Chart 1 I 8 - 6 H 4 Axis Title G fx 1 O Chart Title 2 F O 0 80 70 60 50 40 30 20 10 0 Axis Title D 1 C с y 8.5564x+4.7336 R=0.9924 <--Series1 Linear (Series1) O O Y B 25 37 49 55 75 X A 1 2.5 2 3.7 3 4.9 4 6.1 5 8.2 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Determine the value of the linear correlation coefficient, R2 for the following data: X 2.5 3.7 4.9 6.1 8.2 Y 25 37 49 55 75 a. 0.9879 b. 0.9898 C. 0.9979 d. 0.9924 e. 0.9899
Chart 1 I 8 - 6 H 4 Axis Title G fx 1 O Chart Title 2 F O 0 80 70 60 50 40 30 20 10 0 Axis Title D 1 C с y 8.5564x+4.7336 R=0.9924 <--Series1 Linear (Series1) O O Y B 25 37 49 55 75 X A 1 2.5 2 3.7 3 4.9 4 6.1 5 8.2 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Determine the value of the linear correlation coefficient, R2 for the following data: X 2.5 3.7 4.9 6.1 8.2 Y 25 37 49 55 75 a. 0.9879 b. 0.9898 C. 0.9979 d. 0.9924 e. 0.9899
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter3: Molecules, Moles, And Chemical Equations
Section: Chapter Questions
Problem 3.108PAE: 3.108 As chip speeds increase, the width of the interconnects described in Problem 3.107 must be...
Related questions
Question
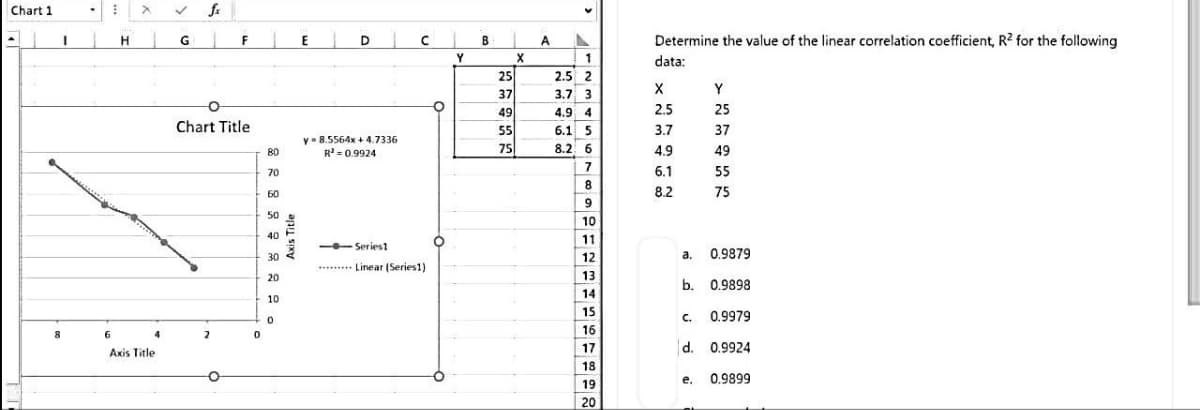
Transcribed Image Text:Chart 1
8
WASSERSY
6
H
X
Axis Title
4
✓ fx
G
F
Chart Title
2
0
80
70
60
50
40
30
20
10
0
Axis Title
y 8.5564x +4.7336
R²=0.9924
--Series1
*****
Linear (Series1)
O
Y
B
25
37
49
55
75
X
A
▲
1
2.5 2
3.7 3
4.9 4
6.1 5
8.2 6
7
8
9
10
11
FOSS7322
12
13
14
15
16
17
18
19
20
Determine the value of the linear correlation coefficient, R² for the following
data:
X
2.5
3.7
4.9
6.1
8.2
Y
25
37
49
55
75
a. 0.9879
b. 0.9898
0.9979
d. 0.9924
C.
e. 0.9899
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning