When cyclohexene is treated with m-chloroperbenzoic acid and H,0, trans-cyclohexane-1,2-diol is produced. Propose a mechanism for this reaction, accounting for the observed stereochemistry. Hint: Recall what a МСРВА + Enantiomer H2O "OH peroxyacid does to an alkene.
When cyclohexene is treated with m-chloroperbenzoic acid and H,0, trans-cyclohexane-1,2-diol is produced. Propose a mechanism for this reaction, accounting for the observed stereochemistry. Hint: Recall what a МСРВА + Enantiomer H2O "OH peroxyacid does to an alkene.
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 36AP
Related questions
Question
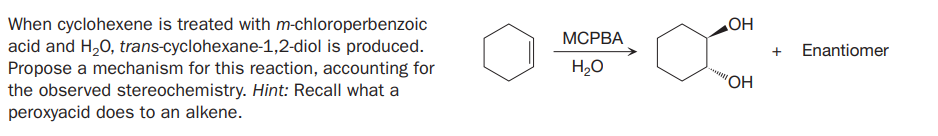
Transcribed Image Text:When cyclohexene is treated with m-chloroperbenzoic
acid and H,0, trans-cyclohexane-1,2-diol is produced.
Propose a mechanism for this reaction, accounting for
the observed stereochemistry. Hint: Recall what a
МСРВА
+
Enantiomer
H2O
"OH
peroxyacid does to an alkene.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 4 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning