Calculation Tables Table 1 Finding the number of moles of sodium acetate (NAC2H3O2) in solution A Molarity of sodium acetate on bottle 0.200 M Volume of sodium acetate solution used in 5.00 mL dilution (mL) Volume of sodium acetate solution used in 0.005 L dilution (L) Moles of sodium acetate used in dilution (mol) Table 2 Finding the molarity of acetic acid (HC2H3O2) in solution B Molarity of acetic acid solution on bottle 1.00 M Volume of acetic acid solution used in dilution 25.0 mL Volume of acetic acid solution used in dilution (L) 0.025L Moles of acetic acid solution (mol) Total volume of solution in dilution (mL) (m) 100.0 mL Total volume of solution in dilution (L) Molarity of acetic acid in diluted solution (M) Table 3: Moles of Acetic Acid: Molarity of Acetic Acid Solution (M) Volume of Acetic Acid Volume of Acetic Acid Moles of Acetic Acid Solution Added (mL) Solution Added (L) Added (mol) D.025M 0.025M 0.002 L 0.004 L. 025M D.025M Table 4 Volume of Moles of Moles of moles of sodium acetate (mol NaAc 807 mol HAc eyd of Acetic Hd Acetic Sodium moles acetic acid Acid Acid Acetate Solution Added 4.74 4.00 4.74 6.00 4.74 4.74 Data Sheet Acidic Wavelength (nm) Basic 0.117 380 0.097 0.157 0.114 420 0.088 0.0500 0.238 0.0400 0.227 0.042 0.177 480 0.067 0.115 520 0.100 690' 540 0.157 0.036 0.229 0.012 580 0.325 0.005 0.44 0.003 009 605 0.467 900'0 610 0.484 800'0 615 0.494 0.005 620 0.493 0.005 625 0.478 630 0.444 0.005 635 0.397 0.004 640 0.342 0.001 099 680 0.135 0.001 0.042 0.004 Volume of Acetic Acid Solution Added (mL) Absorbance (Ax) 0.312 0.227 0.176 0.150
Calculation Tables Table 1 Finding the number of moles of sodium acetate (NAC2H3O2) in solution A Molarity of sodium acetate on bottle 0.200 M Volume of sodium acetate solution used in 5.00 mL dilution (mL) Volume of sodium acetate solution used in 0.005 L dilution (L) Moles of sodium acetate used in dilution (mol) Table 2 Finding the molarity of acetic acid (HC2H3O2) in solution B Molarity of acetic acid solution on bottle 1.00 M Volume of acetic acid solution used in dilution 25.0 mL Volume of acetic acid solution used in dilution (L) 0.025L Moles of acetic acid solution (mol) Total volume of solution in dilution (mL) (m) 100.0 mL Total volume of solution in dilution (L) Molarity of acetic acid in diluted solution (M) Table 3: Moles of Acetic Acid: Molarity of Acetic Acid Solution (M) Volume of Acetic Acid Volume of Acetic Acid Moles of Acetic Acid Solution Added (mL) Solution Added (L) Added (mol) D.025M 0.025M 0.002 L 0.004 L. 025M D.025M Table 4 Volume of Moles of Moles of moles of sodium acetate (mol NaAc 807 mol HAc eyd of Acetic Hd Acetic Sodium moles acetic acid Acid Acid Acetate Solution Added 4.74 4.00 4.74 6.00 4.74 4.74 Data Sheet Acidic Wavelength (nm) Basic 0.117 380 0.097 0.157 0.114 420 0.088 0.0500 0.238 0.0400 0.227 0.042 0.177 480 0.067 0.115 520 0.100 690' 540 0.157 0.036 0.229 0.012 580 0.325 0.005 0.44 0.003 009 605 0.467 900'0 610 0.484 800'0 615 0.494 0.005 620 0.493 0.005 625 0.478 630 0.444 0.005 635 0.397 0.004 640 0.342 0.001 099 680 0.135 0.001 0.042 0.004 Volume of Acetic Acid Solution Added (mL) Absorbance (Ax) 0.312 0.227 0.176 0.150
Chapter29: Mass Spectrometry
Section: Chapter Questions
Problem 29.5QAP
Related questions
Question
Need guidance in filling out this form using numbers given.
please show steps to solution. Thanks
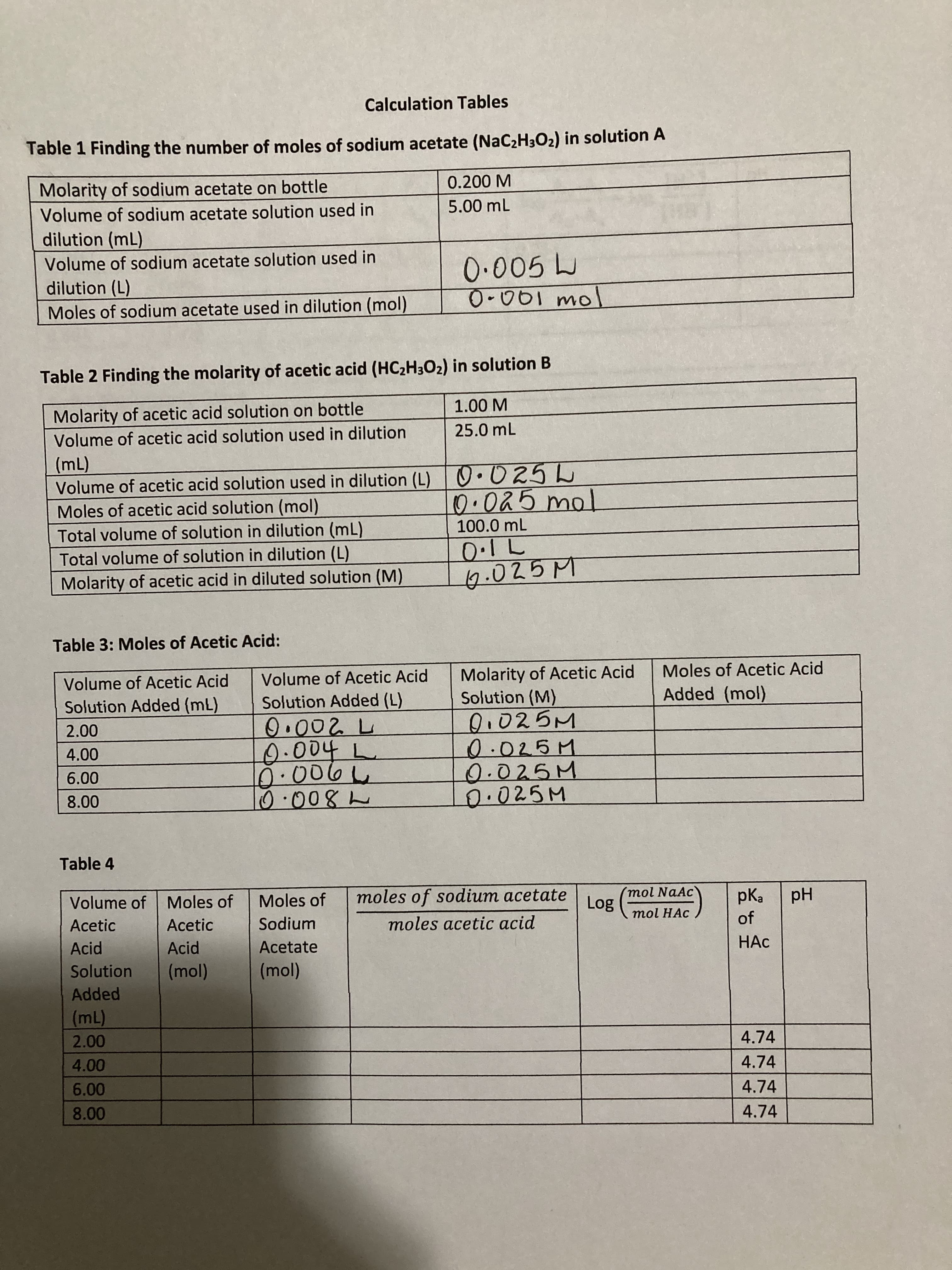
Transcribed Image Text:Calculation Tables
Table 1 Finding the number of moles of sodium acetate (NAC2H3O2) in solution A
Molarity of sodium acetate on bottle
0.200 M
Volume of sodium acetate solution used in
5.00 mL
dilution (mL)
Volume of sodium acetate solution used in
0.005 L
dilution (L)
Moles of sodium acetate used in dilution (mol)
Table 2 Finding the molarity of acetic acid (HC2H3O2) in solution B
Molarity of acetic acid solution on bottle
1.00 M
Volume of acetic acid solution used in dilution
25.0 mL
Volume of acetic acid solution used in dilution (L) 0.025L
Moles of acetic acid solution (mol)
Total volume of solution in dilution (mL)
(m)
100.0 mL
Total volume of solution in dilution (L)
Molarity of acetic acid in diluted solution (M)
Table 3: Moles of Acetic Acid:
Molarity of Acetic Acid
Solution (M)
Volume of Acetic Acid
Volume of Acetic Acid
Moles of Acetic Acid
Solution Added (mL)
Solution Added (L)
Added (mol)
D.025M
0.025M
0.002 L
0.004
L.
025M
D.025M
Table 4
Volume of Moles of
Moles of
moles of sodium acetate
(mol NaAc
807
mol HAc
eyd
of
Acetic
Hd
Acetic
Sodium
moles acetic acid
Acid
Acid
Acetate
Solution
Added
4.74
4.00
4.74
6.00
4.74
4.74
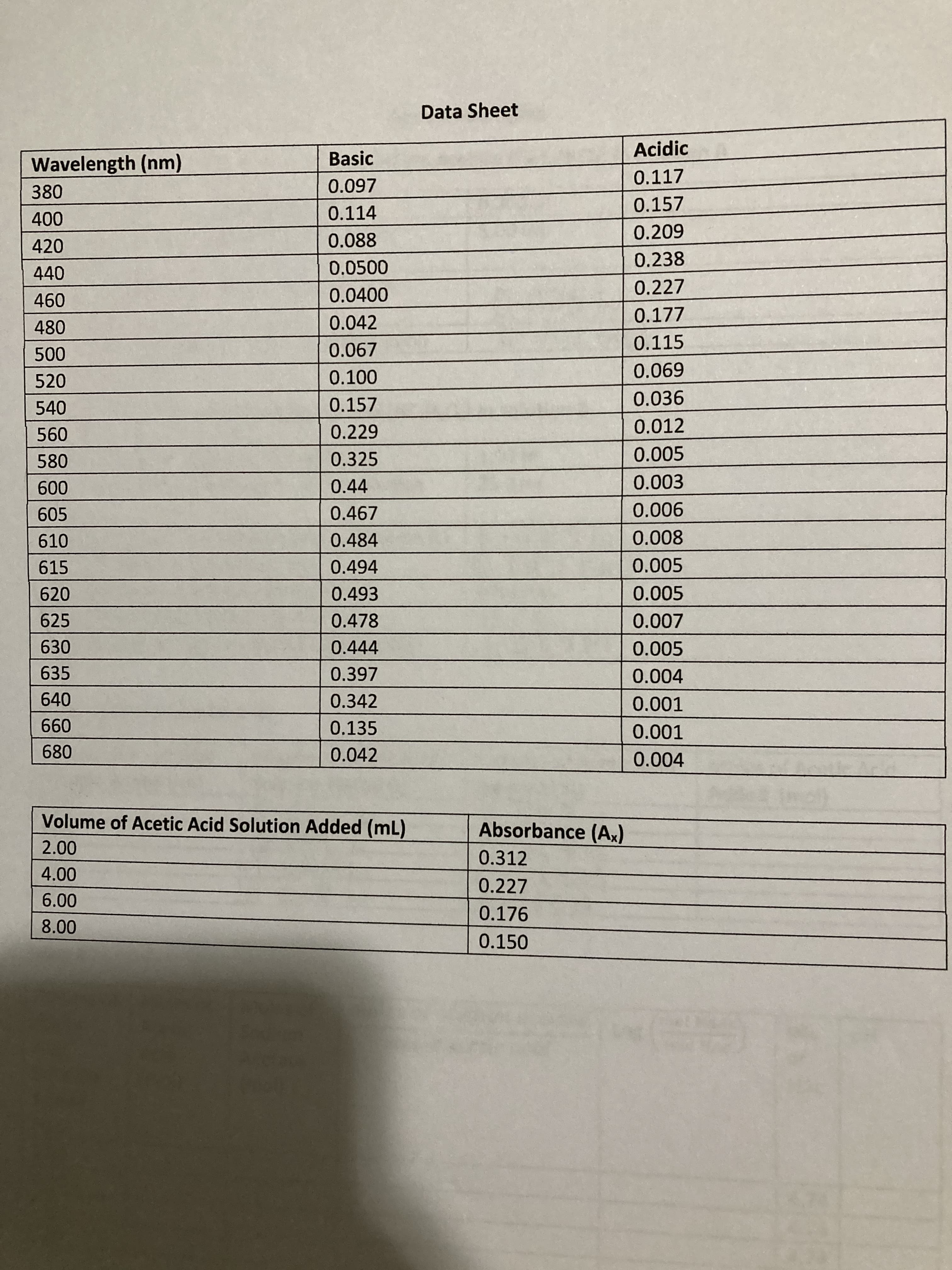
Transcribed Image Text:Data Sheet
Acidic
Wavelength (nm)
Basic
0.117
380
0.097
0.157
0.114
420
0.088
0.0500
0.238
0.0400
0.227
0.042
0.177
480
0.067
0.115
520
0.100
690'
540
0.157
0.036
0.229
0.012
580
0.325
0.005
0.44
0.003
009
605
0.467
900'0
610
0.484
800'0
615
0.494
0.005
620
0.493
0.005
625
0.478
630
0.444
0.005
635
0.397
0.004
640
0.342
0.001
099
680
0.135
0.001
0.042
0.004
Volume of Acetic Acid Solution Added (mL)
Absorbance (Ax)
0.312
0.227
0.176
0.150
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning