
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 39E
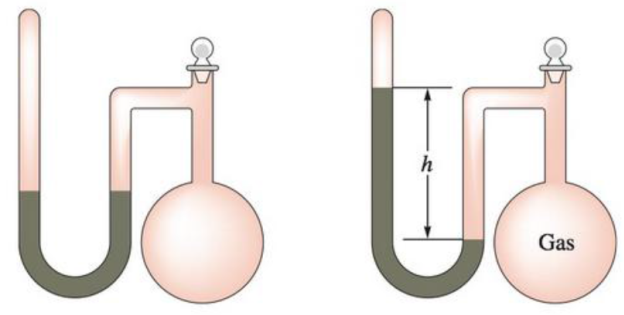
A sealed-tube manometer (as shown below) can be used to measure pressures below atmospheric pressure. The tube above the mercury is evacuated. When there is a vacuum in the flask, the mercury levels in both arms of the U-tube are equal. If a gaseous sample is introduced into the flask, the mercury levels are different. The difference h is a measure of the pressure of the gas inside the flask. If h is equal to 6.5 cm, calcúlate the pressure in the flask in torr, pascals, and atmospheres.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 8 Solutions
Chemistry: An Atoms First Approach
Ch. 8 - Prob. 1RQCh. 8 - Prob. 2RQCh. 8 - Prob. 3RQCh. 8 - Prob. 4RQCh. 8 - Prob. 5RQCh. 8 - Prob. 6RQCh. 8 - Prob. 7RQCh. 8 - Prob. 8RQCh. 8 - Prob. 9RQCh. 8 - Why do real gases not always behave ideally? Under...
Ch. 8 - Prob. 3ALQCh. 8 - Prob. 4ALQCh. 8 - Prob. 6ALQCh. 8 - Prob. 8ALQCh. 8 - Prob. 11ALQCh. 8 - Prob. 12ALQCh. 8 - Prob. 15ALQCh. 8 - Prob. 16ALQCh. 8 - Draw molecular-level views that show the...Ch. 8 - Prob. 20QCh. 8 - Prob. 21QCh. 8 - Prob. 22QCh. 8 - Prob. 23QCh. 8 - Prob. 24QCh. 8 - Prob. 25QCh. 8 - Consider two different containers, each filled...Ch. 8 - Prob. 27QCh. 8 - Prob. 28QCh. 8 - Prob. 29QCh. 8 - Prob. 30QCh. 8 - Prob. 31QCh. 8 - Prob. 32QCh. 8 - Prob. 33QCh. 8 - Prob. 34QCh. 8 - Prob. 35QCh. 8 - Prob. 36QCh. 8 - Prob. 37ECh. 8 - Prob. 38ECh. 8 - A sealed-tube manometer (as shown below) can be...Ch. 8 - Prob. 40ECh. 8 - A diagram for an open-tube manometer is shown...Ch. 8 - Prob. 42ECh. 8 - Prob. 43ECh. 8 - Prob. 44ECh. 8 - Prob. 45ECh. 8 - Prob. 46ECh. 8 - Prob. 47ECh. 8 - Prob. 48ECh. 8 - Prob. 49ECh. 8 - Prob. 50ECh. 8 - The Steel reaction vessel of a bomb calorimeter,...Ch. 8 - A 5.0-L flask contains 0.60 g O2 at a temperature...Ch. 8 - Prob. 53ECh. 8 - A person accidentally swallows a drop of liquid...Ch. 8 - A gas sample containing 1.50 moles at 25C exerts a...Ch. 8 - Prob. 56ECh. 8 - Prob. 57ECh. 8 - What will be the effect on the volume of an ideal...Ch. 8 - Prob. 59ECh. 8 - Prob. 60ECh. 8 - An ideal gas is contained in a cylinder with a...Ch. 8 - Prob. 62ECh. 8 - A sealed balloon is filled with 1.00 L helium at...Ch. 8 - Prob. 64ECh. 8 - Consider the following reaction:...Ch. 8 - A student adds 4.00 g of dry ice (solid CO2) to an...Ch. 8 - Air bags are activated when a severe impact causes...Ch. 8 - Concentrated hydrogen peroxide solutions are...Ch. 8 - In 1897 the Swedish explorer Andre tried to reach...Ch. 8 - Sulfur trioxide, SO3, is produced in enormous...Ch. 8 - A 15.0-L rigid container was charged with 0.500...Ch. 8 - An important process for the production of...Ch. 8 - Consider the reaction between 50.0 mL liquid...Ch. 8 - Urea (H2NCONH2) is used extensively as a nitrogen...Ch. 8 - Prob. 75ECh. 8 - Prob. 76ECh. 8 - Prob. 77ECh. 8 - A compound has the empirical formula CHCl. A...Ch. 8 - Prob. 79ECh. 8 - Given that a sample of air is made up of nitrogen,...Ch. 8 - Prob. 81ECh. 8 - Prob. 82ECh. 8 - Prob. 83ECh. 8 - Prob. 84ECh. 8 - Consider the flasks in the following diagram. What...Ch. 8 - Consider the flask apparatus in Exercise 85, which...Ch. 8 - Prob. 87ECh. 8 - At 0C a 1.0-L flask contains 5.0 102 mole of N2,...Ch. 8 - Prob. 89ECh. 8 - A tank contains a mixture of 52.5 g oxygen gas and...Ch. 8 - Prob. 91ECh. 8 - Helium is collected over water at 25C and 1.00 atm...Ch. 8 - At elevated temperatures, sodium chlorate...Ch. 8 - Xenon and fluorine will react to form binary...Ch. 8 - Methanol (CH3OH) can be produced by the following...Ch. 8 - In the Mthode Champenoise, grape juice is...Ch. 8 - Hydrogen azide, HN3, decomposes on heating by the...Ch. 8 - Prob. 98ECh. 8 - Some very effective rocket fuels are composed of...Ch. 8 - The oxides of Group 2A metals (symbolized by M...Ch. 8 - Prob. 101ECh. 8 - Prob. 102ECh. 8 - Prob. 103ECh. 8 - Prob. 104ECh. 8 - Prob. 105ECh. 8 - Prob. 106ECh. 8 - Prob. 107ECh. 8 - Prob. 108ECh. 8 - Prob. 109ECh. 8 - Prob. 110ECh. 8 - Prob. 111ECh. 8 - Prob. 112ECh. 8 - Prob. 113ECh. 8 - Prob. 114ECh. 8 - Prob. 115ECh. 8 - Prob. 116ECh. 8 - Prob. 117ECh. 8 - Prob. 118ECh. 8 - Prob. 119ECh. 8 - Prob. 120ECh. 8 - Prob. 121ECh. 8 - Prob. 122ECh. 8 - Prob. 123AECh. 8 - At STP, 1.0 L Br2 reacts completely with 3.0 L F2,...Ch. 8 - Prob. 125AECh. 8 - A 2.747g sample of manganese metal is reacted with...Ch. 8 - Prob. 127AECh. 8 - Cyclopropane, a gas that when mixed with oxygen is...Ch. 8 - The nitrogen content of organic compounds can be...Ch. 8 - Prob. 130AECh. 8 - A 15.0L tank is filled with H2 to a pressure of...Ch. 8 - A spherical glass container of unknown volume...Ch. 8 - Prob. 133AECh. 8 - A 20.0L stainless steel container at 25C was...Ch. 8 - Metallic molybdenum can be produced from the...Ch. 8 - Prob. 136AECh. 8 - Prob. 137AECh. 8 - One of the chemical controversies of the...Ch. 8 - An organic compound contains C, H, N, and O....Ch. 8 - Prob. 140AECh. 8 - The total volume of hydrogen gas needed to fill...Ch. 8 - Prob. 142AECh. 8 - Prob. 143CWPCh. 8 - Prob. 144CWPCh. 8 - A certain flexible weather balloon contains helium...Ch. 8 - Prob. 146CWPCh. 8 - A 20.0L nickel container was charged with 0.859...Ch. 8 - Consider the unbalanced chemical equation below:...Ch. 8 - Prob. 149CWPCh. 8 - Which of the following statements is(are) true? a....Ch. 8 - A chemist weighed out 5.14 g of a mixture...Ch. 8 - A mixture of chromium and zinc weighing 0.362 g...Ch. 8 - Prob. 153CPCh. 8 - You have an equimolar mixture of the gases SO2 and...Ch. 8 - Methane (CH4) gas flows into a combustion chamber...Ch. 8 - Prob. 156CPCh. 8 - Prob. 157CPCh. 8 - Prob. 158CPCh. 8 - You have a helium balloon at 1.00 atm and 25C. You...Ch. 8 - Prob. 160CPCh. 8 - You are given an unknown gaseous binary compound...Ch. 8 - Prob. 162CPCh. 8 - Calculate w and E when 1 mole of a liquid is...Ch. 8 - The preparation of NO2(g) from N2(g) and O2(g) is...Ch. 8 - In the presence of nitric acid, UO2+ undergoes a...Ch. 8 - Silane, SiH4, is the silicon analogue of methane,...Ch. 8 - Prob. 167IPCh. 8 - Prob. 168IPCh. 8 - Prob. 169MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- You have an equimolar mixture of the gases SO2 and O2, along with some He, in a container fitted with a piston. The density of this mixture at STP is 1.924 g/L. Assume ideal behavior and constant temperature and pressure. a. What is the mole fraction of He in the original mixture? b. The SO2 and O2 react to completion to form SO3. What is the density of the gas mixture after the reaction is complete?arrow_forwardAcetone peroxide, C9H18O6(s), is a powerful but highly unstable explosive that does not contain nitrogen. It can pass undetected through scanners designed to detect the presence of nitrogen in explosives like TNT (trinitrotoluene, C7H5N3O6), or ammonium nitrate. (a) Write a balanced equation for the combustion (burning in oxygen) of acetone peroxide producing steam and carbon dioxide. (b) What pressure is generated in a 2.00-L bottle when 5.00 g of acetone peroxide is ignited to 555C and burned in air? Assume 100% combustion.arrow_forward
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY