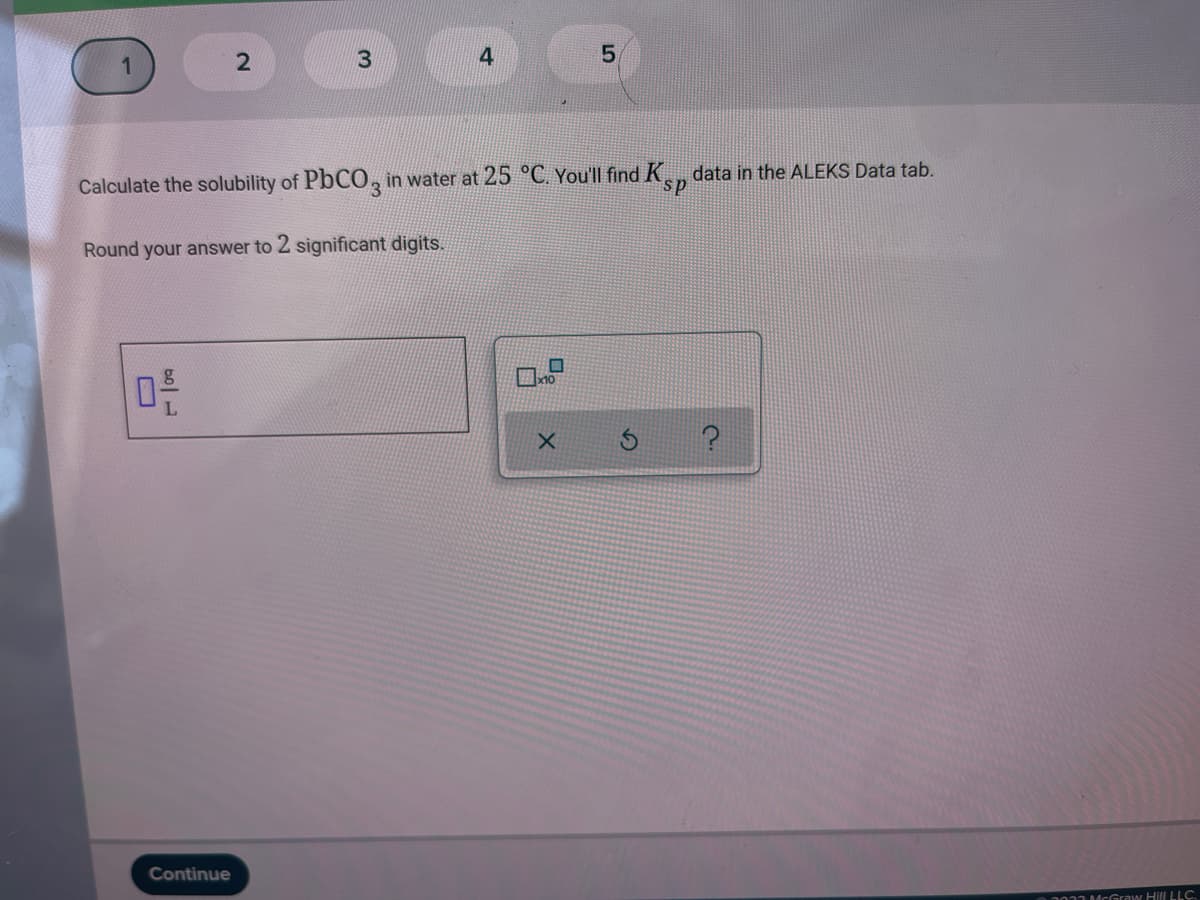
4 5, Calculate the solubility of PbCO, in water at 25 °C. You'll find K, data in the ALEKS Data tab. Round your answer to 2 significant digits. Continue 001McGraw Hill LLC. 2.
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Entropy is the degree of disorderness. Increasing order of entropy, solid < liquid <…
Q: Write the balanced nuclear equation for each of the processes described below. 1. beta emission of…
A:
Q: 0.750 M KOH(aq) is added to a 100.0 mL 0.500 M CH3COOH/NaCH3COO buffer. What is the expected pH of…
A: Given pKa = 4.74 Molarity of Acid = 0.50 M Molarity of salt = 0.50 M Volume of buffer = 100 mL Mmol…
Q: positron emission of E 76 35 Br neutron emission of xe 131y 54Xe
A:
Q: The acid-dissociation constant of hydrocyanic acid (HCN) at 25.0°C is 4.9 × 10-10. What is the pH of…
A: Given, Concentration of the sodium cyanide (NaCN) solution = 0.080 M Acid dissociation constant…
Q: What and how many stereoisomers are present in Ramipril, a drug used to treat health conditions such…
A:
Q: rite the balanced nuclear equation for each of the processes described belca 1. positron emission of…
A: For all the given reactions, there will be either change in atomic number or mass number or change…
Q: A small amount of Mg-EDTA is often added in the titrimetric determination of water hardness. Why?…
A: EDTA a complexing agent designed to bind metal ions quantitatively, forming stable, water soluble…
Q: Which statement best describes the reaction TiCl4(l) + 2H2O(l) --> TiO2(s) + 4HCl(aq) at a low…
A:
Q: The Kea for the equilibrium below is 3.52 x 10-2 at 480.0 °C. 2Cl2 (g) + 2H20 (g) = 4HCI (g) + 02…
A: Given:: 2Cl2 + 2H2O <=> 4HCl + O2 -----(i) K1=3.52×10-2
Q: At 25.0° C, a 10.00 L vessel is filled with 5.00 atm of Gas A and 7.89 atm of Gas B. What is the…
A:
Q: The pH of a solution prepared by mixing 50.0 mL of 0.125 M NaOH and 30.0 mL of 0.125 M HNO3 is…
A:
Q: gasoline (C8H18) in a car engine, results in a large amount of energy released as heat. 2 C8H18 (l)…
A: Entropy is the degree of randomness and disorder. As number of gaseous moles in product are higher…
Q: The gas in a 250.0 mL piston experiences a change in pressure from 1.00 atm to 2.80 atm. What is the…
A: Given :- Initial pressure (P1) = 1.00 atm Initial volume (V1) = 250.0 mL Final pressure (P2) =…
Q: 1. Give one example of chemical derived benzene. 2. Give the uses and source 3. Show the synthesis…
A: 1. Example for benzene derivatives are phenol, toluene and aniline. 2. Uses of Phenol - lower…
Q: freezing point of the solution
A:
Q: Match the appropriate tests with the indicated procedure form the table above. Ceric Sodium Ferric…
A: This question can be answered based on tests used for functional group detection.
Q: Write the equilibrium constant expressions for each of the following balanced chemical reactions: 1.…
A: Equilibrium constant is the ratio of product of concentration of products raised to their…
Q: A 2.00 g sample of a salt (MX(s)) is added to 20.0 g of water at 22.0 °C in a calorimeter. The…
A: Given that, a 2.00 g of a sample of a salt MX(s) is added to a 20.0 g of a water at 22.0oC. So, the…
Q: It was discovered that the rate law for this reaction is given by the following equation: d[CS2] (3.…
A: Given the rate law for the reaction, -d[CS2]dt=(3.07*10-4M-1s-1)[CS2]2 In general terms,…
Q: 2.0000 g of the unknown compound is burned in a bomb calorimeter. During the combustion, the…
A: Given data mass of compound =2.0000 g molar mass of compound =148.16 gmole Initial temperature :…
Q: Consider the following reaction at 25 °C: 3 NiO(s) + 2 NH (g) → 3 Ni(s) + N,(g) + 3 H̟O(g) kJ If AG°…
A:
Q: Consider a redox reaction for which E° is a negative number. What is the sign of AG° for this…
A:
Q: Which of the following acids or bases will have the lowest pOH? Select one: O A 0.10 M H;S04 (pKa,…
A: Given data: Solution Concentration with pKa and acid or salt A 0.10 M H2SO4 , pKa = 1.92 B…
Q: - 1 attachment - Which one of the following substances will have hydrogen bonding as one of its…
A:
Q: At 1000°C, the reaction NO2(3) + SO2(3) = NO(g) + SO3(g) has K. = 5.04. Suppose 0.287 mol of NO2 and…
A:
Q: Can you please draw how you get the S configuration on borneol and camphor? + NaocI + NacI t HzO -…
A:
Q: Tochloric acid, the following tion takes place s) + 2 HCI(aq) → ZNCI2(aq) 1) culate the volume of H2…
A: Given, Temperature =30 °c = 303 K Mass of Zn = 65.7 g Moles of Zn = mass/Molar mass =…
Q: What is the solution that the concentration of [OH-]= 2.5 c 10^-9M?
A: Given:: [OH-] = 2.5×10-9 M Nature of solution = ?
Q: Provide an etfficient synthesis of each molecule using the starting materials indicated. While no…
A: We have to carry out the required synthesis. The required transformation is shown below (a)
Q: Draw the major organic product that results when this compound is treated with PCC. Do not include…
A:
Q: For each reaction, write the equilibrium expression, Kc. A) 2KI(aq) + Pb(NO3)2(aq) ⇄ PbI2(s) +…
A: Given :- A) 2KI(aq) + Pb(NO3)2(aq) ⇄ PbI2(s) + 2KNO3(aq) B) N2O(g) + 2H2O(g) ⇄ NH4NO3 (s) C) Fe2O3…
Q: 1. Calculate the pH of a solution prepared by dissolving 2.13 g of picric acid, (NO2);C6H2OH (229.11…
A:
Q: What is the mass (in grams) of 25.00 L of propane vapor (C3H8) at STP? R= 0.08314 L·bar/mol · K.
A: Detail mathematical calculation is shown below
Q: Calculate the cell potential for the galvanic cell in w
A:
Q: Q. Which of the following sets gives sec-butyl alcohol with water in an acid medium? (i) 1-butene…
A: When 1-butene is treated with water, in acidic medium then sec- butyl alcohol (2-butanol) is formed…
Q: /hich of the following would be classified as cephalins? Select all that apply.) CH2 -C-(CH2)14CH3…
A:
Q: Neptunium-237 undergoes a radioactive decay chain before it reaches nuclear stability, forming the…
A: Here we are required to radioactive decay chain reaction of Neptunium-237 to reach the final…
Q: Show the steps of the multistep synthesis including reagents and intermediates. Only the product…
A: 
Q: Calculate the values of the buffer capacity at pH = 7 and pH = 9 in 0.08 M ammonium chloride…
A:
Q: Which of the following may have caused the formation of bands of the components? (base on the…
A: It is asked in the question to answer for which reason the formation of bands of the components…
Q: Be sure to answer all parts. A freshly isolated sample of 90Y was found to have an activity of 2.9 x…
A:
Q: 4. Write the product and give mechanism. (i) *CO .Fe Ph, Et (ii) E CH3 RCN.. CRh Me,P PMes
A: The first reaction is migratory insertion of Et to CO carbon. In the second reaction, metal (Rh) act…
Q: Calculate the potential measured for the following cell at 298 K. Cr(s) | Cr3+(aq) (0.0070 M) ||…
A: Given-> Cr(s)/Cr3+(aq)(0.0070M)//Ni2+(aq)(0.97M)/Ni(s) E°cell = +0.50 Volt
Q: Balance the half-reaction that reduces iodate to iodide in an acidic aqueous solution. 10, (aq) = I…
A:
Q: 5 p.m. on Saturday. And, they are considering offering seniors a 10% discount on all private pay…
A:
Q: What volume (in L) of 0.246 M HNO3 solution is required to react completely with 38.6 mL of 0.0515 M…
A:
Q: 3. For each of the following pairs of ions, identify the stronger base: (a) OH or NHz (b) F or Ch…
A:
Q: A 100. mL aqueous solution prepared by dissolving 25.0 g of a non-electrolyte solute had a freezing…
A: Given : Kf = 1.86 °C/m Freezing point of solution (Tf) = -1.26 °C Mass of solute = 25.0 gm…
Q: A mixture of two gases with a total pressure of 2.00 atm contains 0.70 atm of Gas A. What is the…
A: Given:: Total pressure =2.00 atm PA=0.70 atm PB= ?
Step by step
Solved in 2 steps with 2 images

- The following evidence was obtained from an experiment to determine the solubility of calcium chloride at room temperature. A sample of saturated calcium chloride solution was evaporated to dryness, and the mass of solid residue was measured.EvidenceVolume of solution (mL) = 15.0Mass of empty beaker (g) = 90.54Mass of beaker and residue (g) = 101.36The solubility of calcium chloride is g/100 mLAt a certain temperature, the solubility of zinc(I) chromate is (7.6x10^-5) M. What is the Ksp? Report your answer to 2 sig figs. Note: Your answer is assumed to be reduced to the highest power possible279.28 mL of 1.22 M organic salt was mixed with 168.14 mL of Ethanol (d= 0.789 g/mL, MW 46.07g/mole). Pure ethanol has a vapor pressure of 0.059 atm what will be the vapor pressure of the solution?
- Please complete the data. PLEASE SHOW HOW DID YOU GET THE DATA. Step by step solution please. For table 1 and 2, please show the step by step solution. You may just fill in the first trials.The solubility of Ni(OH)2 in water at 25c is measured to be 4.9 x 10^-4 g/L. Use this information to calculate Ksp for Ni(OH)2.Boiling Point Elevation/Freezing Point Depression T = m K where, for freezing point depression: T = T(pure solvent) - T(solution) and for boiling point elevation: T = T(solution) - T(pure solvent) m = (# moles solute / Kg solvent) Kb = boiling point elevation constant. Kf = freezing point depression constant. Kb and Kf depend only on the SOLVENT. Below are some common values. Use these values for the calculations that follow. Solvent Formula Kb(°C / m) Kf(°C / m) Water H2O 0.512 1.86 Ethanol CH3CH2OH 1.22 1.99 Chloroform CHCl3 3.67 Benzene C6H6 2.53 5.12 Diethyl ether CH3CH2OCH2CH3 2.02
- Calculate the solubility of BaSO4 in water at 25°C. You'll find Ksp data in the ALEKS Data tab. Round your answer to 2 significant digits.A batch of 500 ?? of ??? dissolves in water to form a saturated solution at 350 ? (77 °?) where the solubility is 30% by weight of ??? in water. The solution cools at 293 ? (20 °?) at which the solubility is 25.4% by weight. Determine the number of crystals obtained if 3.5% of the original water evaporates on cooling. Also, determine the capacity of the container in which this solution is cooled if it is 1.20 times the volume of the solution. The density of the solution is ???? ??/??List 7 real-life applications in Chemical Equilibrium in a Liquid Phase and list 7 real-life applications in Distribution of a Solute between Immiscible Solvents.
- How come we are not looking at the solubility data for hot and room temperature for part a and b?An excess amount of Ba(NO3)2 was added to 2 liters of water at 25 oC until the solution is saturated. Because Ba(NO3)2 is only slightly soluble in water, the excess Ba(NO3)2 was filtered out to get a solid-free mixture. The proponents of the experiment thought about using the concept of colligative properties, specifically boiling point elevation, to determine Ksp. It was observed in their experiments that the solution boils at 100.15 deg C. Kb of water = 0.51 K-kg/mol. Barium nitrate dissociates using: Solve the ff: a) Amount of Ba(NO3)2 dissolved in grams. MW of Ba(NO3)2 = 261.3 g/molb) Solubility product, Kspc) Vapor pressure of the solution in kPaAnswers should be in 4th decimal places An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (SG=1.114; MW = 62.07 g/mole) and water at 20oC. The density of the mixture is 1.070 g/mL. Express the concentration of ethylene glycol asa) Volume percentb) Mass percentc) Molarityd) Molalitye) Mole fraction

