LL 0 Chem101 B Chem101- Survey of Chemistry X SWAN - Clayton State University X A app.101edu.co Bing Vex 3 Game Apps clayton.k12.ga.us bookmarks Question 34 of 35 A patient is prescribed to take 2 pills/day of a drug for a month. The drug is available in 10.0 mg/pill How many grams per week of the drug will the patient receive? STARTING AMOUNT ADD FACTOR ANSWER 00L 140 2 0.01 30.0 0.02 10.0 0.00286 0.140 1. 2.86 0.1 2 Type here to search g/week week days 7. pills/day days/week bu 35°F Clou dy 91 %24 8J 3. 4. 00 6
LL 0 Chem101 B Chem101- Survey of Chemistry X SWAN - Clayton State University X A app.101edu.co Bing Vex 3 Game Apps clayton.k12.ga.us bookmarks Question 34 of 35 A patient is prescribed to take 2 pills/day of a drug for a month. The drug is available in 10.0 mg/pill How many grams per week of the drug will the patient receive? STARTING AMOUNT ADD FACTOR ANSWER 00L 140 2 0.01 30.0 0.02 10.0 0.00286 0.140 1. 2.86 0.1 2 Type here to search g/week week days 7. pills/day days/week bu 35°F Clou dy 91 %24 8J 3. 4. 00 6
Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.11PAE: Some farmers use ammonia, NHS, as a fertilizer. This ammonia is stored in liquid form. Use the...
Related questions
Question
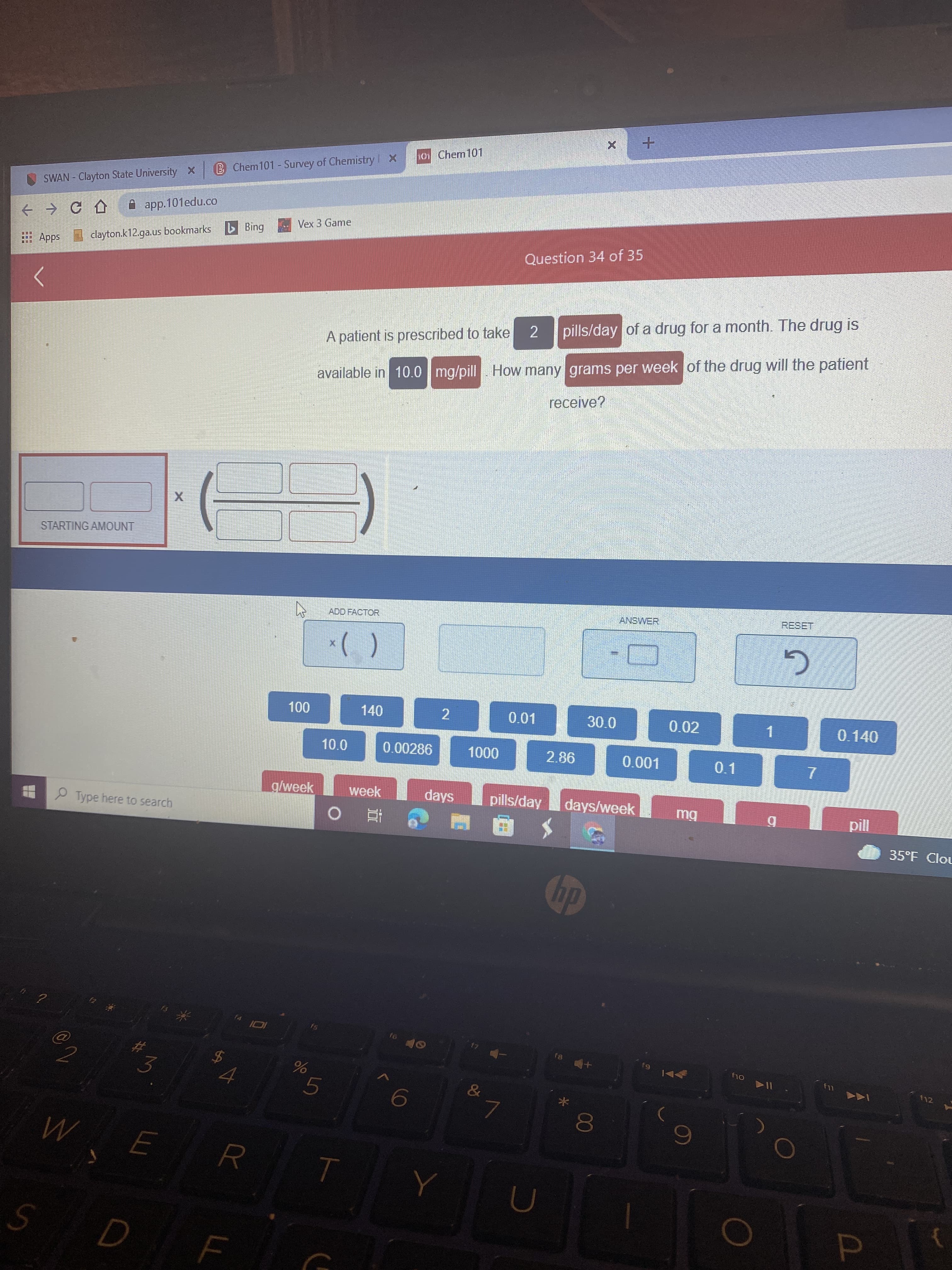
Transcribed Image Text:LL
0 Chem101
B Chem101- Survey of Chemistry X
SWAN - Clayton State University X
A app.101edu.co
Bing
Vex 3 Game
Apps clayton.k12.ga.us bookmarks
Question 34 of 35
A patient is prescribed to take
2
pills/day of a drug for a month. The drug is
available in 10.0 mg/pill How many grams per week of the drug will the patient
receive?
STARTING AMOUNT
ADD FACTOR
ANSWER
00L
140
2
0.01
30.0
0.02
10.0
0.00286
0.140
1.
2.86
0.1
2 Type here to search
g/week
week
days
7.
pills/day
days/week
bu
35°F Clou
dy
91
%24
8J
3.
4.
00
6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning