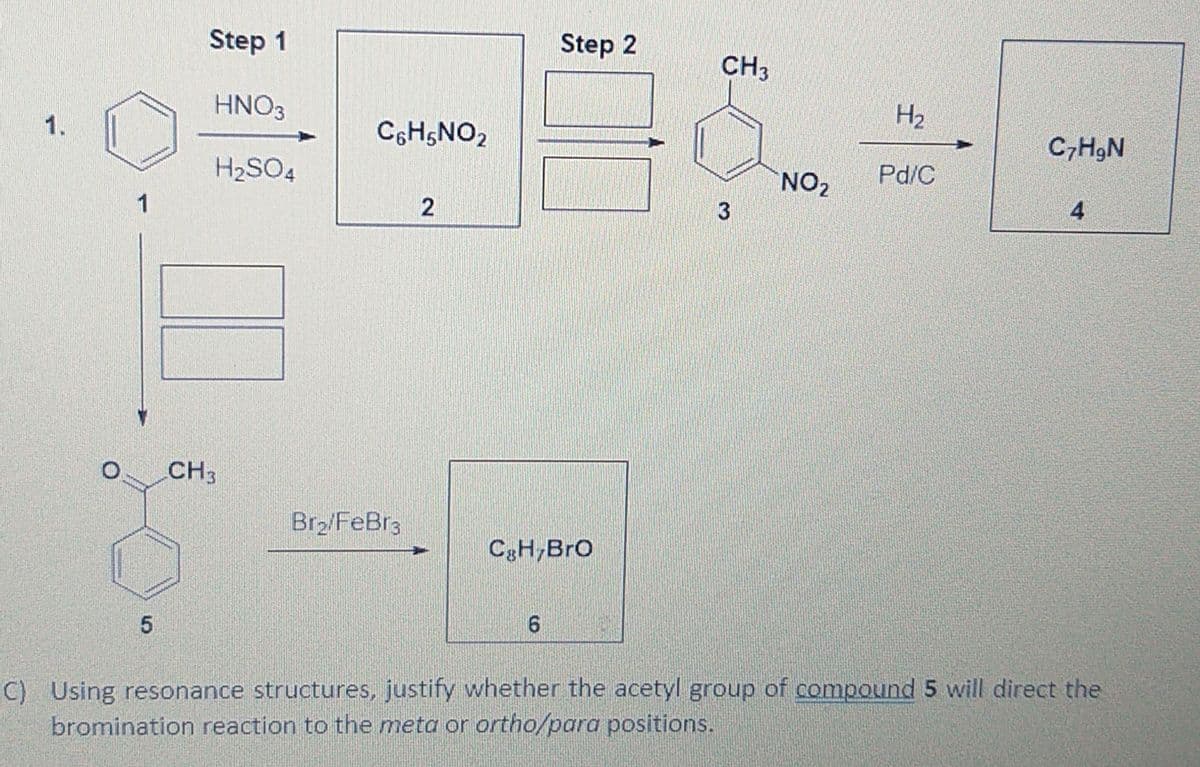
CH3 Br2/FeBr3 C3H,BrO 6. C) Using resonance structures, justify whether the acetyl group of compound 5 will direct the bromination reaction to the meta or ortho/para positions.
Q: Explain the following result. Although alkenes are generally more reactive than alkynes toward…
A: Given, Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction…
Q: Would you expect the substituent N+(CH3)3 to more closely resemble N..(CH3)2 or-NO2 in its…
A: The resemblance of N(CH3)2 or-NO2 with N+(CH3)3 based to the rate effect and regioselectivity in the…
Q: A ?? в
A: The synthesis has been done by the following step- allylic bromination, hydrogenation of alkene and…
Q: Write the structure of the major organic product isolated from the reaction of 3-hexyne with (a)…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three sub…
Q: Which of the following alkylhalides is used in the synthesis of 2-hexanone a. CH;CH2B.,…
A:
Q: Write the structure of the principal organic product formed in the reaction of 1-propanol with each…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: H2O, H-SO4 1. NaNH2, NH, 2. CH,CH,Br Compound E Compound F
A:
Q: Designing Synthetic Schemes with Aromatic Rings: For the reaction below, begin with benzene and…
A:
Q: Aniline reacts with bromine in the absence of a Lewis acid to give monosubstitution products. Draw a…
A: Electrophilic aromatic substitution reactions are those reactions in which replacement of positively…
Q: For each horizontal row of substituted benzenes, indicate a. the one that is the most reactive in an…
A: The reactivity toward the electrophilic substitution reaction depend upon the substituent that…
Q: Draw the structure of two different epoxides that would yield 2-methyl-2-pentanol…
A: Epoxides: These are organic ether group present in cyclic form. These have O as main heteroatom.…
Q: Give the products (if any) expected from the treatment of each of the following compounds with ozone…
A: Ozonolysis:Alkene reacts with ozone which cleaves the double bond followed by work up with dimethyl…
Q: он pyridine 1. SOCI, SO2 ont Aqueous acetone H20 H3C HBr 2. H3C Br H OH optically active racemic a =…
A: The questions are based on the concept of organic chemistry. It involves determination of type of…
Q: 3. Write in the product of 4-bromo-3-cyclopentyl-1-octyne with the following reagents: 2. НаОг. ОН…
A: Addition of HBr follow markovnifov rule
Q: Write the major alkene formed in the following E2 reactions.
A:
Q: Pyrrole undergoes electrophilic aromatic substitution preferentially at the 2 position as…
A:
Q: Draw the four resonance structures formed during bromination of methoxybenzene, CH3OC6H5, with…
A: Methoxy group in methoxy benzene is a ortho-para directing group. Therefore, bromination of methoxy…
Q: Imagine the reaction coordinate diagram for the reaction of methoxide ion in MeOH (CH3O¯, an…
A: Elimination Reactions are the reactions in which a small molecule is eliminated in the presence of…
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: NaNH2 with liquid ammonia acts as a strong base. It is used in those reactions where hydrogens are…
Q: True or false? In an SN2 reaction with HBr, 1-propanol will react faster than 2-propanol to give the…
A: SN2 reaction refers to the substitution reaction between a nucleophile and an electrophile. A…
Q: Provide the major aromatic product(s) for the reactions below. so, H,50, KMno, NaOM H20, heat NaH,…
A:
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: In organic reactions, chloronium ion formed, which can then be attacked by a nucleophile.
Q: 2. Provide the final product for each of the following reactions: a) 1-methylcyclohexene reacts with…
A: For the all the given reactions we can expect alcohol as the product and the position of OH group is…
Q: Give two sets of reactants (each set including an alkyl halide and a nucleophile) that could be used…
A: Please find below the first set of reactants
Q: Without reading ahead in L Chapter 20, state whether it should be possible to carry out each of the…
A: The reactivity of the acid derivative depends on the partial positive charge on the carbonyl carbon…
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: The major organic product isolated from the reaction of 3-hexyne with BH3 followed by CH3COOH is…
Q: Explain the following result. Although alkenes are generally more reactive than alkynes towards…
A: Given,Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction…
Q: 5. Consider the synthesis of 2-butanone from butyne: Hg2+ || CH3CH,-C=C–H CH;CH,-Ĉ-CH3 H3o* (iv)…
A: The given reaction represents the nucleophilic addition in alkynes. It is performed in the presence…
Q: 2. Provide products corresponding to the following reactions (remember to apply Markovnikov's rule):…
A:
Q: Give the structures of the substitution products expected when 1-bromohexane reacts with(a)…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: Addition of hydrogen halides to alkyne The alkyl halides such as HCl, HBr, or HI undergo addition…
Q: Write the resonance structures of methyl benzoate that show that the CO2CH3 group is a deactivating…
A: Given molecule is methyl benzoate C6H5CO2CH3 To prove, it is a deactivating group in ortho and para…
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A:
Q: 3. Write in the product of 3-bromo-4-chloro-1-pentyne with the following reagents: 1. BH, 2. HO2,…
A:
Q: 2. Are the methoxy groups on benzene likely to direct substitution to the ortho and para positions,…
A: Answer - a benzene with a group like OCH3 attached to it. the carbocations for the “ortho” and…
Q: Propose a synthetic scheme to convert bromobenzene into HOCH,- CH:
A: Bromobenzene can be converted into the given compound by the following steps: Friedel craft…
Q: 5) If the reactant provided is neutral what can be done in basic conditions to produce a…
A: In a SN2 reaction a nucleophile attack the leaving group contaning Carbon and remove leaving group…
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: The given reaction is,
Q: A tautomerization reaction can occur with an imine in a way analogous to that of a ketone or…
A:
Q: Does perchlorate (ClO4-) have an extremely low nucleophilicity because it participates in a lot of…
A:
Q: b) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically…
A:
Q: the compound can be synthesized by an SN2 reaction. Suggest a combination of haloalkane and…
A:
Q: provide the name of the major product(s) of the given reactions. For disubstituted aromatic rings,…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Complete these electrophilic aromatic substitution reactions. Where you predict meta substitution,…
A: The given electrophilic aromatic substitution reactions to be completed.
Q: Draw structural formulas for the major product(s) formed by reaction of 3-hexyne with each of these…
A: The given reactant is 3-hexyne. The structure formula of the product formed when 3-hexyne reacts…
Q: i. When But-1-ene (CH3CH2-CH=CH2) is treated with HBr in the presence of a peroxide, give the name…
A: (i) Given reaction,
Q: Four alkenes are formed from the E1 reaction of 3-bromo-2,3-dimethylpentane and methanol. Draw the…
A: In the presence of methanol, tertiary alkyl halide undergoes E1 reaction in which carbocation is…
Q: 2. What product is expected to form from the SN2reaction of 1-bromopentane with а. КОН? B. Nal?
A:
Part c
Step by step
Solved in 2 steps with 1 images

- Could someone help me with ranking these with how reactive they would be with an SN2 reaction? Thanks :)In parts 1 and 2 draw the two organic products of this reaction, showing any nonzero formal charges. Then, in part 3 answer the question regarding purification of the reaction mixture. 1. Draw the product with the higher molecular weight here: 2. Draw the product with the lower molecular weight here:the mechanism of halogenation of thiophene, when the halogen added to the position 2 and 3 , also provide the resonance structure and final product
- Identify the two most likely sites for an S2 reaction to take place in the molecule shown below.For SN1 Explain if 3o ( tertiary) alkyl halides reacted (fastest or slowest) explain why. Be sure to explain if alkyl halides did not react or did react and why. 3o (tertiary) compounds listed are: 2-chloro-2-methylpropane (see picture) Base your explanations on the following considerations: the nature of leaving group, the effect of structure, steric hindrance and any other feature.Assign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).
- Please help me with the organic chemistry problem below: Consider the reaction below: (Check the attached image) (it is between Furan and maleic anhydride, a DIels-Alder reaction) a) Will this reaction for an endo product (with a melting point of 80-81 degrees) or the exo product (with a melitng point of 114 degrees)? b) Carefully explain why the product must have been formed the way it did (exo or endo). c) Provide a mechanism for this reaction.1. State the effects of substituents on a benzene derivative towards further aromatic substitution. 2.Based on the above, suggest the various types of substituents that can be attached to Benzene.Is NaOCH2CH3 also a strong nucleophile? if so and Br wasn't a 3 degree alkyl halide, would it also undergo an Sn2 reaction? what would the major product be then?
- Order each of the following sets of compounds with respect to SN1 reactivity.Repeat Problem 14.36 for [8]annulene. Draw the π MO energy diagram for [10]annulene, similar to those in Figures 14-19 and 14-21. Fill up the orbitals with the appropriate number of π electrons. Based on this diagram, should [10]annulenebe aromatic or antiaromatic? Explain.Is the major product for 2-butanol and HCl racemic since its SN1?

