Which of the following deviations from the experiment could cause a decrease in the calculated mass of Mg(s)? a. Incorrectly recording the barometric pressure. b. Using a beaker with a wider diameter. c. Using a eudiometer of different length. d. Not accounting for the vapor pressure of water. Which of the following deviations from the experiment would most considerably cause an increase in the final calculated %yield of the Mg(s)? a. Placing 13.0 mL instead of 10.0 mL of 6.0 M HCI into the eudiometer. b. Immediately reading the volume of gas right after the reaction ceased. Not adding enough water to fill the eudiometer to the brim, leaving a headspace after inverting the eudiometer. С. d. Consciously inserting 15.0 mg of folded Mg ribbon instead of the prescribed 20 to 30 mg into the eudiometer. Which of the following correctly explains why the vapor pressure of water has to be included in the calculations? a. As the H2(g) is bubbled up the reaction solution, some of the H2(g) molecules are solubilized by water in the aqueous solution, effectively decreasing the measured pressure above. b. The pressure exerted by the evolved H2(g) is reduced because of the co-presence of water vapor, thus Pwater Some of the liquid water evaporates into the gas, and increases the total pressure generated by the trapped gas. d. The pressure contributed by the H2(g) is greater than the measured total pressure because of the contribution of partial pressure of water vapor. has to be added to Pdry gas. vapor С. Recap of the Experiment Record Volume of the Place 10.0 Cover the enclosed gas, the height difference Insert folded mL of 6.0 M mouth of the Mg ribbon (approx. 0.02-0.03 g) Add water HCI into a eudiometer with to fill between the water finger and invert into a glass that is 2/3 filled w/ eudiometer eudiometer levels inside and partially filled w/ distilled to the brim. into the outside the eudiometer, eudiometer. and the temperature of water. water. the water outside.
Which of the following deviations from the experiment could cause a decrease in the calculated mass of Mg(s)? a. Incorrectly recording the barometric pressure. b. Using a beaker with a wider diameter. c. Using a eudiometer of different length. d. Not accounting for the vapor pressure of water. Which of the following deviations from the experiment would most considerably cause an increase in the final calculated %yield of the Mg(s)? a. Placing 13.0 mL instead of 10.0 mL of 6.0 M HCI into the eudiometer. b. Immediately reading the volume of gas right after the reaction ceased. Not adding enough water to fill the eudiometer to the brim, leaving a headspace after inverting the eudiometer. С. d. Consciously inserting 15.0 mg of folded Mg ribbon instead of the prescribed 20 to 30 mg into the eudiometer. Which of the following correctly explains why the vapor pressure of water has to be included in the calculations? a. As the H2(g) is bubbled up the reaction solution, some of the H2(g) molecules are solubilized by water in the aqueous solution, effectively decreasing the measured pressure above. b. The pressure exerted by the evolved H2(g) is reduced because of the co-presence of water vapor, thus Pwater Some of the liquid water evaporates into the gas, and increases the total pressure generated by the trapped gas. d. The pressure contributed by the H2(g) is greater than the measured total pressure because of the contribution of partial pressure of water vapor. has to be added to Pdry gas. vapor С. Recap of the Experiment Record Volume of the Place 10.0 Cover the enclosed gas, the height difference Insert folded mL of 6.0 M mouth of the Mg ribbon (approx. 0.02-0.03 g) Add water HCI into a eudiometer with to fill between the water finger and invert into a glass that is 2/3 filled w/ eudiometer eudiometer levels inside and partially filled w/ distilled to the brim. into the outside the eudiometer, eudiometer. and the temperature of water. water. the water outside.
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter9: Gases
Section: Chapter Questions
Problem 67E: The chlorofluorocarbon CCl2F2 can be recycled into a different compound by reaction with hydrogen to...
Related questions
Question
Hi! I need answers on these multiple-choices questions. The recap of the experiment can be seen below. This experiment is about testing the

Transcribed Image Text:Which of the following deviations from the experiment could cause a decrease in the calculated mass of
Mg(s)?
a. Incorrectly recording the barometric pressure.
b. Using a beaker with a wider diameter.
c. Using a eudiometer of different length.
d. Not accounting for the vapor pressure of water.
Which of the following deviations from the experiment would most considerably cause an increase in the
final calculated %yield of the Mg(s)?
a. Placing 13.0 mL instead of 10.0 mL of 6.0 M HCI into the eudiometer.
b. Immediately reading the volume of gas right after the reaction ceased.
Not adding enough water to fill the eudiometer to the brim, leaving a headspace after inverting
the eudiometer.
С.
d. Consciously inserting 15.0 mg of folded Mg ribbon instead of the prescribed 20 to 30 mg into the
eudiometer.
Which of the following correctly explains why the vapor pressure of water has to be included in the
calculations?
a. As the H2(g) is bubbled up the reaction solution, some of the H2(g) molecules are solubilized by
water in the aqueous solution, effectively decreasing the measured pressure above.
b. The pressure exerted by the evolved H2(g) is reduced because of the co-presence of water vapor,
thus Pwater
Some of the liquid water evaporates into the gas, and increases the total pressure generated by
the trapped gas.
d. The pressure contributed by the H2(g) is greater than the measured total pressure because of the
contribution of partial pressure of water vapor.
has to be added to Pdry gas.
vapor
С.
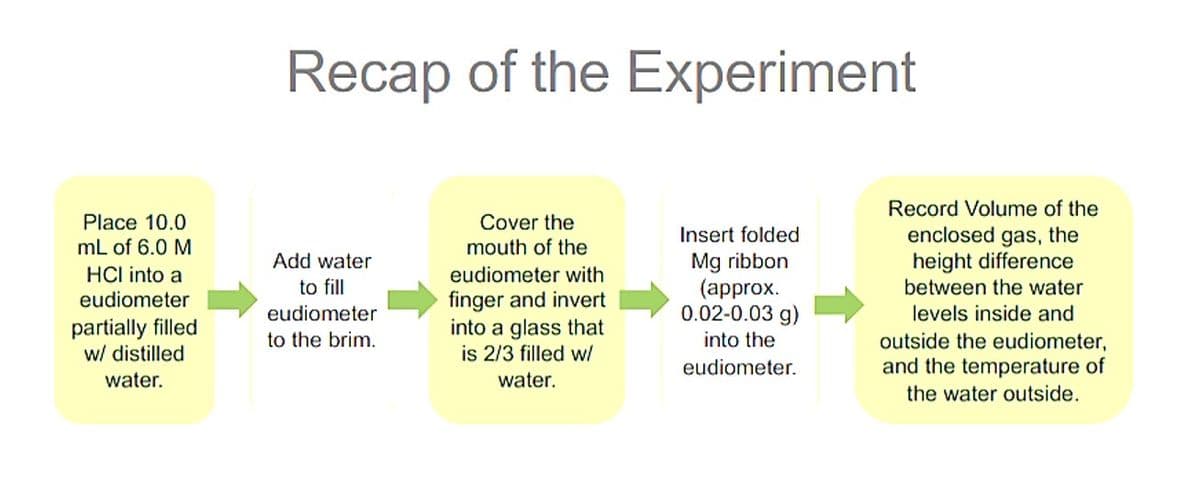
Transcribed Image Text:Recap of the Experiment
Record Volume of the
Place 10.0
Cover the
enclosed gas, the
height difference
Insert folded
mL of 6.0 M
mouth of the
Mg ribbon
(approx.
0.02-0.03 g)
Add water
HCI into a
eudiometer with
to fill
between the water
finger and invert
into a glass that
is 2/3 filled w/
eudiometer
eudiometer
levels inside and
partially filled
w/ distilled
to the brim.
into the
outside the eudiometer,
eudiometer.
and the temperature of
water.
water.
the water outside.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning