Q: Define the structural relationship between the following molecules. CH3 он H он .Co,H H- OH VS Br OH...
A:
Q: Complete the balanced neutralization equation for the reaction below: HCIO,(aq) + CSOH(aq) →
A: Complete the balanced neutralization equation for the given above reaction--
Q: 4. Calculate the work done under isothermal conditions for 5.5 mols of an ideal gas expanding from 1...
A:
Q: etivity.do?locator%=Dassignment-take E! N Update Magnesium hydride reacts with water to produce magn...
A:
Q: The following chemical reaction takes place in aqueous solution: AgF(aq)+NH,Cl(aq) →AgCl(s)+NH,F(aq)...
A: We have to predict net ionic equation.
Q: 8-5. Find the activity coefficient of each ion at the indicated ionic strength: a. So? (u= 0.01 M) b...
A:
Q: Aqueous potassium phosphate reacts with aqueous aluminum chloride to form aqueous potassium chloride...
A: The formula for potassium phosphate, aluminium chloride, potassium chloride and aluminium phosphate ...
Q: Which of the following is NOT a strong acid? a. HNO3 b. HCIO3 с. НI d. HBr a O b O d
A: high the value of acid dissociation constant, Ka more will be the acidic strength and strong acid it...
Q: Arrange the fertilizers in decreasing order of mass percentage of nitrogen. Highest percent nitrogen...
A:
Q: A cereal contains 11.0 grams of sucrose (C12H22O11) per 60.0 grams of cereal. How many grams of cere...
A: Concept: here we can use molar concept As we know that Mole = weight/ M.W.
Q: Combustion analysis of 63.8 mg of a C, H and O containing compound produced 145.0 mg of CO2 and 59.3...
A: Given data, Mass of a C,H and O containing compound = 63.8 mg Mass of CO2 = 145.0 mg Mass of H2O ...
Q: What Quantity in moles of KBr are in 67.0 grams of KBr?
A: Given is, Mass of KBr = 67.0 g We know , Molar mass of KBr= 119.002 g/mol
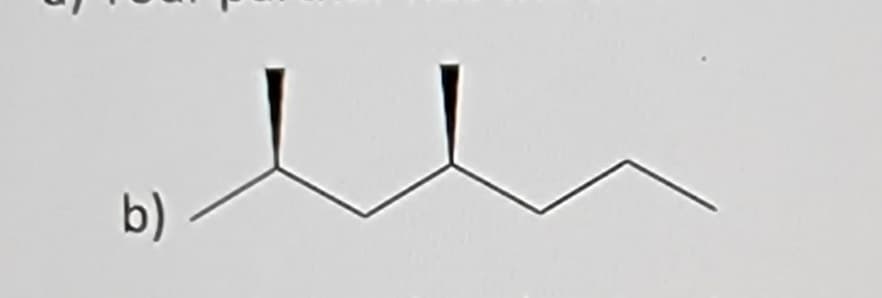
Q: 4) Explain the difference between an enantiomer and diastereomer.
A:
Q: 1. Recall the following relationship developed in lecture: ƏP - P ƏT a) What is the name of this rel...
A:
Q: Rank the indicated bonds from shortest to longest in length. a Shortest
A: There are different types of bond , Single bond , double bond and triple bond.
Q: A 79.0 mL portion of a 1.20 M solution is diluted to a total volume of 228 mL. A 114 mL portion of t...
A: We have to calculate the final concentration.
Q: Calculate the molarity of 0.550 mol of Na, S in 1.30 L of solution. molarity: Calculate the molarity...
A:
Q: There is an "8 hour average" and "1 hour average" in the US EPA's 6 criteria air pollutants. What is...
A: Given, In the US EPA 6 criteria air pollutants the average time"8 hour average " and "1 hour averag...
Q: A solution of CaCl, in water forms a mixture that is 32.5% calcium chloride by mass. If the total ma...
A: We have to calculate the mass of CaCl2 and mass of water.
Q: 23, and so on Which of the following was one of Empedocles elements (from ancient Greece)? Water O S...
A: According to the greek philosopher , Empedocles , all matters was generally composed from four eleme...
Q: A. For each set of quantum numbers below, determine and identify whether the combination of the quan...
A: Check each set one my one starting form the value of principal quantum number (n)
Q: In the grignard reaction, preparation of triphenylcarbinol summarize entire experiment in detail.
A: ph-C(ph)(OH)-ph : triphenylcarbinol ph-MgBr : Grignard reagent
Q: Write the balanced chemical equation for the reaction shown. = NO = NO, balanced equation:
A:
Q: When it is treated with sodium ethoxide in deuterated ethanol (C2H5OD), cyclopentadiene exchanges al...
A: The structure of given cyclopentadiene is depicted as follows: The chemical formula of the above st...
Q: Many metals react with halogens to give metal halides. For example, iron reacts with chlorine to giv...
A: To solve all the given questions we will use the stoichiometric ratio of the given balanced chemical...
Q: The melting point of the sample assigned to your group matches the melting point of a sample assigne...
A:
Q: Calculate the formula mass of magnesium iodite, Mg(I0,),, in atomic mass units (amu or u). formula m...
A: The sum of all mass of all atoms in the empirical formula of the compound is known as Formula mass o...
Q: The kinetic reaction data of the decomposition of substance S is given below. a. Determine the ord...
A:
Q: Identify the strongest attractive forces between the particles of each of the following Drag the app...
A: Interpretation: We have to identify the forces present in the molecules.
Q: What is the mass (in kg) of 6.89 × 1025 molecules of CO2? The molar mass of CO2 is 44.01 g/mol. O 3....
A:
Q: Methyl orange is an indicator that changes color over a range from pH =3.1 to pH =4.4 . What is Ka o...
A: Ka = 1.6 × 10-4
Q: When 1-chloro-1-methylcyclopentane is converted with sodium ethoxide in hot ethanol a product mixtur...
A: Given incomplete reaction is : 1-chloro-1-methylcyclopentane + C2H5ONa , C2H5OH --------> ? What ...
Q: Formula Valence Diagram Drawn Total Electronic Molecular Hadridiz-Polar (yor N) Electrons Bonding Do...
A:
Q: Show a dipeptide with two of your amino acids bonded to each other. Draw the structure. The amino ac...
A:
Q: A bicycle tire is inflated to a pressure of 3.74 atm at 15°C. If the tire is heated to 35°C, what is...
A: A numerical problem based on gas law, which is to be accomplished.
Q: How many milliliters of 10.0 M HC1(aq) are needed to prepare 580.0 mL of 1.00 M HCI(aq)? % = ml
A:
Q: What is the molarity of a solution of nitric acid if 0.216 g of barium hydroxide is required to neut...
A:
Q: Sulfuric acid is also known as battery acid with a Molecular Formula H2SO4 and Molar Mass of 98 gram...
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: Which of the following set of quantum numbers (n, I, mı, ms respectively) refers to the last filling...
A:
Q: 3) M. The solubility product constant of AgI is 8.3 x 10-17. A) 3.8 x 10-11 B) 3.0 x 10-10 C) 9.1 x ...
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Determine the number of NH+ and CO, ions required to form a neutral ionic compound. 2- Cations Anion...
A: the number of NH4+ and CO32- ions required to form a neutral compound:
Q: The monomers HO OH and HN- NH would react with each other to form O a polyester O a polyamide a poly...
A: Given: Monomers: Dicarboxylic Acid & Diamine
Q: What is the maximum number of electrons in an atom that can have the following set of quantum number...
A: For particular 'n' th orbital , value of 'l' is (n-1). For each l value, possible 'ml' values are fr...
Q: Ascorbic acid (C,H3O6) is also known as Vitamin C. How many molecules of C6H;O6 does a Vitamin C dri...
A:
Q: Enter your answer in the provided box. Even at high temperatures, the formation of NO is not favored...
A:
Q: 26 mL of oxygen gas at a temperature of 22 degrees Celsius is heated to 188 degrees Celsius. What is...
A:
Q: Arrange the fertilizers in decreasing order of mass percentage of nitrogen. Highest percent nitrogen...
A: Arrange the given compound in decreasing order of mass percentage of nitrogen ?
Q: Why is important to allow the saturated solution to cool down slowly during the recrystallization?
A: Recrystallization is most useful purification technique. It is used for purify solid compounds.
Q: 4.2 A groundwater used as a drinking water supply contains 4.6 mg/L of Fe2+. Calculate the amount of...
A: Here we have to calculate amount of O2 needed to oxidize Fe2+ and amount of Fe(OH)3 will be produced
Q: solution of CaCl2 is made by dissolving 55.5g of CaCl2 in enough water to yield 0.750 L of solution....
A: 1- First we will calculate the concentration of CaCl2 in 0.750L volume of the solution. moles of Ca...
Step by step
Solved in 2 steps with 2 images


