Q: Use curved arrow notation to show how the first resonance structure can be converted to the second.
A:
Q: mistry For each example, specify whether the two structures are resonance contributors to the same…
A: Resonance is the property of organic compounds to exhibit more than one structures for particular…
Q: Hydrocarbon A possesses a significant dipole, even though it is composed of only C-C and C-H bonds.…
A: The given compound contains two cyclic rings. The seven-membered ring contains seven pi electrons…
Q: H. HC= :NH HC : CH2
A:
Q: Follow the curved arrows to draw a second resonance structure for each species. a. H-C=0: b. CH-…
A: (a) The resonance structure for given aldehyde is drawn in step two.
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A: It is given a molecule of N-methylethanimine and its resonating structure needs to be drawn.…
Q: In Naproxen, is the lone pair of the oxygen atom delocalize? I’m other words, can the oxygen…
A: In Naproxen, is the lone pair of the oxygen atom delocalize? In other words, can the oxygen…
Q: Methyl isocyanate, shown as resonance structure 1, can also be represented by other resonance…
A: Resonating structures refers to the different structures which are obtained by movement of pi…
Q: Answer the following: How many H? Most polar bond? Least polar bond?
A: We can redraw the structure of our given molecules as shown in step 2 and count the number of…
Q: Draw a curved arrow starting from the t bond of Draw all atoms of the resulting resonance structure…
A: The set of Lewis structures which explains the delocalization of electrons in a molecule is known as…
Q: What is the resonance hybrid of the anion in the box below N - A В
A:
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A:
Q: Using appropriate curved arrows, draw one additional resonance structure for each of the following…
A:
Q: Give a reasonable set of resonance structures for each compound below. Use Resonance Arrows, and…
A: Resonance is the partial delocalisation of electron pairs which can be shown by arrows.
Q: 2.Use curved arrows to draw all the significant resonance structures for the following molecule and…
A: Interpretation - To draw all the significant resonance structure for the following molecule which…
Q: 6. Draw all significant resonance structures for the following compound, including curved arrows to…
A: Resonance structures of any compound are those structures which shows different possible Lewis…
Q: Add curved arrows to both structures to show the delocalization of electron pairs needed to form the…
A: The given molecule exhibits a continuous delocalisation of pi electrons (resonance).The resonance…
Q: :0: H H. H. H H A B +Z-
A:
Q: a. Draw as many possible resonance structures of the molecule, (show the direction of electron…
A:
Q: 7. a) Draw the resonance arrows to form the structure on the right, b) Circle the major resonance…
A: The transformation given is,
Q: Draw as many resonance structures as you can for each of the following structure, and use…
A: Resonance structures arise when there exists a conjugated double bond/vacant orbital relative to an…
Q: Draw two significant resonance structures for the molecule below (start at the same site of…
A: Here we have to draw the resonance structure and justify which is most stable and which is least…
Q: Additional Questions: 11 Use curved arrows to draw any additional resonance structu Fredict which…
A: Resonance: Resonance is a method of describing the delocalization of electrons in some molecules in…
Q: Based on trends discussed in class, which of the following molecules would you predict to include…
A: Alkanes are organic compounds that consist entirely of single-bonded carbon and hydrogen atoms and…
Q: What happens to the bond angle (column 4) as the # of lone pairs (column 2) increases? Based on…
A: We know that order of repulsions is l.p-l.p>l.p-b.p>b.p-b.p
Q: are these aromatic, antiaromatic, or nonaromatic? Draw in all lonepairs and indicate the orbital…
A: for molecule have aromatic, the compound must be cyclic and planner and follow the huckel rule the…
Q: Add the missing curved arrow notation, lone pair electrons, and formal charges to all atoms. 20 C +…
A: The given reaction mechanism has been completed below.
Q: Name the following unbranched molecules from the numbered cards. When using a locator number use a…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Draw a more stable resonance structure for the following molecule. Use a curved arrow to show how to…
A: The phenomena by which the compound is represented by structures, non of which can exactly explain…
Q: Compare columns I and II, and write A if the value in column I is greater than in II B if the value…
A: The compounds given are Column I: 1H-phenalene Column II: 1,4-dihydrophenanthrene
Q: ne resonance hybrid a
A: Rules to identify the best resonance contributors are as follows: Greatest number of full octets…
Q: PLEASE Answer all subquestions 1) Draw resonance structures AND resonance hybrid for following…
A:
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A:
Q: O Draw the curved arrows on the left resonance structure to account for formation of the right…
A: In the given structure the resonance structure is given. The curved arrow in the left structure has…
Q: For each of the following pairs of resonance structures, circle the one that would make greater…
A: IN PART A 1st structure contributes to the actual resonance hybrid because the + ve sign is on less…
Q: For each structure below (1) show all resonance forms (do not increase overall # of formal charges),…
A: More the Resonance structure more will be the stability.
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A:
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonanre…
A:
Q: 2. Draw 2 resonance structures for the molecule below including arrows!
A:
Q: including arrows For the following structures, complete resonance structures electron flow and…
A: The ion given is,
Q: Draw the most important resonance form for SO₂ (bonded O-S-O). Draw the molecule on the canvas by…
A: In the given question we have to draw the resonance structure of SO2 the resonance structure will…
Q: Use the "+" and "" tools to add any missing formal charges in the compound below. Please wait for…
A: Lewis structures depict the structure of particular molecule in the simplest way. It exhibits the…
Q: Follow the curved arrows to draw a second resonance structure for each species.
A: a) Please find below the second resonance structure for given species.
Q: e) Circle the most basic nitrogen atom in the compound below. NH2
A: Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: From what we know today, what do the two Kekulé structures for benzene really represent? August…
A: Friedrich August Kekule, in 1865, suggested that the benzene contain a ring of six carbon atom in…
Q: For each structure below (1) show all resonance forms (do not increase overall # of formal charges),…
A:
Q: How many hydrogens are present in the following molecules? a) b)
A: The number of hydrogen molecules in the given structure has to be given,
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A: Given structure:
Q: A. Draw all significant resonance structures of each compound below, using appropriate arrows, and…
A: Given
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A: The solution is given below -
Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE.
Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.
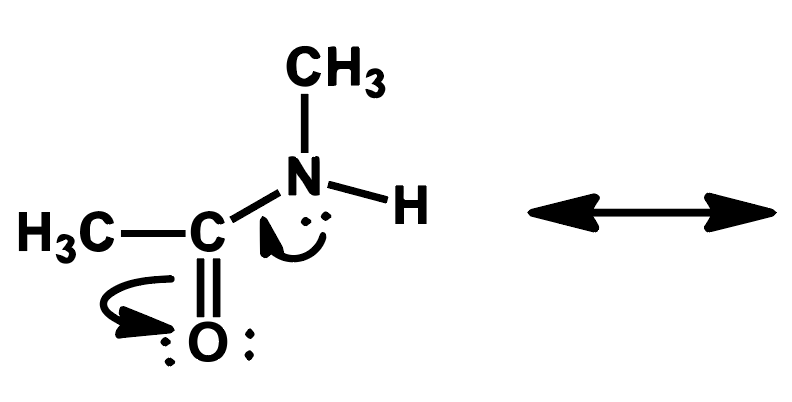
Step by step
Solved in 2 steps with 1 images


