Use of a Graduated Cylinder Trial 1 Trial 2 Trial 3 Mass of graduated cylinder C64.107 864.063 804.079B 103.105 btragt 103-034, 839.55SE Mass of graduated cylinder + water (03.877 103.105-4,l7 38938 39.814 Mass of water 40.5 Volume of water 40.1 41 cm3 C cm³ DENSITY OF WATER g/cm³ 3 g/cm3 g/cm³ AVERAGE DENSITY OF WATER g/cm³ 3 STANDARD DEVIATION g/cm³ 7 PERCENT ERROR
Use of a Graduated Cylinder Trial 1 Trial 2 Trial 3 Mass of graduated cylinder C64.107 864.063 804.079B 103.105 btragt 103-034, 839.55SE Mass of graduated cylinder + water (03.877 103.105-4,l7 38938 39.814 Mass of water 40.5 Volume of water 40.1 41 cm3 C cm³ DENSITY OF WATER g/cm³ 3 g/cm3 g/cm³ AVERAGE DENSITY OF WATER g/cm³ 3 STANDARD DEVIATION g/cm³ 7 PERCENT ERROR
Chapter32: Gas Chromatography
Section: Chapter Questions
Problem 32.15QAP
Related questions
Question
Pls help me with the density of water, average density of water, standard deviation, and percent error
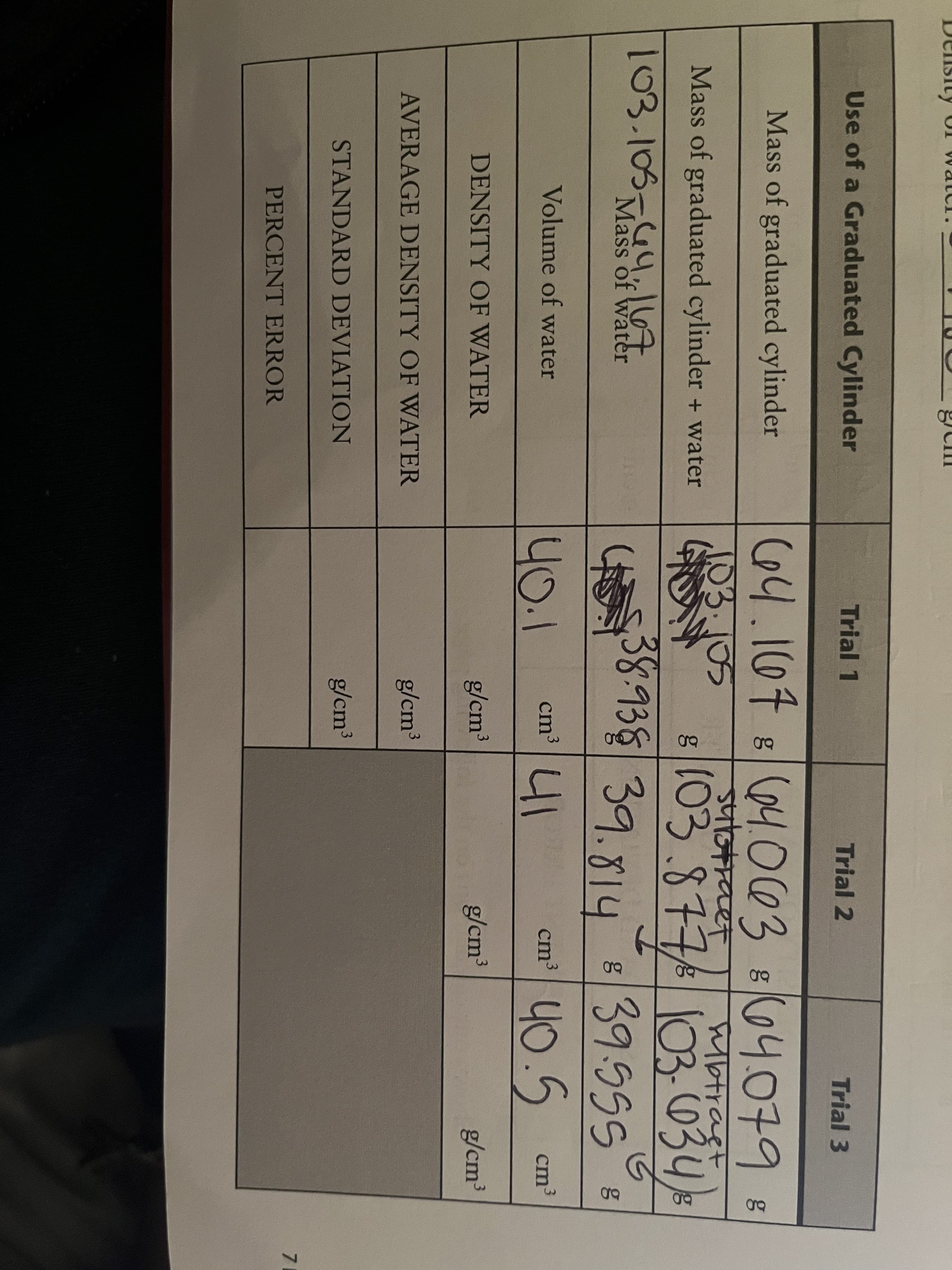
Transcribed Image Text:Use of a Graduated Cylinder
Trial 1
Trial 2
Trial 3
Mass of graduated cylinder
C64.107 864.063
804.079B
103.105
btragt
103-034,
839.55SE
Mass of graduated cylinder + water
(03.877
103.105-4,l7
38938
39.814
Mass of water
40.5
Volume of water
40.1
41
cm3
C
cm³
DENSITY OF WATER
g/cm³
3
g/cm3
g/cm³
AVERAGE DENSITY OF WATER
g/cm³
3
STANDARD DEVIATION
g/cm³
7
PERCENT ERROR
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

