Show the symmetry operations for all the symmetry elements present, and determine the transformation matrices by choosing the basis set as (a) all the atoms in A (b) CO groups in C molecules.
Show the symmetry operations for all the symmetry elements present, and determine the transformation matrices by choosing the basis set as (a) all the atoms in A (b) CO groups in C molecules.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter13: Introduction To Symmetry In Quantum Mechanics
Section: Chapter Questions
Problem 13.41E: a Unlike methane, bromochlorofluoromethane (CHBrClF) is chiral. Determine all symmetry elements that...
Related questions
Question
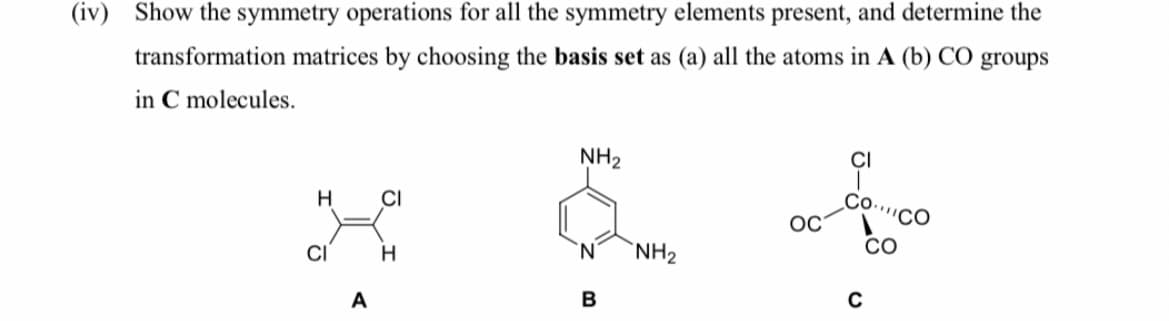
Transcribed Image Text:(iv) Show the symmetry operations for all the symmetry elements present, and determine the
transformation matrices by choosing the basis set as (a) all the atoms in A (b) CO groups
in C molecules.
NH2
CI
H
CI
Со.
''CO
OC
CI
H.
'N'
NH2
CO
A
B
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,