Q: Organic chemistry subject, please provide the correct solution for the following. Naming;
A: The following steps should be followed during the IUPAC nomenclature of the given organic compound: ...
Q: Using the electronic configuration of the transition metals, briefly explain why salts of Cu(II) are...
A: when any iron have an electron then it will colourful because when electromagnetic wave radiation in...
Q: te a balanced equation for the decomposition of ammonium nitrate to form molecular ogen, molecular o...
A: balancing of a chemical reaction means there should be number of atoms be equal in both side reactan...
Q: what volume of sulfur dioxide at 25c and 1 atm pressure is produced when one boxcarload (4 x 10 ^3 c...
A: Chalcopyrite ore is roasted at a temperature of 25°C and 1atm pressure and a volume of 4×103 ft3 is ...
Q: There are 2 balloons in a room. They are identical in size and material. One balloon is filled with ...
A: Air is a mixture of various gases. The parameters of gases like volume, temperature, pressure are re...
Q: 17) Organic chemistry subject, please provide the correct solution for the following.
A: Under acidic condition epoxide opening occur from the greater substituted carbon. This the nucleophi...
Q: Calculate the pH when 54.0 mL of 0.150 M KOH is mixed with 20.0 mL of 0.300 M HBrO (Ka = 2.5 × 10⁻⁹)
A: Calculate mole of each substance then applied neutralissation reaction. limiting reagent is HBRO and...
Q: What is the mechanism of the preparation of cinnamic acid?
A: First pyridine is a base and it will release H+ from highly acidic Hydrogen .carbocation will attac...
Q: If I had 100g of each- water, copper, gold, and silver- and I applied 100J of energy to each startin...
A: The substances considered are water, copper, gold and silver.
Q: A typical atmospheric pressure in Leadville, Colorado (elevation 10,200 feet) is 68 kPa. Use the gra...
A: Boiling Point - The point at which, the external atmospheric pressure is equaled by the increasing v...
Q: 5) Organic chemistry subject, please provide the correct solution for the following. Naming;
A: In order to do the naming of this compound, it is to be noted that this is an ether functional group...
Q: COmplete the equations for these reactions by giving the structures of the products. Assume that the...
A: First NH2 will attack on nucleophilic site which is present in anhydride and then O- will attack Int...
Q: Name the following compounds using correct ionic or covalent naming methods C4S3
A: An ionic bond can be defined as the electrostatic force of attraction between cation and anions. Her...
Q: 210 Calculate the dose equivalent rate 1.6 m from an unshielded 3.0 x 10" Bq ²1º Po – Be source whic...
A:
Q: The following chemical reaction takes place in aqueous solution: FeSO,(aq)+2 NAOH(aq) →Fe(OH),(s)+Na...
A: Given The given ionic compounds are NaOH and FeSO4.
Q: Ice naturally floats on water due to its lower density. If ice did not float on top of water, descri...
A: if ice did not float life underwater would be impossible it stays on the top and ice slowly gets thi...
Q: A cylinder measuring 2.0 cm wide and 2.4 cm high is filled with gas and the piston pushed down with ...
A: pressure = (force ÷ Area ) Force = pressure × Area
Q: The pressure of a gas in a 5.5 L container is 6.789 atm. What is the pressure if thevolume is increa...
A: The relationship between pressure and volume for a gas at constant temperature is given by Boyle's l...
Q: answer this question please
A: For the first-order kinetics: ln[A]0[A]t= kt Where, [A]0=...
Q: A sample of neon gas at a pressure of 0.830 atm and a temperature of 22.6 °C, occupies a volume of 7...
A:
Q: How would the magnitude of ∆ change if the H2O ligands in [Ti(H2O)6]3+ were replaced with NH3 ligand...
A: Given: The magnitude of ∆ change if the H2O ligands in [Ti(H2O)6]3+ were replaced with NH3 ligands
Q: The syrup is diluted three consecutive times by a factor of 2. If the concentration of red dye is th...
A: The dilution factor is calculated by dividing the final volume of the solution by the initial volume...
Q: Heating 3.0 mol of a coordination compound gives 1.5 mol of ammonia, 3.0 mol of water 1.5 mol of HCl...
A: The compounds in which the metal atom is surrounded by the ligands is known as coordination compound...
Q: 1. Write a balanced chemical equation, including physical state symbols, for the combustion of liqui...
A:
Q: 20) Organic chemistry subject, please provide the correct solution for the following.
A: Here, elimination followed by hydration of alkene occurs following Markovnikov rule.
Q: The% K present in the compound K4Fe(CN)6
A: The formula of percent composition is as follows: Mass % element=Mass of the element in the compound...
Q: Provide the balanced net ionic chemical equation between IO3- and I-
A: The net Ionic reaction for the given chemical equation can be given as: (1) The balanced reaction of...
Q: Ammonium carbamate (NH₂COONH₄) is a salt of carbamicacid that is found in the blood and urine of mam...
A: Given:
Q: Using the Bohr model, determine the energy, in joules, necessary to ionize a ground-state hydrogen a...
A: A hydrogen atom has one electron. This electron is present in 1s orbital.
Q: Free radicals are highly reactive molecules that often contain ________. They are reactive due to th...
A: A reaction intermediate can be defined as the molecular entity which is formed by the reactant speci...
Q: #1. Thank you.
A: The boiling point of a liquid describes the temperature point in which the vapor pressure belongs to...
Q: Convert benzene into attached compound. You may also use any inorganicreagents and organic alcohols ...
A:
Q: Write the ground-state electron configuration for each atomand ion pair-Zr, Zr2+, Co, Co2+, Tc, Tc3+...
A: Follow the steps below to write the electronic configuration of any element: 1. Identify the element...
Q: Dinitrogen tetraoxide is a liquid boiling at 21°C (1atm). The standard vaporizing enthalpy is +28,8 ...
A: The reaction of hydrazine and dinitrogen tetraoxide can be represented by the balanced chemical equa...
Q: what volume of sulfur dioxide at 25c and 1 atm pressure is produced when one boxcarload (4 x 10 ^3 c...
A:
Q: Many computer chips are manufactured from silicon, which occurs in nature as SiO2. When SiO2 is heat...
A: Silicon is produced industrially by electrochemical reduction of silica or SiO2 with coke which is s...
Q: Which of the following is true when you pour a mixture of liquids to be separated into a distilling ...
A: Distillation is a process by which mixture of liquids having different boiling points. The liquid ha...
Q: Carl is approaching the Canadian border on a trip from Vermont to Montreal, Canada, and the gas in h...
A: The amount of gas in liter that Carl can buy from $20 US dollar is; Vgas=1 gal$2.45×$200.264 gal/L=3...
Q: Calculate the wavelength (in m) of a photon that has a frequency of 4.72 x 1013s1. Select one: O 6.3...
A: The required equation is c=λν where c is the speed, λ is the wavelength and ν is the frequency. ...
Q: Some bismuth tablets, a medication used to treat upset stomachs, contain 262 mg of bismuth subsalicy...
A: Stoichiometry involves the calculation of concentration of solutions in the given conditions of volu...
Q: An atomic isotope radioactively decays and has a half-life of 23.7 days. How many days will it take ...
A: In order to solve this question, we will follow the rule that all radioactive disintegration decays ...
Q: When 0.514 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises ...
A: When a chemical reaction occurs, some energy change takes place. In the case of combustion, energy i...
Q: Illustrate cis-1,3-Dibromocyclopentane contains a plane of symmetry ?
A: Given compound: cis-1,3-Dibromocyclopentane
Q: 1. Aluminum sulfate can be made by the following reaction: 2AlCl3(aq)+3 H2SO4(aq)➡Al(SO4)3(aq)+6HCl(...
A: As you not specified so I am giving answer of first question First convert mass into mole by diving ...
Q: Show how Novocain, a painkiller used frequently by dentists, can be prepared from benzene and compou...
A: The reaction scheme for the generation of the Novocain is shown below. Benzene undergoes Friedel-Cra...
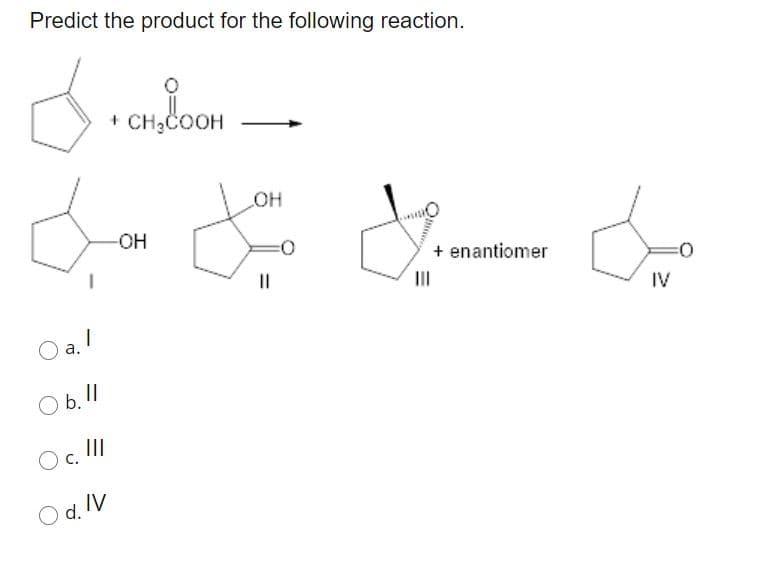
25)
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Predict the Product of the reaction and indicate regiochemistry and stereochemistry when relevant. PLEASE EXPLAINE! Thank you!In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:w how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.
- Predict the product of the following reaction and classify the reaction Pb+FeSO4---> PbSO4+ ______cmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)Predict major and minor products of bicyclo[4.4.0]dec-4-ene when reacted with NBS in peroxide.
- When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.There are two isomeric cyclohexa-1,4-diene products when benzoic acid undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads tothe formation of the major product. (b) Will the Birchreduction of benzoic acid occur faster or slower than theBirch reduction of benzene itself? Hint: Is –CO2H anelectron-donating or an electron-withdrawing group?(a) What happens when CH3—O—CH<sub3 is heated with HI?(b) Explain mechanism for hydration of acid catalyzed ethene :CH2 = CH2 + HzO CH3—CH,—OH
- The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?OChem reaction scheme help... Please provide the structures for the products obtained in the reaction schemes shown in the attaced imagepredict the stating materials, reagents, or products of the followinf reactions



