Q: The decreasing order of boiling point of the following alcohols is * 1-pentanol < 2-methyl-2-butanol…
A: When branching increases then boiling point of same molecular formula is decreases. So,
Q: Vhich of the following is the common name for this alcohol? CH3- -C CH3 CH3 1. sec-butyl alcohol 2.…
A: Alcohols are the compounds having hydroxyl group bonding to an aliphatic carbon. The common name of…
Q: Which of the following molecules can hydrogen bond to another molecule of itself? Which can hydrogen…
A: Hydrogen bonding is a special type of dipole-dipole attraction between molecules, not a covalent…
Q: When heated with H2SO4, both 3,3-dimethyl-2-butanol and 2,3-dimethyl-2-butanol are dehydrated to…
A: On the basis of mechanism, chemical reactions can be classified as substitution reactions, addition…
Q: What is produced when most aldehydes undergo Grignard reaction? O A. primary alcohol B. secondary…
A: Grignard's reagent is an organometallic reagent that gives very useful products by reacting with…
Q: Classify the alcohol shown. CH H,C-C-CH,CH, O primary O secondary O quaternary tertiary None of the…
A: The structure of the primary alcohol is--- Here, the alcoholic -OH group attached with carbon, that…
Q: What type of alcohol is (S)-2-methylbutan-1-ol? o primary secondary o tertiary o quaternary
A: A primary alcohol is an alcohol in which the hydroxy group is bonded to a primary carbon atom. A…
Q: 1. Which alcohol has a higher boiling point? a. (i) 2-methylpropan-2-ol or (ii) butan-2-ol b. (i)…
A: Boiling point of alcohols: Instead of having a similar molecular mass alcohols having the higher…
Q: 13. What is the product of an aldehyde reacting with 2 molecules of an alcohol in the presence of a…
A: The reaction of aldehyde with 1 molecule of alcohol in the presence of strong acid results in the…
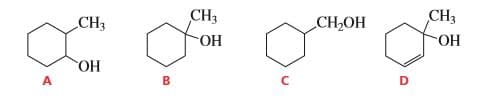
Q: Which of the following alcohols dehydrates the fastest when heated with acid?
A: The alcohols form a carbocation when heated with an acid. This carbonation is an intermediate which…
Q: What products are formed when each alcohol is oxidized with K2Cr2O7? a. CH3CH2CH2CH2CH2OH
A: This is a basic oxidation reaction of alcohol in presence of Potassium dichromate(acidic). Here, the…
Q: Draw the products formed when each alcohol is dehydrated with H 2SO 4.
A: Given alcohol,
Q: redict the relative ease of dehydration of the following alcohols ОН CH3 .CH2 H3C CH H3C- -C- CH3…
A:
Q: What products are formed when each alcohol is oxidized with K 2Cr 2O 7? In some cases, no reaction…
A: K2Cr2O7 is a potent oxidising agent like KMnO4. Potassium dichromate can oxidise alcohols and…
Q: Clear All CH;CH,C-H Lowest boiling point CH,CH2CH2CH3 Intermediate boiling point Highest boiling…
A:
Q: 6. Draw a circle around the compound in each horizontal row with the highest boiling point and draw…
A:
Q: Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with…
A: Hydrogen bonding- The interaction between electropositive ‘H’ atom of first molecule and…
Q: Examples: Dehydration of Alcohols Complete the following reactions: CH3CH2CH2OH H2SO4 140°C CH3CHCH3…
A: In chemistry, dehydration is a chemical reaction in which water molecule is eliminated from the…
Q: A- Each of the following alcohols is namea incorrectly. Give the correct IUPC name for each alcohol.…
A:
Q: Which of the following compounds is expected to give a positive Tollen's test? HC-CH2-CH3 CH…
A: Tollen's test given by aldehyde.
Q: Draw all the products of dehydration of the following alcohol, and predict which one is major…
A: The structure of CH3CHOHCH2CH(CH3)2 is; Dehydration refers to the loss of water.
Q: Draw the products formed when each alcohol is dehydrated with H 2SO 4. Use the Zaitsev rule to…
A: Since you have posted a question with multiple sub parts, we will solve only first three sub parts…
Q: 13. Which of the following has the highest water solubility? b. 1-butanol a. butanal C. 1-hexanol d.…
A: We know Polar is soluble in polar solvent. So the compound having highest polarity will have highest…
Q: Complete the example of Fischer esterification. Assume that the alcohol is present in excess.
A: The acetic acid is treated with 3-methyl-1-butanol in the presence of an acid to form isopentyl…
Q: or a. CH;(CH,),CH, b. CH-C–CH,CH;CH, CH-C-CH, or CH-CH-CH,CH,CH, OH
A: a. CH3COCH3 will be more soluble in water as compared to CH3(CH2)2CH3. b. CH3CH(OH)CH2CH2CH3 is more…
Q: Which alcohol is the most soluble in water? A CH;CH2CHOHCH3 B CH;CH2CH2CH2OH C (CH3)2CHCH2OH D)…
A:
Q: Rank the following according to predicted water solubility. Enter a number between 1-7 where 1 =…
A: Alcohols are more soluble than ethers which are more soluble than alkanes. This is due to their…
Q: 12.60 Which of the following aldehydes or ketones are soluble in water? (12.3) |3| a. CH-CH,-C-CH,…
A: B acetaldehyde is soluble in water
Q: Which one of the following alcohols would dehydrate most rapidly when treated with sulfuric acid? OH…
A: As, the speed of dehydration will be proportional to the stability of carbo-cation formed after the…
Q: Which of the following compounds would be most soluble in water? CH CH-CH CH3 CH-CH-CH-C-+H O…
A: They functional group is highly polar and allows the C3H7COOH acid to form strong intermolecular…
Q: OH CH-CH3 H2SO4 Heat I or OH CH-CH3 H2SO4 Heat II
A:
Q: 2. What alkenes could be produced, if each of the following alcohols were dehydrated? 2-propanol Ke…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: Which of the following alcohols would dehydrate the fastest when heated with acid? CH CH CH3 CH2OH…
A: In order to determine which of the following undergoes a faster dehydration or elimination reaction,…
Q: What is the classification of the following structure? CH3 H3C-CH2-CH2-CH-OH OA. Primary Alcohol O…
A: The name of the given compound is as follows - 2-Pentanol
Q: Rank each set of compounds in order of increasing boiling points.ethanol, dimethylamine, dimethyl…
A: Boiling point : It is the temperature in which liquid will turn into vapor. Boiling point depends…
Q: What two alkenes give rise to each alcohol as the major product of acid-catalyzed hydration?
A: Acid catalyzed hydration requires the reagent water in the presence of an acid (say sulfuric acid)…
Q: Solve this attached file
A:
Q: Which of the following compounds would be more soluble in water? A CH3CH2-N-CH2CH3 CH3-N-CH3 CH2CH3…
A: Amines can form hydrogen bond with the water molecule so their solubility is affected by the…
Q: In the Cr* oxidation of cyclohexanols, it is generally true that sterically hindered alcohols react…
A: Oxidation reactions are one of the major class of organic reactions. In oxidation reactions,…
Q: What is the formula of the hemiacetal of methanol and propanal? A CH3CH2CH(OCH3)OH CH3CH(OC2H5)CH2OH…
A:
Q: 1. Give the IUPAC names for the following alcohols. (а) ОН OH (Ь) ОН (с) НО CH3CHCH2CHCHCH3…
A: Rules for IUPAC nomenclature: 1) select the longest carbon chain as parent chain. 2) identify the…
Q: Which of the following is true regarding the boiling points of alcohols having the same number of…
A:
Q: What is produced when ketones undergo Grignard reaction? A. primary alcohol O B. secondary alcohol O…
A:
Q: . What is the structure of the cyclic hemiacetal that is formed from the compound below there's an…
A:
Q: Which of the following would have the lowest boiling point? a. 2-methyl-2-butanol (2-metil-2…
A: Please find your solution below : Boiling point increases with increase in the intermolecular forces…
Q: estion 9 Which compound below has the highest boiling point? H3C CH3 нн H-C-C-O H H CH3CH2 O- CH2CH3…
A:
Q: What is the formula of the hemiacetal of methanol and propanal? A CH3CH2CH(OCH3)OH B CH3CH(OC2H5)OH…
A: Hemiacetals are formed by the action of alcohols on aldehydes.
Q: Which of the following compounds can form hydrogen bonds with 2-Butanol? ОН CH3-CH-CH2-CH3 2-Butanol…
A:
Q: List the products of each alcohol reaction. CH3 a. CH,-C-OH CH, NazCrO b. CH3-CH-CH;-CH2-OH c.…
A: The objective of the question is to write the products of the given reactions.
Q: Which of the following compounds would be most soluble in water? c. CH-CH-CH-CH-OH a. CH;-CH-CH C-H…
A: Solubility of a compound follows the rule of like dissolves like. This means that polar solute…
Which of the following alcohols dehydrates the fastest when heated with acid?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


