Chapter 12 Kine 674 () [A]o = kt %3D In [A], The initial concentration of C4H3, [A]o, is not provided, but the provision that 80.0% of the sample h decomposed is enough information to solve this problem. Let x be the initial concentration. in which e the concentration after 80.0% decomposition is 20.0% of x or 0.200x. Rearranging the rate law to isolata . and substituting the provided quantities yields: [x] [0.200x] t = ln %3D = In 5 x 1 %3D 9.2x 10-3 s-1 = 1.609 x 1 %D 9.2 x 10-3 s-1 = 1.7x 102 s %3D Check Your Learning Iodine-131 is a radioactive isotope that is used to dia Codine-131 decays to xenon-131
Chapter 12 Kine 674 () [A]o = kt %3D In [A], The initial concentration of C4H3, [A]o, is not provided, but the provision that 80.0% of the sample h decomposed is enough information to solve this problem. Let x be the initial concentration. in which e the concentration after 80.0% decomposition is 20.0% of x or 0.200x. Rearranging the rate law to isolata . and substituting the provided quantities yields: [x] [0.200x] t = ln %3D = In 5 x 1 %3D 9.2x 10-3 s-1 = 1.609 x 1 %D 9.2 x 10-3 s-1 = 1.7x 102 s %3D Check Your Learning Iodine-131 is a radioactive isotope that is used to dia Codine-131 decays to xenon-131
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter13: Mass Spectrometry
Section: Chapter Questions
Problem 2Q
Related questions
Question
I am not sure how they calculated the ln 5. I would like to possibly see a more detailed step for this solution.
![Chapter 12 Kine
674
()
[A]o
= kt
%3D
In
[A],
The initial concentration of C4H3, [A]o, is not provided, but the provision that 80.0% of the sample h
decomposed is enough information to solve this problem. Let x be the initial concentration. in which e
the concentration after 80.0% decomposition is 20.0% of x or 0.200x. Rearranging the rate law to isolata .
and substituting the provided quantities yields:
[x]
[0.200x]
t = ln
%3D
= In 5 x
1
%3D
9.2x 10-3 s-1
= 1.609 x
1
%D
9.2 x 10-3 s-1
= 1.7x 102 s
%3D
Check Your Learning
Iodine-131 is a radioactive isotope that is used to dia
Codine-131 decays to xenon-131](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F9dc111da-6604-4898-805c-db1b932cb297%2Fdd765229-9318-4167-89f9-4937a824665e%2F6i61d56.jpeg&w=3840&q=75)
Transcribed Image Text:Chapter 12 Kine
674
()
[A]o
= kt
%3D
In
[A],
The initial concentration of C4H3, [A]o, is not provided, but the provision that 80.0% of the sample h
decomposed is enough information to solve this problem. Let x be the initial concentration. in which e
the concentration after 80.0% decomposition is 20.0% of x or 0.200x. Rearranging the rate law to isolata .
and substituting the provided quantities yields:
[x]
[0.200x]
t = ln
%3D
= In 5 x
1
%3D
9.2x 10-3 s-1
= 1.609 x
1
%D
9.2 x 10-3 s-1
= 1.7x 102 s
%3D
Check Your Learning
Iodine-131 is a radioactive isotope that is used to dia
Codine-131 decays to xenon-131
Expert Solution
Step 1
The initial concentration, which 100% of C4H8 is not known. Let it be x.
About 80% of C4H8 is decomposed.
The final concentration of C4H8 at time t is 100% of x- 80% of x = 20% of x
That is 20/100(x) = 0.2x present at time t.
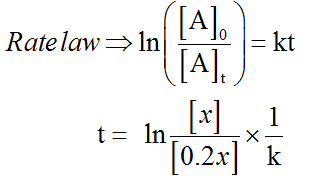
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole
