Q: Draw the product of attached reductive amination reaction.
A: The product of the given reductive amination reaction has to be given.
Q: Rank the solubilities (from least to most soluble) of PbS, CuCl, HgS, CaCO3, and BaSO4, and explain ...
A: Compounds and thier ksp's are given as following - ★ PbS Ksp = 3.2*10^-28 ★ CuCl Ksp = 1*10^-6 ★ H...
Q: Draw the condensed structures of the compounds formedfrom (a) butanoic acid and methanol, (b) benzoi...
A: Condensed structures of any compound are the structures that can be written in a single line and wh...
Q: A solution of 4.85 g of a carbohydrate in 100.0 g of water has a density of 1.017 g/mL and an osmoti...
A: Mass of carbohydrate (Solute)=4.85 g Mass of solvent (water)=100.0 g Mass of solution =4.85 + 100.0...
Q: Please answer numbers 2 and 3 and please show the complete solution
A:
Q: How does hydrogen bonding effect OH and NH groups in an intrared spectrum compared to the IR peaks f...
A:
Q: put answer in this format
A: protons and electrons are elementary particle of atoms .
Q: Calculate the concentration of each species present in a 0.050-M solution of H2S. (Ka1 = 1.1 x 10^-7...
A: H2S is an example of a diprotic acid. It has two acidic protons therefore, two dissociation constant...
Q: A 5.00 mL tap water sample was measured out with a volumetric pipette, and added to a 25 mL Erlenmey...
A:
Q: Define about Polymerization ?
A: The definition is given in the following steps.
Q: Determine the percent sulfuric acid by mass of a 1.31 m aqueous solution of H2SO4.
A: The molality of the sulfuric acid solution is = 1.31. m We know that the molar mass of sulfuric acid...
Q: Which compound has the largest wavenumber of absorption (considering all the vibrations). Explain a)...
A: IR spectroscopy is used for the detection of functional groups in unknown compounds. It observes the...
Q: The first question was wrong please provide good answer
A: In the given molecule: Angle a: H-C-O Angle b: C-O-H So, angle a is around the central atom of carbo...
Q: Draw the products of attached reaction
A: Hydrolysis is a reaction in which the hydration of the compound is done to beak the bond. Unsaturati...
Q: 3. 4) Which substance is a mixture? (could be more than 1) (a) Gold. (b) Water. (c) Milk. (d) Fruit ...
A: Since you have asked multiple question we will solve the first question for you. If you want any spe...
Q: Plz do Asap...!
A: The rate constant of a first-order reaction can be expressed in term of time taken and the concentra...
Q: I'm having hard time predicting the product can you explain the mechanism?
A: Alcohols in presence of acids such as H2SO4 undergo an elimination reaction and form alkene as the m...
Q: Antifreeze solutions are aqueous solutions of ethylene glycol, C2H6O2 ( d = 1.12 g/ml) . In ...
A:
Q: A student wishes to determine the heat capacity of a coffee-cup calorimeter. After she mixes 107 g o...
A: Given mass of water added to the coffee-cup calorimeter(ma) is 107 g and initial temperature(tai) of...
Q: What is the major product of the following reaction?
A: Inorganic chemistry, Cyanide is used to increase the carbon numbers in a molecule. It is very toxic ...
Q: Answer a, b & c
A: Substance having strong intermolecular forces would have higher boiling point too.
Q: Balance the following equation and express the rate in terms of the change in concentration with tim...
A:
Q: The density of dodecane, C12H26, is 0.7495 g/mL. Calculate the volume (in mL) occupied by 3.405 mole...
A:
Q: A reaction of the form aAProducts gives a plot of inA) versus time (in seconds), which is a straight...
A: From the given plot type it is clear that the reaction is first order in nature.
Q: A one-dimensional polymer chain consists of 1200 segments, each 1.125 nm long. If the chain were ide...
A: The expression of root mean square separation of the ends of the chain is NL where N is the number o...
Q: Consider the balanced reaction 5 A + 6 B → 2 C + 3 D. If 3.629 grams of D (MW = 25.47) are produced,...
A: In Given Reaction : 5A + 6B → 2C + 3 D moles =massmolar mass Given mass of D = 3.629 g molar mass =...
Q: The standard molar entropy of benzene is 173.3 J K-1 mol-1. Calculate the change in its standard mol...
A: The given data contains, Standard molar entropy of benzene= 173.3 J/K.mol Temperature T1= 25°C=298 K...
Q: Which of the bonds shown in red in each compound or pair of compounds is shorter?
A: The answer for part (a) is given below. Kindly repost the other parts as separate one and with clear...
Q: (i) What is meant by chirality of a compound? Give an example.(ii) Which one of the following compou...
A: A compound is said to be chital if it exists in two stereoisomeric forms that are non-superimposable...
Q: How many grams of S are in 515 g of SO2?
A: The atomic mass which is denoted in grams indicates the gram atomic mass. The relationship between t...
Q: An organic chemist synthesizes the molecule below:(a) Which of the orientations of hybrid orbitals s...
A: In the above structure, the electron groups around carbon (C-1), sulfur, and oxygen atoms are 4. Hen...
Q: To identify a diatomic gas (X2X2), a researcher carried out the following experiment: She weighed an...
A: A numerical problem based on gas law, which is to be accomplished.
Q: Sodium is a highly malleable substance , while sodium chloride is not . Explain the difference in pr...
A: Metals are described as malleable (can be beaten into sheets) this is because of the ability of the ...
Q: Take pictures of each step of the procedure while you are performing the experiment. Submit the comp...
A:
Q: Draw structural formulas for the aminoalcohol and enamine formed in the following reaction
A: When amines react with aldehyde or ketone, water is eliminated giving imine derivatives.
Q: When a 4.31 g sample of liquid octane (C8H18) is burned in a bomb calorimeter, the temperature of th...
A: Gdhh
Q: The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of...
A: Given, salt A benzene diazonium chloride is dissolved in 10cm3 of water, cooled and added drop by dr...
Q: Write the structures of two specific alcohols.
A: Alcohols are generally denoted by R-OH. R represents any organic substituent where carbon and hydrog...
Q: I need help please (Not honor class) (Not grading)
A: Water is the best polar solvent and hexane is the best non polar solvent. Most of the ionic salts ar...
Q: Question is attached
A: Specific heat is defined as the amount of heat required to raise the temperature of one gram of a su...
Q: An ice freezer behind a restaurant has a freon leak, releasing 43.31 g of C2H2F3Cl into the air ever...
A:
Q: Draw the products formed when cembrene A is treated with O3 followed by CH3SCH3. Label each product ...
A: Alkene reaction with O3 and followed by CH3SCH3 is an ozonolysis reaction. The alkene bond breaks ...
Q: When cyclohexene is treated with m-chloroperbenzoic acid and H,0, trans-cyclohexane-1,2-diol is prod...
A: Given, The mechanism for the above reaction has to be proposed.
Q: The density of a gas is 1.43 g/Lg/L at a temperature of 23 ∘C∘C and a pressure of 0.789 atmatm. Calc...
A: Solution PV=nRT 0.789atm x 1L= n x 0.082 L.atm/mol.K x 296K n= 0.0325 mol mol= mass/ molar mass ----...
Q: The thermite reaction, used for welding iron, is the reaction of Fe3O4 with Al. 8 Al (s) + 3 Fe3O4 (...
A: The question is based on the concept of reaction stoichiometry. First, we determine the limiting rea...
Q: how much energy is (kcal) is required to melt 77g of ice at (0.0C) to liquid?
A: Given: The mass of ice=77 g Tempearature=0 °C The heat of fusion for water =333.6 J/g
Q: (S)-2-Bromobutane has a specific rotation of +23.1°. Suppose that a solution is made by dissolving 2...
A: At a given temperature, the chiral compound will rotate the plane polarised light at a particular an...
Q: Draw curved arrows and identify the thermal and photochemical products of the electrocyclic reaction...
A: Given: To determine the thermal and photochemical products of electrocyclic reaction of 2Z,4Z,6E-Oct...
Q: When ozone (O3) is placed in contact with dry, powdered KOH at –15°C, the red-brown solid potassium ...
A: Ideal gas equation is the combined result of simple gas laws such as Boyle’s law, Charles’ law and A...
Q: 1.20 mol of a nonvolatile, nonelectrolyte hydrocarbon is dissolved in 1,115.7 g of benzene. What is ...
A: Elevation in Boiling point is the rise in the boiling point of the solution on addition of solute to...
Draw the structure(s) of the monomer(s) used to make each
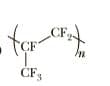
Step by step
Solved in 2 steps with 1 images


