Q: Consider the following two Lewis resonance structures for HCNO. What is the formal charge on C in st...
A: Formal charge of an atom is calculated by the formula= (Number of valence electrons) - {electrons in...
Q: Imagine that we have excess NaCl solid on the bottom of a saturated aqueous solution of NaCl. Please...
A: A saturated solution is a solution in which no more solute can be dissolved in it at a particular te...
Q: b) 1- Study the following diagram and then answer the following: 1- Write the primitive formula of ...
A: Interpretation - To study the following digram and we have to give answer of the following question...
Q: Which substance will conduct electricity? o Pure water (H2O) o Epsom salt dissolved in water (MgSO4)...
A: Electrolytes: It is a substance that ionizes or dissociates completely into ions when dissolve in aq...
Q: Use BaSO4 to distinguish between solubility, molar solubility, and solubility product.
A: BaSO4 is the inorganic ionic compound, when it is dissolve in aqueous solution, it dissociates into ...
Q: Calculate the number of meq. of H2C2O4.2H20 (fw= 126.1) in 0.5 gm of the pure compound assuming that...
A: The convenient method of expressing the amount of a substance is the mole concept. Measurement can b...
Q: An ideal gas is placed in a piston at 0°C under 1 atm pressure. How does the density of this gas cha...
A:
Q: enumerate the properties of alcohols contributing to their reactivity with an oxidizing reagent?
A:
Q: In the gravimetric determination of barium (FW=137.33 g/mol) as BaCrO4 (FW=253.32 g/mol), what weigh...
A: The ratio of mass of particular component to the mass of sample multiply by 100 is known as percenta...
Q: What is margarine
A: Margarine is made up of vegetable oils that contains unsaturated fats which reduced the bad LDL chol...
Q: In the first-order decomposition of dinitrogen pentoxide at 335 K : N2O5 (g) (yields) 2 NO2 (g) + ½ ...
A: Given: Sample weight in g initially = 2.50 g Sample weight after 109 s = 1.50 g Time, t = 109 s The ...
Q: The reaction below was found to have a Kc = 4.2 at a 448°C. Find the value of K, at that temperature...
A: Kc and Kp are the equilibrium constant at particular concentration and pressure respectively. The re...
Q: What is the Kb of the base PO−34 given that a 0.48M solution of the base has a pH of 12.70? The equa...
A: Given, pH = 12.70 [base] = [PO43-] = 0.48 (M) Kb is the equlibrium constant for the ...
Q: Draw 2- methyl propyl
A: -yl subscript is used for the function groups of --alkyl( Cn H2n+1 ).
Q: Use Ideal gas law equation to solve the problem.1. Calculate the pressure(P) exerted by a 0.25 mole ...
A: The given data: The number of moles of Sulfur hexafluoride (SF6) = 0.25 mole The volume of the SF6 =...
Q: Calculate the concentration of Ni2+ in the solution prepared by mixing a) 50 mL 0.03 M Ni2+ with 50....
A:
Q: Answer for "#1": 1.936 x 10-18 J How would the answer to #1 compare to the amount of energy (in jou...
A: The expression of energy states can be known from Bohr model.
Q: Combustion of hydrocarbons such as methane (CH,) produces carbon dioxide, a "greenhouse gas." Greenh...
A: Given; The combustion of methane.
Q: For the following second order reaction, the rate constant is 0.752 1/M s and the [A]0 = 2.86 M. Ca...
A: Given, Rate constant of the reaction is 0.752 (Msec)-1. Concentration of sample initially, [A]0 = 2...
Q: Which of the following three compounds has the lowest boiling point? (Consider Van der Waals forces ...
A: .The boiling point of alkanes increases with the number of carbon atoms because surface area increas...
Q: Which properties would be changed if cotton fiber had amino group
A: Interpretation - To tell about that which properties would be changed if cotton fiber had amino gro...
Q: Electromagnetic radiation behaves both as particles (called photons) and as waves. Wavelength (A) an...
A: Given information, Frequency (v) = 2.60 GHz Wavelength = ? Speed of light (c) = 3.00 x 108 ms-1
Q: The Ksp of AgBr is 5.0 x 10^-13 . What is the molar solubility of AgBr.
A: Given data is as follows: The solubility product constant (Ksp) = The molar solubilty (S) of AgBr =...
Q: Use Gay-Lussac equation to solve the given problem.At 293 K a confined ammonia gas has a pressure of...
A: Gay-Lussac's law is a gas law that illustrates the relationship of temperature and pressure of a giv...
Q: For the following second order reaction, the rate constant is 0.0462. If [A]0 = 8.27, calculate [A]...
A: Given data is as follows: The equation for second order reaction for calculation of [A] is as follo...
Q: May I please get some help on this, Please include a description of your process of solving this pro...
A:
Q: 6. The rate of formation of D for the reaction 3A(g) + B(g) +2C(g)D(g) + 2E(g) is k[A][B]?. If the c...
A: We are given the following reaction: 3A(g) + B(g) + 2C(g)---------->D(g) + E(g).
Q: A galvanic cell was formed using Avium and Beanium:[hypothetical elements] Reduction Potentials: Av ...
A:
Q: Help me please
A: The distribution of electrons in an element's atomic orbitals is described by its electron configura...
Q: Which of the following solutions can act as a buffer: (a) KCl/HCl, (b) KHSO4/H2SO4, (c) KNO2/HNO2?
A:
Q: B. Solid AgCl (Ksp = 1.8×10-10 ) is dissolved in 1 Liter of water , then 0.585 gm of sodium chloride...
A: AgCl will dissociate into ions according to the equation: AgCl (s) ⇌ Ag+(aq) + Cl-(aq) If M is the m...
Q: Why is CCl4 used in a bromination test?
A: Bromination test in organic chemistry is done to determine the presence of unsaturation in organic c...
Q: The stability const.(formation const.) of EDTA is (ionization const.) is 8.35 x 10-22. when the inst...
A: We have given that for EDTA The instability constant = 8.35×10-22 And we have to find the stabilit...
Q: Read the question carefully and encircle the correct answer.1. What law explains the mechanism of ga...
A: The gas laws are a set of rules that control how gases behave by establishing links between the fore...
Q: Imagine that we have a saturated aqueous solution of NaCl. Please choose the correct term to fill in...
A: We are given the NaCl solution.
Q: Q: How is salicylic acid residue detected when preparing aspirin
A: Aspirin is an important analgesic and antipyretic medicine.It is prepared on reaction of salicylic a...
Q: Determine the % atom economy for the production of 1-bromobutane from 1-butanol (reaction shown belo...
A: A numerical problem based on atom economy, which is to be accomplished.
Q: Note: Provide a clear & complete solution
A: Introduction : The reaction between methane and chlorine is a type of substitution reaction.Methane...
Q: Q2// Draw the chemical structures for common name below ? 1. Supirdyl 2. Spearmint oil 3. Menthol 4....
A:
Q: Use the standard of enthalpies table to determine the change in enthalpy for: SO 2(g) + ½ O 2(g) ---...
A: total energy needed for completion of reaction is defined as enthalpy of reaction.
Q: 4
A: Role of acid in Cyclohexene synthesis is not only limited to form carbocation, i.e., as acid. But al...
Q: Indicate which other analytical techniques TGA can be coupled with and what would be the advantages?
A:
Q: 14. For the following problem, determine the order of the reaction with respect to A and the rate co...
A: time(s) [A] mol L-1 ln[A] ...
Q: Consider a cell buffered at pH 2.00 and consists of Fe2+(aq) (0.004 M), Fe3+(aq) (0.070 M) and MnO4-...
A: Given, concentration of Fe2+(aq) = 0.004 M Concentration of Fe3+(aq) = 0.070 M Concentration of MnO4...
Q: Calorimetry NaOH using a coffec-cup calorimeter which was calibrated using 5.00 mL of 0.900 M HCl an...
A: A coffee-cup calorimeter is made up of a coffee cup, water, a thermometer, and finally a reactant pl...
Q: The freezing point of pure water is 0.0°C. How many grams of ethylene glycol (C2H6O2) must be mixed ...
A:
Q: a. Reaction rate is expressed in terms of changes in concentration of reactants and products. Write ...
A:
Q: Use the standard of enthalpies table to determine the change in enthalpy for: NH 3(g) + HCl (g) ----...
A:
Q: What happens when you drink Ibuprofen and Aspirin together. How would their structures react with ea...
A: Given : What happens when you drink Ibuprofen and Aspirin together. How would their structures ...
Q: a.The decomposition of hydrogen peroxide is a first-order process with a rate constant of 1.06 × 10-...
A: The question has two subparts which has been solved below
Whats the name of this structure
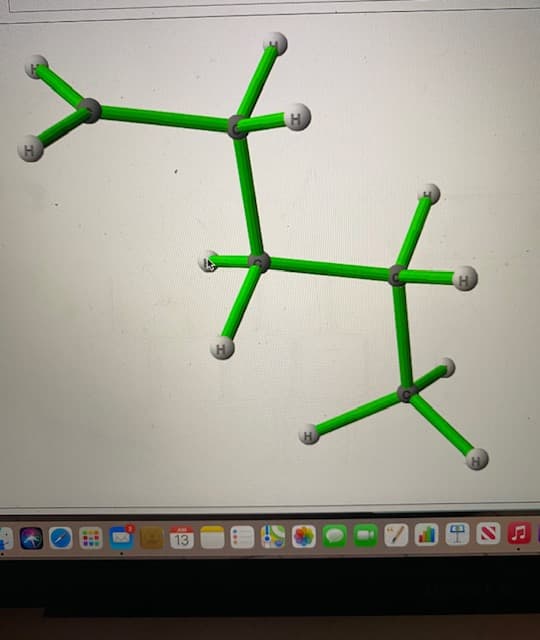
Step by step
Solved in 2 steps with 2 images


