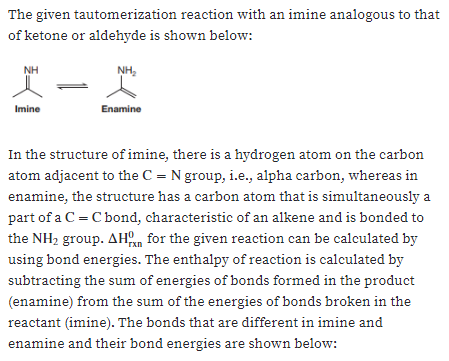
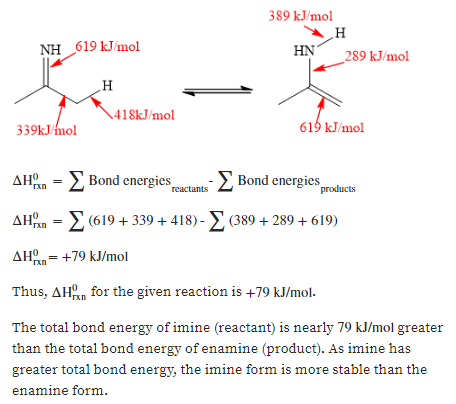
A tautomerization reaction can occur with an imine in a way analogous to that of a ketone or aldehyde. Using the appropriate bond energies from Section 1.4, estimate AHan for this reaction and determine which form is more stable, the imine or enamine. NH NH2 Imine Enamine
Q: clopropanation of allylic ethers as shown in the scheme below. Ph Et₂Zn + CICH₂l Toluene 3 h, -20°C…
A:
Q: For the Electrophilic Aromatic Substitution lab you performed an iodination reaction on salicylamide…
A:
Q: Give the major product(s) of the following reactions, if there is a reaction. For electrophilic…
A: The reactions are as follows:
Q: OMe HO 1. LIN(iso-propyl)2 OMe 2. H20
A:
Q: Draw the SNl mechanism of the reaction between 2,5-dimethyl-2,5-hexanediol and hydrochloric acid.…
A:
Q: The two Grignard reactions and the reduction reaction below are all believed to have similar…
A: Reaction involving organic compounds are called organic reactions. Organic reactions are of…
Q: Alkyl halides 1-chlorobutane 2-chlorobutane Allyl chloride 2-chloro-2-methylpropane…
A: Reactivity of SN2 reaction based on Nature of alkyl group
Q: 1. The dramatic reaction below was reported in the Evans synthesis of colchicine in 1981. MeO CO₂Me…
A: Note : Acid catalyzed electrophilic aromatic substitution followed by dehydration. ( see below).
Q: 1) How will you describe whether any compound has been oxidized or reduced? Support the answer with…
A: Hi, we are supposed to answer one question. To get the remaining questions solved please mention the…
Q: Which set of conditions for the reaction given below would provide the largest amounts of…
A: We have to find a suitable reagent that will give the largest amount of substitution product.
Q: From the list below, select all of the benzene derivatives with activating groups attached. CHO…
A: Activating groups on benzene increases electron density of benzene via +M effect whereas…
Q: Write the structure of the principal organic product formed in the reaction of 1-propanol with each…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Question 4
A: A nucleophile is always in situ to attack an electrophilic center. As carbonyl carbon is…
Q: Aniline reacts with bromine in the absence of a Lewis acid to give monosubstitution products. Draw a…
A: Electrophilic aromatic substitution reactions are those reactions in which replacement of positively…
Q: Provide the reagent sequence for the following reactio O 1) NaNH2, 2) MeCl, 3) NaNH2, 4) EtCI O 1)…
A: Correct answer is (1) NaNH2 , (2) MeCl , (3) NaNH2 , (4) EtCl , (5) Na, NH3
Q: For each horizontal row of substituted benzenes, indicate a. the one that is the most reactive in an…
A: The reactivity toward the electrophilic substitution reaction depend upon the substituent that…
Q: NaOH H20 HO
A: NaOH gives here Michael addition. If its gives direct addition then two OH group present in same…
Q: он pyridine 1. SOCI, SO2 ont Aqueous acetone H20 H3C HBr 2. H3C Br H OH optically active racemic a =…
A: The questions are based on the concept of organic chemistry. It involves determination of type of…
Q: Fifteen different monosubstituted benzenes are represented below. How many of them are "deactivated"…
A:
Q: Identify the compounds in Figure 7 that are an anhydride, amide, acyl chloride, and ester. Ph Ph Ph…
A: Anhydrides, amides, acyl chlorides and esters all are derivatives of acids.
Q: Br Br II Order of increasing reactivity
A: Given that : We have to explain the observed rate of reaction for the following compounds in an SN1…
Q: Imagine the reaction coordinate diagram for the reaction of methoxide ion in MeOH (CH3O¯, an…
A: Elimination Reactions are the reactions in which a small molecule is eliminated in the presence of…
Q: Electrophiles for the electrophilic aromatic substitution reactions have to be very strong to react…
A: In the presence of sulphuric acid, protonation of nitric acid occurs in the first step. Loss of…
Q: OH 1. aqueous H2SO4 CH3 CH3 2. H2SO4 OH H2O 130° a = Proton transfer d = El Elimination f= SN1…
A: Hydration of alkene: Alkene gives hydration reactions in presence of an acid catalyst. In this…
Q: Predict the product of the Robinson annulation reaction between the diketone and alpha,…
A:
Q: 9. Provide the major dehydration product of the following reaction. Recall which of the four…
A:
Q: Reaction 1: Reaction 2: CI CI AICI3 AICI3
A: Friedel craft reaction- Benzene is a nucleophile and addition of electrophile does not takes place…
Q: Without reading ahead in L Chapter 20, state whether it should be possible to carry out each of the…
A: The reactivity of the acid derivative depends on the partial positive charge on the carbonyl carbon…
Q: c) For the following substituted benzenes, please rank their relative rates of reactivity in an EAS…
A: Electrophilic aromatic substitution reactions are organic reactions wherein an electrophile replaces…
Q: CH3 Br2/FeBr3 C3H,Bro 5 6 Using resonance structures, justify whether the acetyl group of compound 5…
A:
Q: Provide the missing structures of products A and B for the following reaction sequence. NH2 1)…
A: → Aniline Aniline is primary amine
Q: An important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and…
A: Given statement: NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is…
Q: 2. Write the structural formula of the major organic product formed in each of the following…
A: a) The product formed from the given reaction is 2-methyl pentan-3-ol.
Q: This
A:
Q: Predict the product of the Robinson annulation reaction between the diketone and alpha,…
A: first base will abstract the acid hydrogen and form thermodynamic enolate which was then attack with…
Q: In both examples below the reactants shown are combined to bring about a nucleophilic substitution…
A:
Q: Determine the order of reactivity for (CH3),CHCH,Br, (CH;);CBr, and CH,CHCH,CH, in substitution…
A: The answer to the given question is as follows---------- In sodium cyanide medium the reactions…
Q: 1.The reduction of butanoic acid can form butanal and butan-1-ol. The boiling points of these…
A: Reduction of carboxylic acid produces alcohol and aldehyde. C=O converts to aldehyde and and when…
Q: 1. H2SO4 Br +] H20 HO, NaBr 2. H3C-O H,C-O-Li Li f= Sn1 Nucleophilic substitution g= SN2…
A: The reactions given are,
Q: Q6 Butryic Acid + Trimethylamine The following equation describes the reaction of butyric acid with…
A:
Q: 2. The hydrolysis of a nitrile under acidic or basic conditions to form a carboxylic acid parent…
A: 2. a. We have to tell why the reaction in the scheme (1) is easily done under mild conditions and…
Q: (ii) Rank the carbonyl compounds given below in terms of their relative reactivity with sodium…
A:
Q: Using your knowledge of general chemistry and organic chemistry, answer the following questions:…
A:
Q: Explain WHY/HOW all please - Propanol CANNOT protonate ethylamine - 3-methylheptan-3-ol CANNOT be…
A: Since you have asked a question with multiple subparts, we will answer only first three subparts for…
Q: b) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically…
A:
Q: Rank the alkyl halides in the following marine natural product in order of increasing reactivity in…
A: Applying concept of SN1 reaction on reactivity of alkyl halide.
Q: Complete these electrophilic aromatic substitution reactions. Where you predict meta substitution,…
A: The given electrophilic aromatic substitution reactions to be completed.
Q: Which site in the molecule below is most reactive in electrophilic aromatic substitution? (а) OH (b)…
A: Answer:- This question is answered by using the simple concept of directive effect of disubstituted…
Q: 2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O to form compound W. Compound X…
A: The details solution for this is provided below in attach image.


Step by step
Solved in 3 steps with 4 images

- Alcohols react with sulfonyl chlorides to form sulfonate esters. Only the O-H bond of the alcohol is broken in the reaction, and so no inversion of configuration occurs. The resulting sulfonate esters are reactive in SN1 and SN2 reactions since the sulfonate group is a very weak base and is therefore a good leaving group.Draw curved arrows to show the movement of electrons in this step of the mechanism.Arrange the structure on the image with regard to the reactivity towards nucleophilic acyl substitution 1 being the least and 3 being the mostEnolates are formed by deprotonation of an α-carbon hydrogen. Answer the following questions about enolate formation. In the molecule shown, select the α-carbon hydrogen that would be removed to form an enolate when NaOEt is used as a base. Draw the thermodynamic enolate that results for the molecule in Part 1. Draw only the enolate resonance form that includes a formal charge on the α carbon. Be sure to indicate that formal charge as well as any lone pair of electrons in your answer.
- 1) How will you describe whether any compound has been oxidized or reduced? Support the answer with suitable examples. 2)Why carboxylic acid with a carbonyl group at 3rd position can be decarboxylated? 3) Explain why electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in Pyridine it takes place at C-3 position? 4) List the following esters in order of decreasing reactivities towards hydrolysis with reason: Methyl benzoate, p-cyano methyl benzoate and p-hydroxy methyl benzoate 5)LDA is the base of choice for carbonyl compound to completely convert into enolate. Why?Provide a step-wise synthesis for the following: image 1 The last step of the synthesis needs to be a nucleophilic aromatic substitution reaction with a secondary amine.Rearrangements can occur during the dehydration of 1° alcohols even though no 1° carbocation is formed—that is, a 1,2-shift occurs as the C— OH2+ bond is broken, forming a more stable 2° or 3° carbocation, as shown in Equation [1]. Using this information, draw a stepwise mechanism for the reaction shown in Equation [2]. We will see another example of this type of rearrangement in Section 16.5C.
- Which or which of the statements given below is correct. I) Maleic anhydride is a carboxylic acid derivative and its reaction with water is a reduction reaction. II) Fumaric acid and maleic acid are stereoisomers of each other III) Since fumaric acid has a more stable structure than maleic acid, its boiling point is higher. A. Solo I B. I and III C. II and III D. I, II, III E. Solo IIISecondary alcohols are often dehydrated in an E2 reaction to give an alkene. Elimination follows Zaitsev's rule to give the more substituted alkene as the major product. Since the reaction occurs via an E2 mechanism, there is no risk of rearrangement of the carbon skeleton as could possibly occur if the elimination occurred via an E1 mechanism with a carbocation intermediate.Draw curved arrows to show the movement of electrons in this step of the mechanism.The two Grignard reactions and the reduction reaction below are all believed to have similar mechanisms. Use the curved arrow formalism to draw a single mechanism for these reactions using :Nuc-1 to indicate the nucleophile. The reacations are: cyclohexylmagnesium chloride + acetone in ether --> 2-cyclohexyl-2-propanol phenylmagnesium bromide + acetone in ether --> 2-phenyl-2-propanol Acetone + NaBH4 --> 2-propanol
- When allyl bromide is refluxed with magnesium metal in ether solvent, the product formed is 1,5-hexadiene. (C6H10). What is the curved arrow mechanism for this reaction?If the alkenes below were possible products of a elimination reaction ,how would you rank their likely order of formation from most likely to least likely?Complete the curved arrow mechanism of the following double elimination reaction when 2,2-dichloro-3,3-dimethylbutane is treated with two equivalents of sodium amide and heated in mineral oil. A.) Use three curved arrows to show the elimination of the first hydrogen chloride. B.) Use three curved arrows to show the elimination of the second hydrogen chloride. C.) Complete the structure of the organic product formed from the double elimination. The ammonia and chloride ion formed in step 1 is not drawn for clarity.



