Show how you would carry out the following disconnections. In each case, show whichbonds are being disconnected and show what the reagents would be for the resultantsynthons.The leaves of the Brazilian Tree Senna multijunga contain a number of pryidine alkaloids that inhibit acetylcholinterinase. Two recentyl isolated isomeric compounds have the strcture have the strcture shown below. (NOTE: M=293) Use the mass spectral data provided to determine the precise location of the hydroxyl group in each isomer. Isomer A: EI-MS, m/z(rel. int): 222(20), 150(10), 136(25), 123(100) Isomer B:EI-MS, m/z(re;. int): 236(20), 150(10), 136(25), 123(100)
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Explain how IR spectroscopy could be used to distinguish between polymers I-III shown below. In youranswer, you should specify the diffrences between the spectra of the polymers that would lead tounambigiguous identifcationsThe structures of two tertiary bicyclic chlorides (compounds 1 and 2) are shown below. Which of the following statements is correct?
- I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?The reaction of Compound B with O3, followed by Me2S gave two products, C and D (Scheme 1). The mass spectrums of products C and D are shown below. Identify the structures of products C and D. Provide explanation for your answer (based on mass spectrometry interpretation).C9H10O2: IR absorption at 1718 cm–1. Propose a structure with data given.

