Chapter21: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
Section21.SE: Something Extra
Problem 30VC
Related questions
Question
Transcribed Image Text:Reset
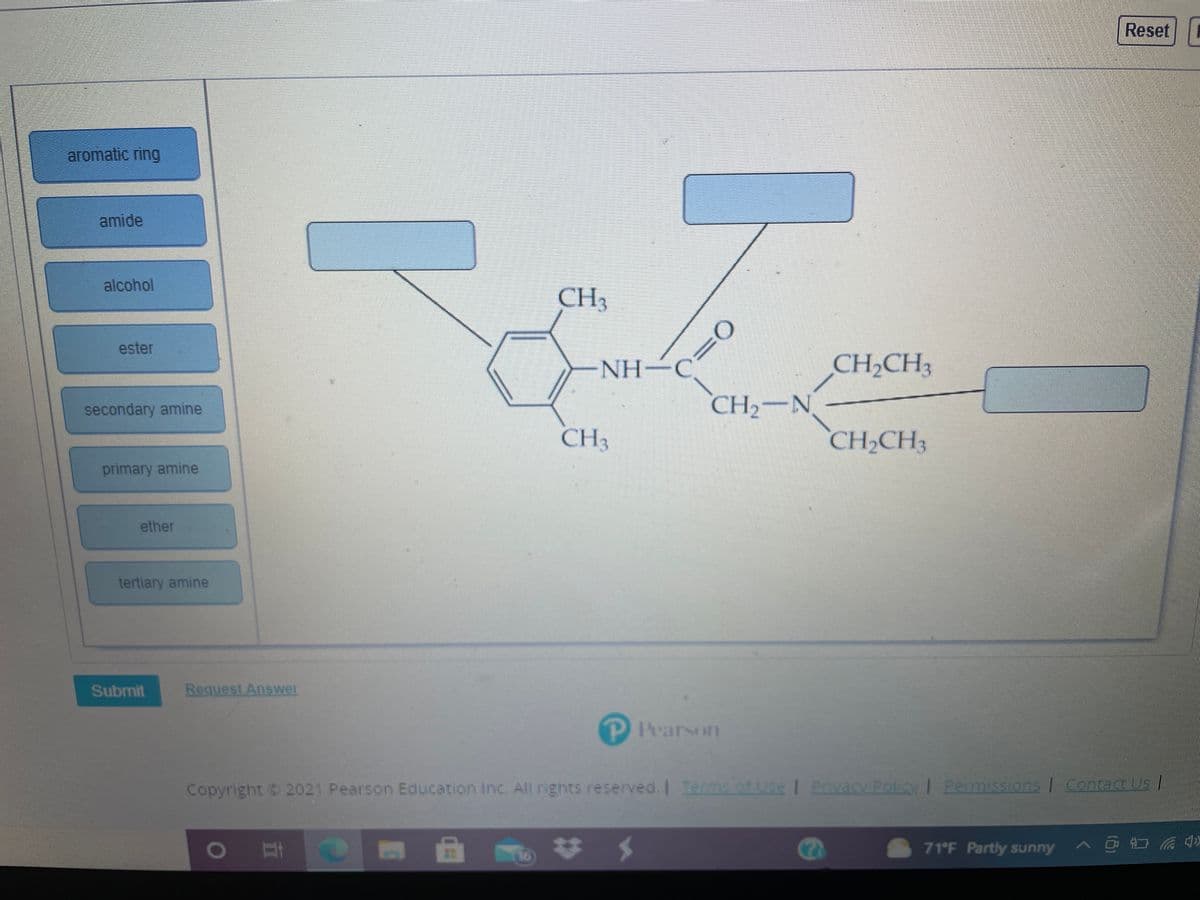
aromatic ring
amide
alcohol
CH3
ester
NH-C
CH2CH3
secondary amine
CH2-N
CH3
CH2CH3
primary amine
ether
tertiary amine
Submit
Request Answer
P Pearson
Copyright 2021 Pearson Education Inc. Anghts reserved. I Terma ofUse I Povacy Poloy I Pernmissions Contact s/
# メ
71°F Partly sunny @ >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning