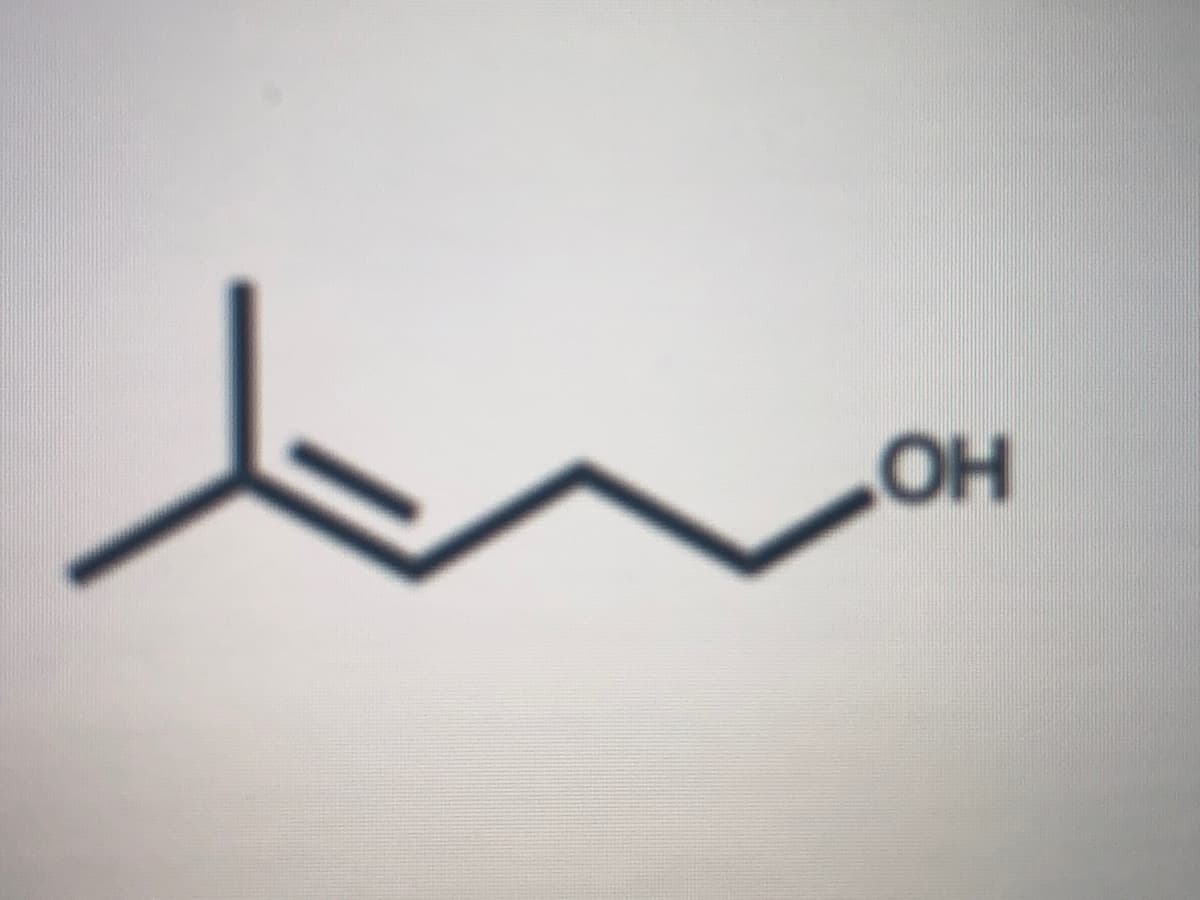
Q: What option would be best to synthesize the following alcohol? OH Link O 1-methylcyclopentene + hydr...
A:
Q: If this element is in period 5, its valence electron configuration is
A:
Q: . Calculate the equilibrium constant at 25°C for the reaction H2 + 2Fe 3+→ 2H+ + Fe2+ E° H+|H2 = 0.0...
A:
Q: Balance the following equations. Classify the type of chemical reaction. (NH),Cr,0, > N + Cr,0, + H,...
A: As you have posted multipart question. We will solve first three parts. Solution: For balancing the...
Q: redict the major product for the reaction shown.
A:
Q: Which reagents react with an alkene by syn addition? Select all that apply. Incorrect selections wil...
A: The stereoselectivity during the addition reactions of alkenes leads to syn and anti-products. In sy...
Q: In preparation of Copper (I) Chloride, the mass of copper is 1.0762. Calculate moles of Copper Foil...
A:
Q: A mixture of gases is prepared from 125 gof O2 and 18.0 g of H2. After the reaction of O2 and H2 is ...
A: Mass of O2 = 125 g Mass of H2 = 18 g
Q: Which 'H-NMR signal would be the MOST downfield? В в C C D D Ан H B A. A
A: 1- In this question, we will see that which protons is most downfield in 1HNMR spectrum. You can se...
Q: - Identify if the Oxidation product form is: Aldehydes, Ketones and no reaction. 2 Dontonol
A: Primary alcohol (1°) oxidises to aldehyde, secondary alcohol (2°) oxidises to ketones and tertiary a...
Q: FeBr3 Br2
A:
Q: B. Surface Area 1. Weigh a small iron nail to the nearest hundredths of a gram and weigh an equal a...
A:
Q: What alkene is best used to produce the following compound? H20/H2SO4 OH
A:
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI ...
A: Energy of electron in ground state is 350 zepta j. Energy of electron in first exited state is 600 z...
Q: ANSWER: Benedict's CH3-CH2 C-HJoluion a. CH3-CH2- CH2-Ċ- H H2 Mi b. Tallen's c CHs - Ct- Ċ - CH3 vol...
A:
Q: Predict the products (if any) that will be formed by the reaction below. If no reaction occurs, writ...
A:
Q: Which of these molecules best corresponds to the below IR spectrum and the chemical formula. 0.8 C,H...
A: Functional group in a compound give characteristic peak IR spectrum due to different bond strength a...
Q: To answer the questions, interpret the following Lewis diagram for NO3 . :0 1. For the central nitro...
A: Lewis structures
Q: What are the mandatory components required for column Chromatography? Please shortly answer at your...
A: Columns chromatography is used for separation of mixtures By using stationary and mobile Phase.
Q: It takes 614./kJmol to break a carbon-carbon double bond. Calculate the maximum wavelength of light ...
A: Given: The amount of energy required to break a carbon-carbon double bond = 614 kJ/mol We have to ca...
Q: contain benzene (C6H6) at molarity levels higher than concentrations considered lethal for rats. A s...
A:
Q: What is the preferred IUPAC name of the alkyne in Figure 2? [Numbers are separated by commas and num...
A: Rules- 1) select the parent chain , it is the longest chain containing triple bond 2) Then number th...
Q: Scre 2. structures. a) b) HO. c) OH d) H. e) OH f) odi worle lidgos NH CH-CH C Deduce the molecular ...
A:
Q: 4. Label the chiral centers in the following molecules with an asterisk (*
A:
Q: 2. Copper has two stable isotopes with the following masses and % abundances: Cu-63 (62.93 amu, 69.0...
A: Calculation of average masses: As per our guideline we have to answer first question only.
Q: Activity 1. Carbohydrates Study the structure and answer the question below. CH2OH C=0 H-C-OH H-C-OH...
A: The structure of glucose , fructose and Galactose have same elements but the orientation of group...
Q: The combustion of methane (CH4) produces carbon dioxide and water. CH4 + 2 O2 → CO2 + 2 H2O Assume t...
A: The combustion of methane (CH4) produces carbon dioxide and water. CH4 + 2O2 → CO2...
Q: Balancing Redox Reaction 1. HNO3 + H2S ➡️ S + NO + H2O 2. K2Cr2O7 + KI + H3PO4➡️ I2 + CrPO4 + K3PO4 ...
A: 1) HNO3 + H2S → S + NO + H2O
Q: Figure 11 O CH3 FeBr3 -c-C-CH2-CH3 CH3
A: Formation of acylated benzene is a well established reaction occuring in the presence of acyl halide...
Q: 3. True or False. a. The relation d In P/ dT AvapH / RT² should not be applied to solid-liquid trans...
A:
Q: 5.) The atomic weights of three compounds are shown. Use a linear system and Cramer's rule to find t...
A:
Q: For each of the following species, determine the number of protons and the number of neutrons in the...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: how many lone pairs of electrons would there have to be on a SN=6 central atom for it to have a line...
A: SN = 6 leads to octahedral geometry i.e geometry consist of 6 sp3d2 arranged in octahedral geometry.
Q: A mixture of 100.0 g of CS2 and 200.0 g Cl2 is passed through a hot reaction tube, where the followi...
A:
Q: The following reaction has two possible products A and B. Which is the major product formed?
A: Reduction of alkenes to alkanes takes place under H2 / Pt
Q: what is the volume of the unit cell in pm3 and in cm3?
A: Given- Metallic barium crystallizes in a body-centered cubic lattice. Metallic radius of Ba is 217 p...
Q: Na oH 4. solid copper(II) carbonate is heated oules tranred durig 0.22 bas (HOh2y0.21 5. solid bariu...
A: Here we have to write the balanced chemical equation and net ionic equation for reactions in solutio...
Q: 3.52 Avobenzone and dioxybenzone are two commercial sunscreens. Using the principles of solubility, ...
A:
Q: Predict the major organic product of the following reaction. NH2 1. NaNO2, HCI 2. Hао NO2 N(CH3)2 HO...
A: The given reaction proceed via diazonium chloride salt formation.
Q: Make an electron-flow-mechanism for this synthetic scheme. This involves predicting major and by. pr...
A: Solution -
Q: (i) List and depict all the symmetry elements and deduce the point group. H CI CI H A
A:
Q: The standard enthalpy of formation of ethylbenzene is –12.5 kJ mol-1 Calculate its standard enthalpy...
A:
Q: How many liters of 0.305 M KPO4 solution are necessary to completely react with 187 mL of 0.0184 M N...
A:
Q: It is usually recommended to clean the glass surfaces of a refractometer with ethanol or isopropyl a...
A: Refractometer is a device that measure the degree to which light changes direction called as angle o...
Q: 1.PROPERTIES OF POLYUNSATURATED FATS: I. Forms single bonds and double bonds with hydrogen II. Under...
A: I am providing solution for only 1 question as per the guidelines. Please post other questions separ...
Q: The constant heat capacity oe a sample perpect press ure of a ZAMOOTAA hren of punad SOM pound CP / ...
A: The above problem is thermodynamic problem. At constant pressure and at constant volume , Perfect ga...
Q: 3. Determine the absolute configuration of the following molecules. а. b. CI
A: R and S Configuration: Steps and Rules(1): Give each atom connected to the chiral center a priority...
Q: Show the symmetry operations for all the symmetry elements present, and determine the transformation...
A:
Q: titration
A:
Q: Other than an arene, identify the other key functional group found in styrene.
A:
Step by step
Solved in 3 steps with 1 images


