Q: 5. Calculate AHrxn (28) using hess's law 2+ Ca(s)+2H2Om - Са (aд) + 20 (ад) + H2(g) AHrxu(28) = ? H…
A: We would use Hess's law to calculate ∆Hrxn of above reaction , using the given values of ∆Hrxn for…
Q: ueous solution of zinc chloride was 50.00 mL of 0.75 M aqueous soluti action. 3.14 kJ of heat is…
A: Given: Mass of zinc powder = 1.34 g Heat=3.14 kJ 0.75 M of HCl
Q: Given the data below, H°rxn for the reaction 3Cl2 (g) + PH3 (g) → PCl3 (g) + 3HCl (g) is…
A: Hess's law states that the amount of heat evolved or absorbed in a process is independent of the…
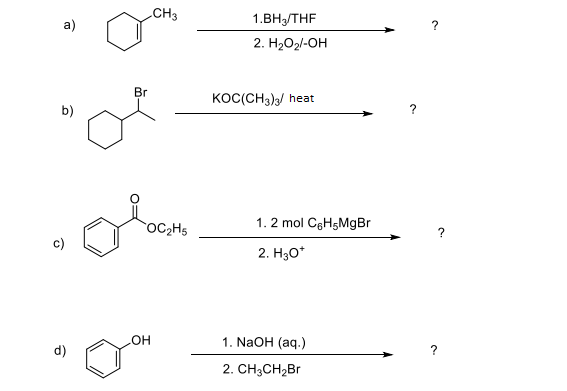
Q: O LI* H -Н20 1. ? OH- heat 2. H2O a. b. C. d.
A:
Q: I) AH>0 shows that reaction is an exothermic. II) Mole fraction of nitrogen (N2) in air is 0.21.…
A: We know, exothermic reaction releases energy, and endothermic reaction absorbed energy. So ∆H>0…
Q: Given the following equation, C3H8(g) + 5 O2(g) → 3 CO2(g) + 4 H2O(g) ΔG°rxn = -2074 kJ Calculate…
A: The desired equation is, 5C3H8(g) +25O2(g) →15CO2(g) +20H2O(g) ;∆G°=? The given…
Q: Mn(g) → Mn“(g)+2 e¯ Mn(s) → Mn(g) S(s) → S(g) S(g) +2 e→ s²(g) Mn (g) + S(g)→ MnS(s) Mn(s) + S(s) →…
A: Mn (g) →Mn 2+ (g) + 2e- +2230 kJ/mole Ionization energy (IE) Mn (s) → Mn (g) ………
Q: Given the following information, what is the AH°F (heat of formation) for Cu2S(s)? Thermochemical…
A:
Q: Stoichiometry find # of o geneale 6.81Kg grams 0, needed of Ilozinunbalanced reartion if The…
A:
Q: Automobiles and trucks pollute the air with nitric oxide, NO (9). At 2, 000 °C. the Key for the…
A: The relation between two equilibrium constants at two different temperatures is calculated by vant…
Q: From experiments, you know the following thermochemical equations: CaO(s) + 2HCl(aq) ---> CaCl2(aq)…
A: Given: CaO(s)+2HCl(aq)→CaCl2(aq)+H2O(l) ∆H1=-186 kJ ---eqn(1)CaO(s)+H2O(l)→Ca(OH)2(s)…
Q: Me CO2ME 1) NaOMe, heat; H3O* Ме 2) K2CO3, CH3CH2Br 3) H3O*, H2O, heat .CO2ME
A:
Q: At 1 atm, how much energy is required to heat 95.0 g H2O(s) 95.0 g H2O(s) at −22.0 ∘C−22.0 ∘C to…
A:
Q: re of 4 mol of H-(g) and 1.5 mol of Na(g) at 25°C and 1 atm or, a sample of 3 r at 25°C and 1 atm
A: A) Higher entropy is 2 mol He gas at 150 °c and 0.2 atm pressure. As temperature increases and…
Q: Consider the reaction A B with AH° = 15.3 kJ/molpm and the reaction B→ C with AH° = 91.3 kJ/molxn-…
A: •The desired reaction is, A→C ; ∆H° = ? • The given reactions are, (i) A →B ; ∆H1° =…
Q: Use Hess's law to calculate AG°,xn using the following information. CIO (g) + O3 (g) CI (g) + 2 O2…
A:
Q: Given the following data 2 ClF(g)+O2(g)→Cl O(g)+F2O(g) Δ H = 167.4 kJ 2 ClF3+2O2(g)→Cl…
A: It is given that three reactions have the change in enthalpies which are shown below with the…
Q: A 12 grams sample of methanol, CH,OH is burn(2 liters at room temperature). This results in a temp…
A: Mass of methanol = 12 g Initial temperature = 25oC final temperature =35oC
Q: 10. How much heat will be absorbed when 38.2 g of bromine reacts with excess hydrogen according to…
A:
Q: Calculate Δ H o rxn for the following: SiO2(s) + 4 HF(g) →SiF4(g) + 2 H2O(l) ΔH o f
A: Given, △H°fSiO2=−910.9 kJ/mol △H°fHf (g) =−273 kJ/mol △H°fSiF4 (g)=−1,614.9 kJ/mol △H°fH2O…
Q: Ozone, O3(g), is a form of elemental oxygen produced during electrical discharge.Is ΔH°f for O3(g)…
A:
Q: Consider the reaction: ICl(g) + Cl₂(g) → ICl₃(s). The ∆G(f)° of ICl(g) is -5.5 kJ/mol and the ∆G(f)°…
A: For the given reaction: ICl(g) + Cl₂(g) → ICl₃(s) ∆G(f)° [ ICl(g)] =-5.5 kJ/mol ∆G(f)° [ Cl₂(g) ] =0…
Q: 0.35 mol of HgO(s) decomposes to Hg(t) and O2(g) (A,H° for HgO =-90.83 kJ/mol) Enthalpy change = kJ…
A:
Q: Acetylene burns in air according to the following equation: C2H2(g) + 5/2 O2(g) →2 CO2(g) +…
A: The standard free energy of formation refers to the energy change that occurs when a compound is…
Q: Reaction Type? Enthalpy? (ΔH) Spont.? (-ΔG)? Yes, No, or Maybe (TD*) C5H12(g) + 8O2(g) -> 5CO2(g) +…
A:
Q: 1. CHJ 1. CH (1) 2. Ag20, H20 3. heat NH 2. Ag,0, H20 3. heat KI (m-n) CuBr EN
A:
Q: Calculate the amount of heat produced when 3.057 L of liquid acetonitrile (C,H,N) is burned in…
A: First calculate mass of acetonitrile by using density and then moles of acetonitrile.
Q: 7- Given the following data: 2H2 (g) + C (s) –→ CH4 (g) 2H2 (g) + O2 (g) → 2H2O (I) C (s) + O2 (g) →…
A: Given, The reaction with unknown free energy change is - Add the equation (2) and (3) and subtract…
Q: Concentration of hydroiodic acid (M) 2.10 Concentration of lithium hydroxide (M) 2.07 Calorimeter…
A:
Q: For the compund + H,O 아 +H,0
A: As you have asked multiple, we will answer the 1st part only. If you want any specific question,…
Q: CO° C. After the car has been driver ncreases to 50° C. Assuming that t hat air behaves as an ideal…
A: To determine the pressure of the tire when the temperature of the tire is increased 20oC to 50oC…
Q: Reaction A (Mg + HCI) Trial 1 Trial 2 ss of Calorimeter (g) 2.384 2.431 16.973 16.842 ss of…
A: 1.) Mass of Hydrochloric acid is calculated as : Mass of HCl = ( mass of calorimeter + HCl solution…
Q: PHNH2, CH3COOH Meo OMe heat EtO2C. CH2 CO2Et + i) CH;COOH H3C H2N CH3 ii) [0]
A:
Q: Given the heats of formations (ΔH°f) listed below, ΔH°rxn for the reaction Ca(OH)2 + 2H3AsO4 →…
A:
Q: b) Using the following data of heats of combustion in cal/g mole, calculate the following: (a) (b)…
A: The enthalpy changes associated with the various reaction is given above.
Q: Consider the reaction A →B with AH° = 43.19 kJ/molxn and the reaction B → C with AH° = 81.98…
A: We know that according to Hess's law, the heat of reaction for a given reaction is the sum of the…
Q: ΔH°rxn
A:
Q: A Al(s) + 12(s) = All3(s) CH4(g) + 4C12(g) = CC14(1) + 4HCI(g) C Fe,03(s) + 3C0(g) = 2Fe(s) +…
A: The correct option is A, B, C.
Q: Given that H2(g)+F2(g)⟶2HF(g)ΔH∘rxn=−546.6 kJH2(g)+F2(g)⟶2HF(g)ΔHrxn°=−546.6 kJ…
A: Since by multiplying reaction 1 with 2 and subtracting reaction 2 from 2 X reaction 1, we will get…
Q: A) 1. Excess CH,I /OH NH2 B) 1. H,O, / heat Br (1) NaCN (2) LİAIH,/H,O
A: Given Since you have posted a question with multiple sub-parts, we will solve first three sub-parts…
Q: = following the Mg) from calorimetry if magnesium reacts with HCl steps (and sample problem 9.6) to…
A:
Q: Part 3: Enthalpy of Neutralization HCl(aq) NaOH(aq) a. Formula of the acid b. Formula of the base c.…
A: Mass of calorimeter + acid = 7.1495 g Mass of empty calorimeter (A) = 2.175 g Mass of Acid solution…
Q: Calculate the ΔGo for X, in kJ, from the data given below at 327 K: 2X(g) → X2(g) So…
A: Since the ΔGo is given by ΔGo = ΔHo - TΔSo Where T = temperature in K = 327 K
Q: Q1/ Calculate the AH, for 3C,H2 (9) AHc (C,H2) = -1.30 J mol1 AHc (C,H.) = -3.303 J mol1 --- C,Hs…
A: The reaction is, 3C2H2 (g)→C2H6 (g) Given that: ∆Hc(C2H2) = -1.30 J/mol ∆Hc(C6H6) = -3.30 J/mol To…
Q: Reaction A (Mg + HCI) Reaction B (MgO + HCI) Trial 1 Trial 2 Trial 1 Trial 2 Mass of Calorimeter (g)…
A: The heat released or evolved in a reaction or process is calculated by the formula Q=m×c×ΔT where,…
Q: How do I get H reaction per mole of MgO consumed ?
A: We have to find out Delta H of a reaction per mole of MgO consumed.?
Q: Titanium(IV) oxide is commonly used as a white paint pigment. The compound is produced by the…
A: For a chemical reaction, the enthalpy of the reaction is the difference between products and the…
Q: The combustion of how many moles of ethane (C2H6) would be required to heat 561 g of water from…
A:
Complete the reactions given below
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Aleks data for PbCO3 is 7.40 x 10^-14.Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)F,g,a? :)
- Ff.22. With explanation...Provided the following information determine Δ?ΔHformation of C2H6(g). C(graphite) + O2(g) ⟶⟶ CO2(g) Δ?=−393.5??ΔH=−393.5kJ H2(g) + 1212O2(g) ⟶⟶ H2O(l) Δ?=−285.8??ΔH=−285.8kJ C2H6(g) + 7272O2(g) ⟶⟶ 2CO2(g) + 3H2O(l) Δ?=−1560.7??Pls help ASAP. Pls look at the correct answer and do the second sked question.
- Why do you multipy the # of H2S mols by 4, when 4 corresponds to the reactory amount of O2?The logic there is somewhat confusing to me.I understand how/why you found out the mols of the equation but dividing the sample's MM by the equation confuses me.Ag video: youtube.com/watch?v=npqooiE6DhEPls help ASAP. DO BOTH THE FUCKING QUESTIONS
- Please answer this stichometrey question and put stepsQUESTION 5 Calculate ΔG°rxn for the reaction below at 25°C. CH3CH2OH (ℓ) + 3O2 (g) → 2CO2 (g) + 3H2O (ℓ) Report your answer in kJ/mol to one decimal place. ΔH°f (kJ/mol) S° (J/(K·mol)) CH3CH2OH (ℓ) -277.7 160.7 O2 (g) 0 205.14 CO2 (g) -393.5 213.7 H2O (ℓ) -285.8 69.1pls help asap on all asked questions.

