When solutions of silver nitrate and potassium chloride are mixed, silver chloride precipitates out of solution according to the equation AGNO3 (aq) + KCI(aq)→A£C1(s) + KNO3(aq) Part A What mass of silver chloride can be produced from 1.70 L of a 0.271 M solution of silver nitrate? Express your answer with the appropriate units. • View Available Hint(s) mass of AgCl = 66.0 g Submit Previous Answers v Correct
When solutions of silver nitrate and potassium chloride are mixed, silver chloride precipitates out of solution according to the equation AGNO3 (aq) + KCI(aq)→A£C1(s) + KNO3(aq) Part A What mass of silver chloride can be produced from 1.70 L of a 0.271 M solution of silver nitrate? Express your answer with the appropriate units. • View Available Hint(s) mass of AgCl = 66.0 g Submit Previous Answers v Correct
Chapter4: Calculations Used In Analytical Chemistry
Section: Chapter Questions
Problem 4.26QAP
Related questions
Question
Pls help ASAP. Pls look at the correct answer and do the second sked question.
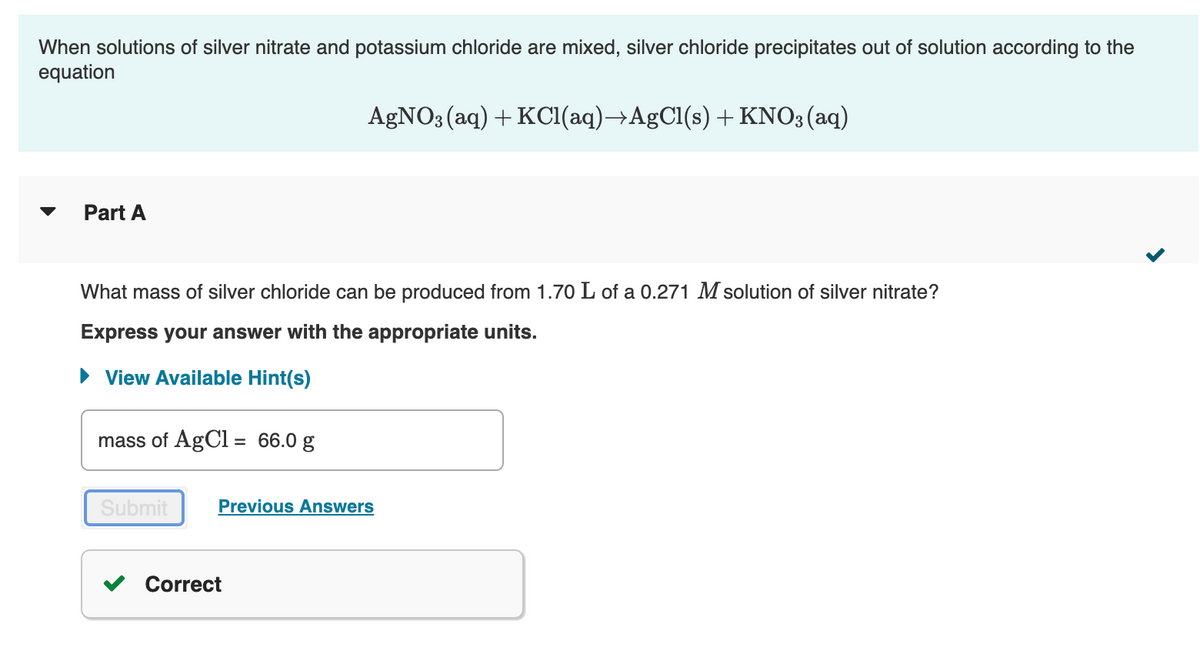
Transcribed Image Text:When solutions of silver nitrate and potassium chloride are mixed, silver chloride precipitates out of solution according to the
equation
AGNO; (aq) + KCI(aq)→AgCl(s) +KNO3(aq)
Part A
What mass of silver chloride can be produced from 1.70 L of a 0.271 M solution of silver nitrate?
Express your answer with the appropriate units.
• View Available Hint(s)
mass of AgCl = 66.0 g
Submit
Previous Answers
Correct

Transcribed Image Text:The reaction described in Part A required 3.56 L of potassium chloride. What is the concentration of this potassium chloride
solution?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning