Q: Identify compounds A and B:
A: The compound A is the ethylene and hydronium ions. The compound B is methane which is derived from…
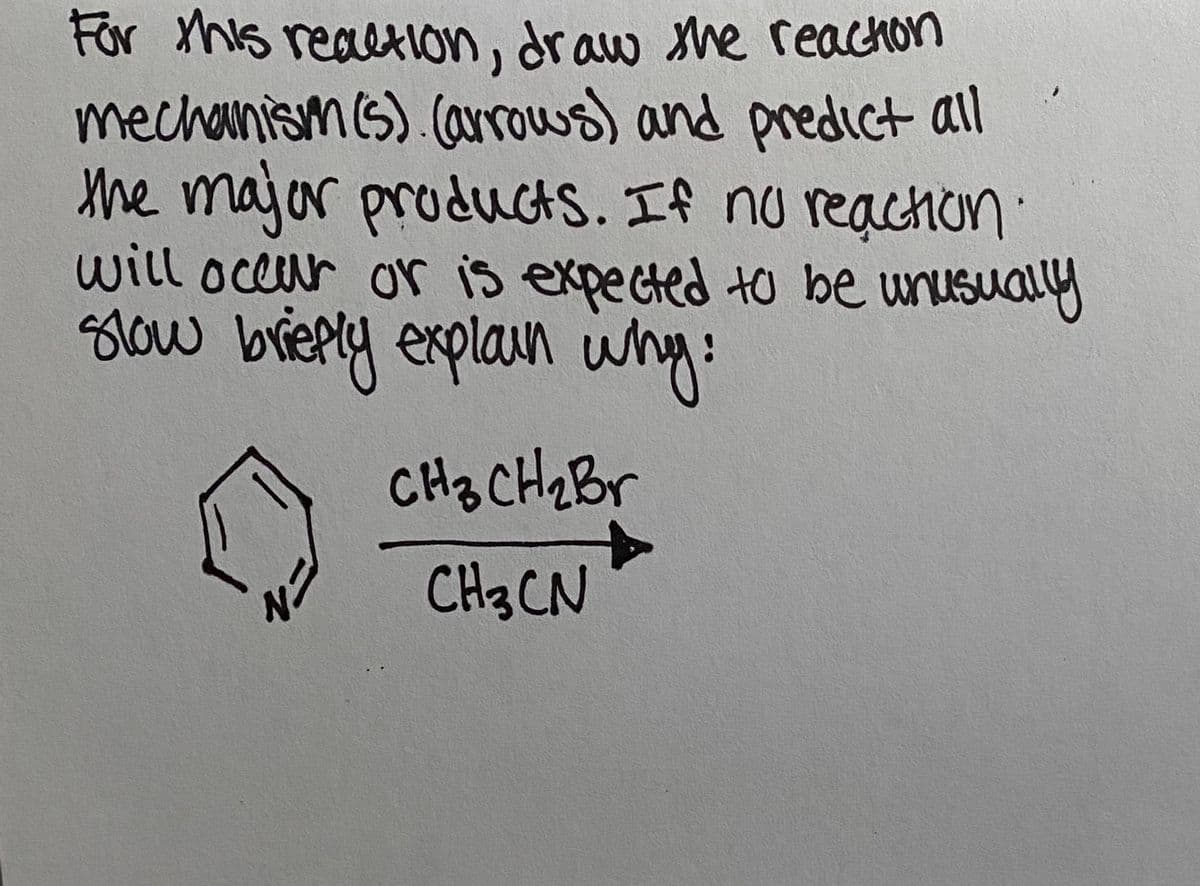
Q: H3C H3C H3C N' H CH3 CI SOCI2 pyridine OH
A:
Q: Identify the catalyst in each equation.
A: a.
Q: CH3 CH3COCI/ AICI3 H₂C. CH3
A: Friedel–Crafts Acylation Involves formation of a complex between the Lewis acid and the chlorine…
Q: CH3CH2CH=CH2 + HBr
A: Protonation of alkene leading to the formation of carbocation intermediate. Attack of halide as…
Q: H3C. CH3 CH3 FIGURE 5
A:
Q: C H3C CH3
A: Explanation Step-1 IUPAC system of nomenclature We know about very well IUPAC nomenclature, any…
Q: 1. CH3 2. НС %3 СССНЗ CH3 3. H3CCH2CHCH2C = CCH2CH3 CH3 A. 5.
A: Answer:- This question is answered by using the simple concept of naming of the organic compounds…
Q: NAOCH3 H3CO O a.
A:
Q: 1. Name the following cc CH2=CHCH,CH=CH2 1) » CH;=CH-CH=CH2 2) 3) CH2=C=CH2
A: According to the answering guidelines, I'm posting the solution for the first 3 problems. Kindly…
Q: a (CH3)2CHCH=C(CH3)2
A: Select the longest carbon chain Numbering Name = prefix + word root + suffix
Q: H2C= CI CH2(CH2);CH2 CI
A:
Q: CH3 CHBV clu.goc Br Br
A: The key components associated with a molecule and are responsible for providing the notable chemical…
Q: CH3NH2 H3O*
A: Since there is a lone pair electron on N in CH3NH2 and the carbon making C=O in ketone is having…
Q: H3C. OH H3C. H3C но. CH3
A: Step 1 Dehydration. Step 2. Ozonolysis of alkene to give aldehydes. Step 3. Reaction of aldehyde…
Q: CH3 Cl2 AICI3 Brz. FeBr3 3. CH2CH3 HNO3, H,SO4 H3CH2C 1. 2.
A:
Q: CH3 CH3 CH3 CH3 H2C H2C CI
A: Positively charged species is a cation and negatively charged species is an anion. The above arrow…
Q: CH3 H3C H3C Name:
A: Rules for IUPAC Nomenclature of Aldehyde: 1. Choose the longest chain for parent naming. Aldehydes…
Q: [D] Br CH ₂ 2 HA C 1|||||| CH₂OH B CH3 | с
A: -> For priority of groups we see first of all atoms directly attached with chiral carbon. ->…
Q: Please name (a) CH3-CH2-CH2-CH-CH3 (b) CH3
A: Alkanes are saturated hydrocarbons. If an alkane has a missing hydrogen atom, then the group is…
Q: H CH₂ CH 3 I -CH3 CH ₂3
A:
Q: (II) CH3 nd
A: The pairs given are,
Q: a. CH,CH20CH, CH3 b. CH, CH2CH2 CHz OH
A: Given isomers are: a) b) What type of isomerism is involved in them?
Q: CI H3C, HO H3C. HCI + H2O + H3C CH3 H3C CH3
A: The above Reaction is between Alcohol and the Hydrogen Halide This is the type of nucleophile…
Q: a. H2C=CH2 b. (CH3)2C=CH2 c. CH3CH=CHCH3
A: We have two tyoes of reactions for haloalkanes , namely SN1 And SN2 Reaction. These reactions are…
Q: compound CH4 CH2F2 H20 NH3
A: Compounds CH4 CH2F2 H2O NH3 What to determine? Lewis structure Hybridization Electronic…
Q: CH3 CH3 HO- CH3 CH2
A:
Q: 1. CH3CH2MgBr 2. H*
A: Attack of nucleophile Protonation(acidic workup)
Q: 1.) (CH3)2CuLi 2.) CH3B 3.) H3O+
A: step 1 is nucleophilic attack of organometallic reagent (CH3)2CuLi . this attack is at 4th position…
Q: OH CH3 H3C он b) CH H3C c)
A:
Q: Name: CH 3 H3C- CH – CH 2- CEN
A: For naming of a compound, 1st numbering the compound so that substituent gets least numbering.…
Q: CH H3C
A: Those carbon which are considered in root chain is marked as star. Functional group is nitrile here.…
Q: NaOCH3 ? CH3OH
A:
Q: CH3 + KBRO3 + HBraq. НОАС H3C
A: Concept: KBrO3 is a oxidizing agent which can oxidized Br- to Br2 in a acidic solution. The…
Q: CH2CI
A: There is an energy barrier that separates the energy level of reactants and products. The activation…
Q: b) H3PO4 HOCH3 CH3
A: First of all alkene react with acid (H3PO3) and carbocation formed then if rearrangement possible…
Q: CH3 ONa H3C `CH3
A:
Q: „CH3 CH,CH2COCI AICI3 он PBR3 CH NH,NH2 pyridine IZ O=U
A: organic reaction mechanism helps us to understand the possible product
Q: Explain Chlorination: CH3CH2CH3 + Cl2
A: Chlorination is the addition of chlorine to a structure. Chlorination of alkane gives a mixture of…
Q: H3C. CH3 CH3 1 CH3
A: Here we have to prepare the following compounds by using an electroplie ( alkyl halide) and organo…
Q: CH3 CH3CHCH3 + Br,
A:
Q: 3CO OCH3 H3CO OCH3 CI + H3C CH3 H3C CH3
A: ANSWER : Tertiary butoxide act as as base which abstract more acidic alpha H-atom from reactant.…
Q: -CH3 2 CH3NH2 h. H3C-CH,-
A:
Q: NH3 H3C CH H3C CH3
A:
Q: Pls help ASAP. WRITE THE IUPAC NAME
A: IUPAC ( International union of pure and applied chemistry ) is an international organisation that…
Q: A) SOC1₂ (CH3)₂CHOH pyridine C1 lot "ylon B) سللية بلده D)
A:
Q: CH,OH + CH3C C Nat?
A: The hydrogen bonded to the sp hybridized carbon becomes very acidic in nature. In the given problem…

Step by step
Solved in 2 steps with 2 images

- Using chair confirmations, draw the final product of each molecule. Pls explain how are you arrived at the answerthank you!what is degree unstaturation of the following ? and draw 2 structures per each numberDraw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2O
- In addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the conguration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.In addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.Predict the major product (or products) for each. Provide the stereochemistry if needed. pls answer all
- 1 -Bromopropane on treatnent with alc KOH produces?Please identify priorities of functional groups and name following molecules based on thier stereochemistry as R or S1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.
- Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain why1. Draw the organic product (ignoring stereochemistry) 2. State whether the process is overall oxidation, overall reduction, or neither.In addition to organic halides, alkyl tosylates react with organocuprates (R2CuLi) to form coupling products R–R′. When 2° alkyl tosylates are used as starting materials (R2CHOTs), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.

