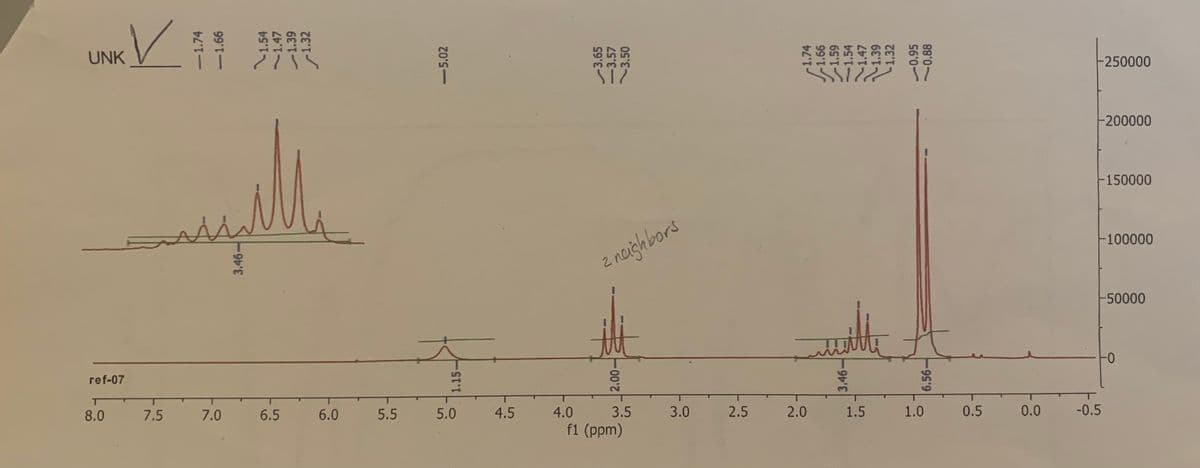
UNK -250000 -200000 -150000 100000 2 noighbors -50000 0- ref-07 8.0 7.5 7.0 6.5 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 -0.5 f1 (ppm) -5.02 c1.59 S6'0-
Q: II rome File Edit View History Bookmarks Profiles Tab Window Help Watch Gilmore Girls | Netflix (2…
A:
Q: Please help with determining the functional groups present in the IR data results of our isoamyl…
A: IR mainly used to help to determine the functional groups present in molecule
Q: A group of students compiled the data shown in data table 1 below. What is the exact calculated…
A: We have to find is the exact calculated order for H+(aq)
Q: Масс-спектр 887 100 69- thil 50 0 50 ИК-Спектр 100 Пропускание, % 100 m/z 4000 Спектр ЯМР Н (300…
A: Note : DBE = C8H15O2Br = 8- (15+1/2)+1 = 9-8 = 1. Carbonyl present. From DEPT six CH2 groups and one…
Q: Arrange the values according to magnitude. Greatest Least Answer Bank 7.6 x 10-6 3.3 x 10-2 9.0 x…
A: The value which is more than zero ( > 0 ) has the greatest magnitude and the Value which is less…
Q: ᴀ ꜱᴛᴇᴇʟ ᴛʀᴀɪɴ ʀᴀɪʟ ɪꜱ 400 ᴍᴇᴛᴇʀꜱ ʟᴏɴɢ, ɪɴ ᴍᴀʀᴄʜ ɪᴛ ɪꜱ -30 ᴅᴇɢʀᴇᴇ ᴄ ᴀɴᴅ 40 ᴅᴇɢʀᴇᴇ ᴄ ɪɴ ᴊᴜʟʏ. ᴡʜᴀᴛ ɪꜱ…
A: Detailed solution is provided.
Q: b a 1466 3333 4000 3000 2000 1500 1000 500 4000 3000 2000 1500 1000 1650 d 1450 1685 3034 3341 2000…
A: To find: the correct specturm and match the structure and to give the reason:
Q: UNK 350000 300000 250000 200000 150000 F100000 50000 ref-02 10.5 10.0 9.5 9.0 8.5 8.0 7.5 7.0 6.5…
A: 1H NMR Spectrum: A peak at ? 2.46 ppm, 3 H, singlet indicating the presence of methyl group attached…
Q: group of students compiled the data shown in data table 1 below. What is the exact calculated order…
A: This question is related to finding out the order of reaction with respect to a particular species.…
Q: Chrome File Edit View History Bookmarks Profiles Tab Window Help 32% O Purple/Black Iridescent x G…
A:
Q: cal BTU Convert 1 g - °C to lbm - °F
A: Cal/g °C =The heat needed to raise the temperature of 1gm of water bt 1°C. BTU/lb °F = The heat…
Q: The largest gem-quality diamond ever found is the Cullinan diamond, found in 1905 in South Africa.…
A: #32.d Given the weight of the diamond = 3106.75 carat Cost per diamond = $ 4 million per carat
Q: a. Convert 125.0km to centimeters: (1km=1000m 1m=100cm) b. Convert 200.0…
A: Unit conversion comprises several steps involving division or multiplication through a specified…
Q: Give two examples of the Personal Error (one of the classification of systematic error). Please do…
A: Personal errors are types of operational errors that arise due to personal limitations of the person…
Q: 1. MF C₂H₂NO LOD TRANSHI LANCETS! D 4000 3000 3328 13 2940 72 3156 79 2902 79 3087 74 1639 4 3066 68…
A:
Q: F5000 F4500 F4000 F3500 3000 F2500 2000 F1500 F1000 -500 4.5 f1 (ppm) 7.0 6.5 6.0 5.5 5.0 4.0 3.5…
A:
Q: 45 3.5 3.0 2.5 1.5 1.0 0.5 8.5 8.0 7.5 7.0 6.5 6.0 5.5 5.0 4.0 2.0 F80'7 F-99'0 SO'E T-86'1 ZI -1.31…
A: Given
Q: 4C: C H15N 100- 80 60 С 40 Я 20- 3.0 4000 2.8 3500 4H 2.6 3292 2.4 2934 2.2 2959 Зега 2876 2810 2.0…
A:
Q: Date Wednesday, November 20, 2019 9:59 AM 1017 100 95- 9어 85 8어 75 3277.4 7어 1448.4 65 1367.9 2936.2…
A: Some important IR peaks of 4-tert-butyl cyclohexanol are discussed below : A. Broad medium peak at…
Q: 52 50- 48- 46- 44 %T ransTmittance 54 3853.08 3744.19 3675.00 3648.53 6.42 2925.17 2855.10 2159.02…
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: Unknown 2.5 Rel. abund. Ret miz obund. miz 12 13 14 15 24 26 26 02 04 0.4 1.0 0.8 60 0.2 61 0.4 62…
A:
Q: fill in the structure with any assumed c & h atoms. how do you know if or if not they belong…
A: Carbon has valence electron -4 Hydrogen has valence electron -1 Hence , carbon make 4 bond .
Q: INSTRUCTIONS: Encode your final answers on the answer sheet for quizzes. For illustration-type of…
A: #1: Lineweaver-Burk Plot is drawn from the reciprocal form of Michaelis-Menten equation which is…
Q: :0: :O: VTITTO DAAMG DELLAL TTITVJTTTV Assessment - Chem 101 002 - Ac X A Achieve > Students:…
A: In a chemical compound, atoms are bonded to each other by a single, double or triple bond depending…
Q: 19- 3853.77- 3820.98- 3736.12- 3649.32- wwwww mang 2362.80 2339.65- hurbark 1751.36- 1685 79- 1604…
A: Aspirin commonly known as acetylsalicylic acid. Two functional groups ester and carboxylic acid…
Q: Part A 5100000 g 5.10×106g 5.1×106g 0.510×107g 0.51×107g 5.1×10-6g Part B 26000 s 2.60×104s…
A: Since Lowest significant digit present in the given questions is also a part of answer means same…
Q: Data: Year # of Pennies 1998 60 2000 2003 50 55 volume of Pennies, mill Ave. vol mi Ave density…
A:
Q: please highlight the correct answer
A: The natural log of the given values are provided below. Maximum size (kg) Base Metabolism…
Q: The Bay of Fundy in Nova Scotia, Canada is reported to have the largest tides in the world with high…
A:
Q: 200 180 160 140 120 100 80 60 40 20 CDS-03-756 ppm
A: Given, C13NMR spectrum
Q: Question 4 Convert 81.9x10-6 Matm to psi. Give your answer in scientific notation (e. g. 1.23E4…
A:
Q: -3800 eso 600 400 3200 -750 3000 700 2800 450 -2600 400 Sample C 2400 550 -2200 500 -2000 450 1800…
A:
Q: 100- 98- 96- 94- 92- 90- 88- 86 84 82 80 78- 76+ 4000 3533 Aña 3402.5 3500 3002.41cm-1 2916.20pm-1…
A: The IR tells about The functional group present in the molecules.
Q: 2,00000000000000000000×9,00000000000000
A: Given: 2,000000000000000000×9,00000000000000
Q: 3000 2000 1500 1000 500 HAVENUMBERI -l 3331 2932 1704 86 1467 1266 1238 13 47 970 21 667 64 4 42 53…
A:
Q: 11% O Mon File Edit View History Bookmarks Profiles Tab Window Help Chrome O S550 Mustan (6 unread)…
A: Given, Gaseous ammonia react with oxygen gas to produce nitrogen monoxide and water vapor, the moles…
Q: PIH A Achieve Students: Register for X Assessment Chem 101 002 - Ac X Canvas LMS…
A:
Q: b 1466 3333 1716 2000* 2941 4000 3000 2000 1500 1000 500 4000 3000 ' 1500 1000 500 3026 1650 d 1450…
A: To solve the question:
Q: 992 96 94 92 90 3278.8 3005 2901 88 1378 3/217.8 86 84 82 80 78 76 74 72 70- 68 66 65 4000 1697…
A:
Q: - MyEPCC с Gmail ► YouTube X o Mail - Chavez, Daniela Mi X A ALEKS-Daniela Chavez X b Chemistry…
A: Here we are required to find the standard entropy of the given Chemical reaction .
Q: Inconc 500 1000 1500 2000 2500 -3 -4 -5 y=0.0007x-4,2935 -6 R0.934 -7 1. 2.
A: First graph will definitely shows the most linear among the given three graphs. Because in that…
Q: 2KCIO3 → 2KCI + 302 Build your Model:
A: A balanced chemical reaction is that in which number of atoms of each type in reactants are same as…
Q: 24°BK → X + -je A
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: 10: C4H,NO 100 08 60 48 20 8-4 4000 1.0 3500 4H 3335.35 3.5 2947.61 2851.15 3000 2752.77 4H 3.0 2500…
A: Double bond equivalence (DBE) of the compound having molecular formula C4H9NO can be calculated by…
Q: Exp 12 step 2 product F11000 10000 -9000 F8000 7000 6000 -5000 -4000 -3000 F2000 1000 10 -1000 8.4…
A: As can be seen from the spectra peaks are:
Q: A group of students compiled the data shown in data table 1 below. What is the exact calculated…
A: For the given reaction, the order for H+(aq) is determined using trials 1 and 4 as follows.…
Q: Rb Blackboard Collaborate Ultra -2 x General Psychology -Fall 20 How to Find a Career Path Using X V…
A:
Q: .4.25×3.10=13.1750 b.3.75×7.35=27.5625 c.83.126×8.12=674.98312
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Q)What is used for heating small amounts of solids at a high temperature? a) Mortar and pestle b)…
A: Q)What is used for heating small amounts of solids at a high temperature? a) Mortar and pestle b)…
Q: 1. Convert 7 orks to oops. Conversion Statements for problems 1-4 1) 1 ahah = 10 eeps = 21ahah 3…
A: In dimensional analysis, we need to keep in mind that the numerator unit in one step must be present…
Based on the following 1H-NMR data, what is the possible structure of the alcohol C5H11OH?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- https://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4 I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this labA batch of raw milk was tested and found to contain a bacterial population of 8xl06cfu/ml. It is to be pasteurised at noc for 33 seconds. The average D value at 52°C for the mixed population is 11 min. The z value is 10°C.i)Explain the terms "D value" and "z value".ii)Calculate how many organisms will be left after pasteurisation.iii)Hpw much time will be needed at 62°C to accomplish the same degree oflethality?iv. How much time will be needed at 82°C to achieve the same degree oflethality?Use this video to answer the questions: https://www.youtube.com/watch?v=BocfSXdbaU0
- Given the following: Solutions Flask 1 Flask 2 Flask 3 0.04 M KI 25.0 mL 25.0 mL 12.5 mL 1% starch 5.00mL 5.00mL 5.00mL 0.03 M Na2S2O3 5.50 mL 5.50 mL 5.50 mL 0.05 M H2SO4 5.00 mL 5.00mL 5.00mL 0.08 M NaCl 0 0 10.0 mL and Solutions Beaker 1 Beaker 2 Beaker 3 0.04 M H2O2 25.0 mL 12.5 mL 25.0 mL deioinized water 0 12.5 mL 0 Following the procedure Run 1 is when Beaker 1 is added to Flask 1 and the timer is started giving the time recorded for the run in the table below. This is repeated with Beaker 2 and Flask 2 for Run 2 and finally Beaker 3 and Flask 3 for Run 3: The results for these runs are given below: Run Time (seconds) 1 35 2 66 3 72 Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions tables) the rates of reaction [I2]/time (calculate the [I2] from [S2O32-] and the reaction: I2(aq) + 2 S2O32-(aq) → 2I-(aq) + S4O62-(aq) Choose the correct rate law for this reaction from your calculations.…You were in the middle of a tropical rainforest and it is very humid in the environment. Do you have a glass of pure water that you have left outside for two days. Nothing has come to drink it and it has not rained either for those two days you measure the amount of water in the glass after these two days and it is exactly the same volume that was present two days before answer 22-251. State the process that is used to separate kerosene and diesel fuel. State the operating conditions of this process. 2. State the process that can be used to yield fraction of heavy fuel oil from crude oil. 3. Describe the difference between straight run naptha and cracked naptha.
- y= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)27. asap plsNano chemistry questions! True or false? 1. Bulk materials have almost constant properties regardless of their size and shape. 2. An important parameter that determines the functioning of nano materials is surface area (SA) to volume (v) ratio 3. Top-down approach are required for synthesis of nanomaterials 4. UV-visible spectroscopy can be used as a simple and reliable method for monitoring the stability of nano particles solutions.
- avg concentration: 8.937x10^-7Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72You work in a research organization that is looking for markers of various diseases that can be used as a diagnostic for the disease. It has been reported in the past that high levels of Cu are found in the sweat of people with cystic fibrosis. One of the research projects is focused on looking for high levels of Cu in samples that can be obtained non-invasively such as saliva, sweat, hair, nails, etc. The lab will analyze large samples for Cu. What instrument would you recommend purchasing to support this work, Atomic absorption spectrophotometer or an inductively coupled plasma atomic spectrophotometer? Explain the basis for your decision.



