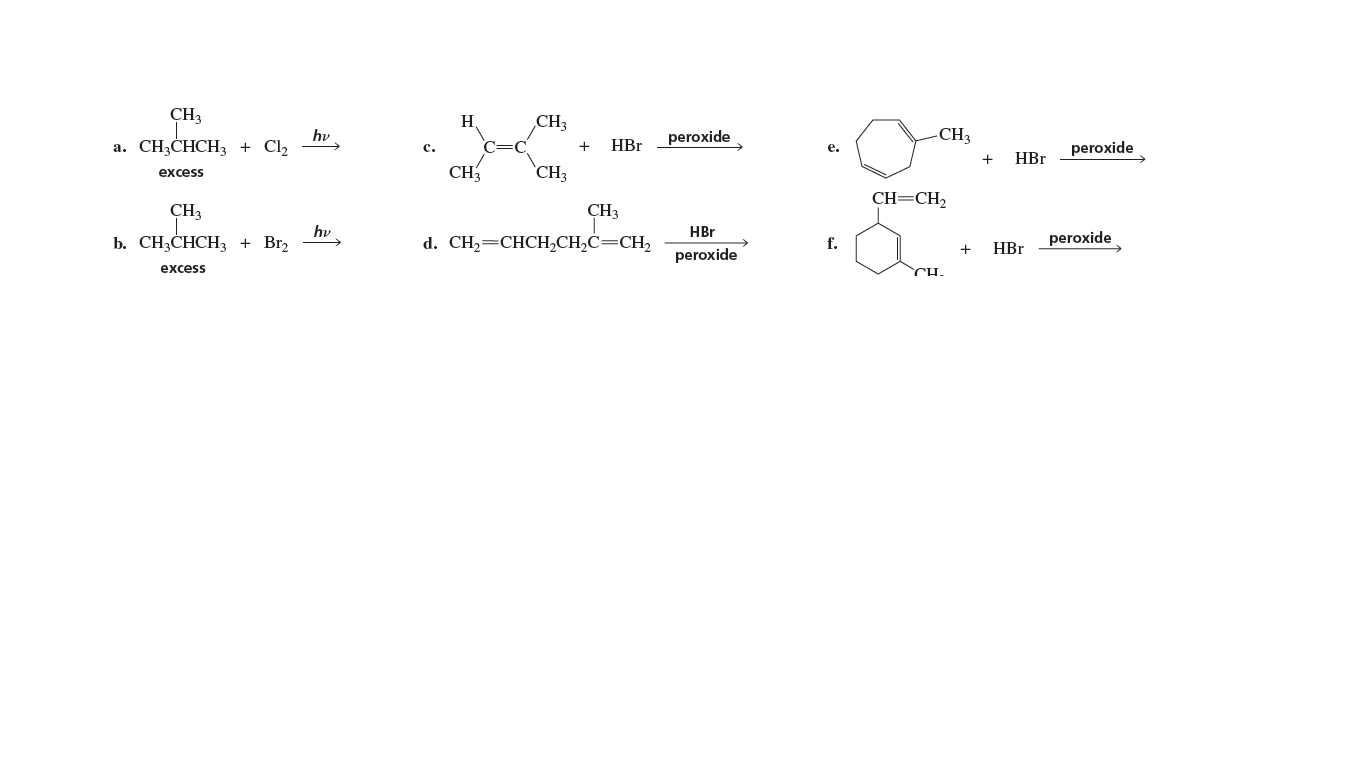
CH3 H CH3 hy а. CH,CHCH, + Clz CH3 peroxide HBr peroxide с. е. HBr CH3 CH3 excess CH=CH2 CH3 CH3 hy b. СН,СHCH + Brz HBr d. CH—CHCН,СH,С—СH, f. peroxide + HBr peroxide excess CH.
Q: Consider the following reaction at 299K. Sn²¹ (aq) + Pb (s) ---- Sn (s) + Pb²+ (aq) Which of the…
A: Given the cell reaction is: Sn2+(aq) + Pb(s) ----> Sn(s) + Pb2+(aq) Here Sn2+(aq) is undergoing…
Q: 10.3 Chlorination of Met Which reaction conditions are best to convert methane (CH4) into CCI4? O…
A:
Q: 14. Acetylene gan, CH, can be prodaced by the seachion of cala CaC, () + 2H,0 - How many lites of…
A:
Q: 1. Calculate Delta H° at 25°C for each of the unbalanced reactions below: a) C2H4 (g) + H2 (g) C2H6…
A: Concept: ∆H° =Σ ∆H°f (products) -Σ ∆H°f (reactants)
Q: In an aqueous solution, ethanal undergoes the reaction CH4CHO (aq) + H2O (l) -> CH3CH(OH)2 (aq). At…
A: The equilibrium constant for a reaction is expressed in terms of the ratio of the concentration of…
Q: In cach reaction box, place the best reagent and conditions from the list provided. 1) Br 2) 3) 4)…
A:
Q: Provide the missing compounds and reagents in the reaction scheme. Identify reagent 5. он reagent 2…
A:
Q: 2. Zn(s) | Zn²+ (0.05 M) || Zn²+ (1 M) | Zn(s) Calculation for E (cal.) (eq. 11): E = 0.0592 K:…
A: Calculating Ecell using Nernst equation. Anodic concentration =0.05M. Cathodic concentration =1M.
Q: The reactions of [PICI4]2- with NH3 (reaction I) and of [PTC14]2– with [NO2]- followed by NH3…
A: Answer: option d: cis -[PtCl2(NH3)2]; II: trans-[PtCl2(NH3)(NO2)]- is a correct option
Q: а. Cr (aq) + Cla (g) + Сr207 (ад) + CI (аф) e° = V b. РЬ?+ (аg) + Сa(s) — Са** (аq) + Рb(s) V
A: Calculate oxidation number of Cr in Cr2 O7 2- Let the oxidation number be x A. Split the…
Q: Calculate the ∆H of the following reaction. Show the complete and step by step solution. 1. 2C(s) +…
A:
Q: Consider the following reactions. 2CH4 (g) ⇌ C2H6 (g) + H2 (g) Kc = 9.5x10–13…
A: Given: (i) 2CH4 (g) ⇌ C2H6 (g) + H2 (g) Kc1 = 9.5x10–13 (ii) CH4 (g) + H2O (g) ⇌…
Q: d) Calculate the atom economy for Ethylene Oxide (CH;CH2O) preparation CH2=CH2 + Cl2 +H2O CICH,CH2OH…
A: Atom economy - It is a measurement of the amount of starting materials (reactants) that end up as…
Q: Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by…
A:
Q: HCI H2O2 d. to 1. Hg(OAc)2, H2О е. 2. NABH4 Br2 f. Lindlar's catalyst, H2 g.
A: Answer:- This question is answered by using the simple concept of chemical reactions of organic…
Q: Nibromethane undergoes combustion CH3 NO₂(1) + according to - - O2(g) A -CO₂(g) + H₂O(g) + - N₂cg- a…
A:
Q: The following reactions: Pb2++ 2l--->Pbl2 2Ce4++ 2l--> l2 + 2Ce3+ HOAc + NH3--> NH4+ + OAc - are…
A: Formation reaction Formation reaction is the formation of one mole of a substance from its…
Q: 10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH;Br + H2S 2…
A: The equilibrium reaction taking place is given as, => CH3Br + H2S ------> CH3SH + HBr Given:…
Q: H2 Pd/C H2 Lindlar's catalyst Na NH3
A: Product of the following reactions are
Q: Given: H2SO3(g) → H2O (g) + SO2 (g) Keq = 100 What will…
A: Equilibrium constant of a reaction, Keq can be written on the basis of law of action of mass. It is…
Q: 4
A: The C-D bond is strong compared to C-H bond which makes C-D bond hard to break. Therefore, compound…
Q: 1. n-Butanol (CH3CH2CH2CH2OH) and t-butanol ((CH3)3COH) are converted to their corresponding…
A: Nucleophilic-Substitution reaction : It is substitution reaction where a neuclephile attack a…
Q: Given: 10 g of o-hydroxybenzoic acid (density: 1.44 g/mL, MW: 138.2) 20 mL wood alcohol (density =…
A: Here 10 gram of o-hydroxybenzoic acid react with 20ml wood alcohol (CH3OH) of density 0.792 g/ml .…
Q: -Er a) H b) MgBr 1. 2. H3O+ ai (second step is just a workup) c) OH D3C- F3C CF3 • of you wholes CD3…
A: a. Aldehyde + alcohol in the presence of acid gives ketal. b. Grignard addition to keto FG. c.…
Q: Create sywthesis worng at least your owN 6or the following reagants: ) Towes 2) LIAIRY )…
A:
Q: 16 What is X in the following reaction? * (i) X CH,-C=C-CH, (ii) H*/Zn CH,-C-C-CH, I| | HNO3 02 03…
A: The question is based on the concept of organic reactions. We have to identify the reagent.
Q: ________ is reduced in the following reaction: Cr2O72- + 6S2O32- + 14H+ → 2Cr3+ +…
A: Loss of electron is called oxidation process and gain of electron is called reduction process. The…
Q: How do I calculate moles used in this lab from the information in the table?
A: The mass of nitric acid used is calculated below with the help of density equation in which m is the…
Q: Methane, CH4, is a potent green house gas. The only significant sink for methane is its reaction…
A:
Q: 1 mole Br /2 moles H,/Pt 2HC1 1 mole H2 (a) Lindlar's cataly st NANH2 -CH3C=CH 1. disiamylborane 2.…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: What are the reagents used and why?
A: NBS in presence of alkene undergoes benzylic halogenation if benzylic hydrogen is present. H2SO4…
Q: 2 C5H10 (1)+15 O₂ (g) → 10 CO₂ (g) + 10 H₂O(l) a. Calculate 4H of the reaction. b. Calculate 4S of…
A: Dear student , since you have posted multiple parts questions we will allow to solve only first…
Q: 10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH,Br + H2S CH3SH…
A: a. The Keq at room temperature (25°C) is 2.335. GivenCH3Br+H2S⇌CH3SH+HBr∆G=2.1KJmol=-2100…
Q: Calculate the ∆Hrxn for the reaction below: MgCl2 (aq) + 2 KOH (aq) ---> Mg(OH)2 (s) + 2 KCl (aq)…
A: Given reaction is- MgCl2 (aq) + 2 KOH (aq) → Mg(OH)2 (s) + 2 KCl (aq) ∆Hreaction of it is given by=…
Q: What is the % of H2O if 102g H2) is produced from 14.6g H2 and excess O2?
A: Given Mass of H2O = 102 gram Mass of H2 = 14.6 gram Percentage yield =?
Q: Exercice 3: Equilibrer les équations suivantes Ni H2SO4 H2 Niz(SO.)3 a. +] -> b. NH4F AlCl3 NHẠCI…
A: The complete balanced equation : b. 3NH4F + AlCl3 →3NH4Cl +AlF3 c. 3Na2CO3 + 2H3PO4 →2Na3PO4 +3H2O +…
Q: CH3CH2CH2CH2CI (1 mole) ? OMe AIClg, heat O Meo Meo Meo OMe O Meo OMe O There is no reaction under…
A: The reaction is the aromatic substitution reaction . The position of the entering group is directed…
Q: Acetic acid: k1.75x10 Ammonia: k=1.8x10s Hydrofluoric acid: 7.2x10+ Hydrogen sulficde: ke5.7x10…
A: #2: For acetic acid, CH3COOH(aq), Ka = 1.75*10-5 For hydrogen sulfide, H2S, Ka1 = 5.7*10-8 , Ka2 =…
Q: Bromine in methylene chloride is added in stoichiometric proportions to cyclopentane, What color is…
A: Bromine in methylene chloride is used for the determination of unsaturated compounds. In the bromine…
Q: Do you expect Step 2 to be easier than Step 1? Why or why not?
A: The reaction of bromobenzene with nitric acid and sulfuric acid results in the formation of…
Q: Find ΔHrxnΔHrxn for the following reaction: N2O(g)+NO2(g)→3NO(g)N2O(g)+NO2(g)→3NO(g) Use the…
A: If A+B →C+ D ∆H=xthen C+ D →A+ B ∆H=-xand2( A+B →C+D ) ∆H=2x
Q: b) Write the products for each of the following reactions (remember to add phase labels for each…
A: Given, Correction: MgNO3(aq) formula is incorrect. The correct formula is Mg(NO3)2(aq). i)…
Q: Consider the given almost irreversible reactions below: HSO3– + CO32– ➜ SO32– + HCO3– HNO2 +…
A: Given reactions are : 1. HSO3– + CO32– ➜ SO32– + HCO3– 2. HNO2 + SO32– ➜ HSO3- + NO2– Which of…
Q: Ch4+2O2->CO2 +2H2O Do I understand this correctly ? If I burn 1m3/hr of CH4 with 2m3/hr O2, do…
A: Methane reacts with oxygen to form carbon dioxide and water. The equation for the balanced chemical…
Q: (e) Provide alternative to the following conventional reaction schemes and state the major advantage…
A:
Q: H2O2 + KMNO. + H2SO. → O2 + MNSO. + K2SO, + H2O The products that were formed without charge…
A:
Q: In each reaction box, place the best reagent and conditions from the list. 2. H,C H CH;CH, он но…
A: The reaction given is,
Q: Predict deltarH for this reaction: C2H4(g) + H2O(l) -> C2H5OH(l) using the enthalpy changes of the…
A: We are having an aim equation that is equation we have to achieve to get ∆Hr For the reaction. We…
Q: H,SO, (A) но 180° K,Cr,0, H,SO, (B) он (F) (E) (D) (C) NaH (G) + ethyl chloride -OH (H) (1) + )…
A: here we are required to predict the product of the reaction when different reagents are used
Q: What reaction conditions are best to carry out the following reaction? ? H. H. c )03 2) (CH3)2S A…
A: The question is based on the concept of organic reactions. we have to Correctly identify the reagent…
What
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 6 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?According to this video, https://www.youtube.com/watch?v=9Ng6Zv9oLzk, draw the dienophile that was used in the example explaining s-cis and explain what is different about this dienophile from common examples?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Cr2(CH3COO)4(H2O)2 was prepared using the following reaction: 2Cr3+ + Zn(s) --> 2Cr2+ + Zn2+ 2Cr2+ + 4CH3COO- + 2H2O --> Cr2(CH3COO)4(H2O)2 (s) Discuss this reaction and how the reagents react with one another to form the product.1. The of alka zeltzer in iced water will give a result and observation of _________. alka zetltzer dissolved readily alka zeltzer partially dissolved No reaction 2. In the Le Chatelier's Principle, Co(H2O)6 with oxidation of +2 is _________ in color. blue violet pinkHow to determine the type of reaction? (SN1/SN2/elimination)?
- Use the following to complete the table of properties of Friedel –Crafts catalysts (none, poor, low, high, good) Friedel-Crafts catalyst recyclability Waste produced Environmental compatability Shape selectivity AlCl3 HF zeoliteI’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Kindly completes parts A-D:
- plzz answer with detail explanation ina clean white psper step by step in detailThe transition (to the right) is brought about by heating the starting material with a) Mg and anh. Ether b) KOH in alcohol c) H2O d) H3PO4Which of the following reaction coordinate diagrams represents SN1 and E1 reactions? A B C D



