Chapter15: Benzene And Aromaticity
Section15.SE: Something Extra
Problem 17VC: Azulene, an isomer of naphthalene, has a remarkably large dipole moment for a hydrocarbon (μ = 1.0...
Related questions
Question
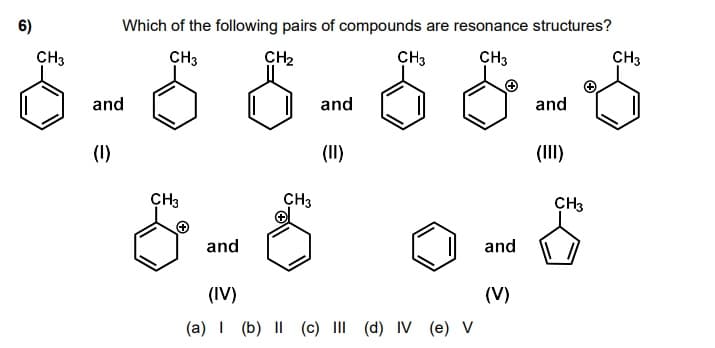
Transcribed Image Text:6)
Which of the following pairs of compounds are resonance structures?
CH3
CH3
CH2
CH3
CH3
CH3
and
and
and
(1)
(II)
(III)
CH3
CH3
CH3
and
and
(IV)
(V)
(a) I (b) I| (c) III (d) IV (e) V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

