LL The vapor pressure of Substance X is measured at several temperatures: temperature vapor pressure -27. °C 0.0449 atm -16. °C 0.112 atm -5.°C 0.257 atm Use this information to calculate the enthalpy of vaporization of X. Round your answer to 2 significant digits. Be sure your answer contains a correct unit symbol. Continue O 2022 McGraw Hill LLC. All Rights Re EPIC MacBook Pro ロ号 F3 F4 F5 F2 24 4. 23 7. 6. 3. 5. A G D. B. N 8
LL The vapor pressure of Substance X is measured at several temperatures: temperature vapor pressure -27. °C 0.0449 atm -16. °C 0.112 atm -5.°C 0.257 atm Use this information to calculate the enthalpy of vaporization of X. Round your answer to 2 significant digits. Be sure your answer contains a correct unit symbol. Continue O 2022 McGraw Hill LLC. All Rights Re EPIC MacBook Pro ロ号 F3 F4 F5 F2 24 4. 23 7. 6. 3. 5. A G D. B. N 8
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter10: Solids, Liquids, And Phase Transitions
Section: Chapter Questions
Problem 31P
Related questions
Question
Transcribed Image Text:LL
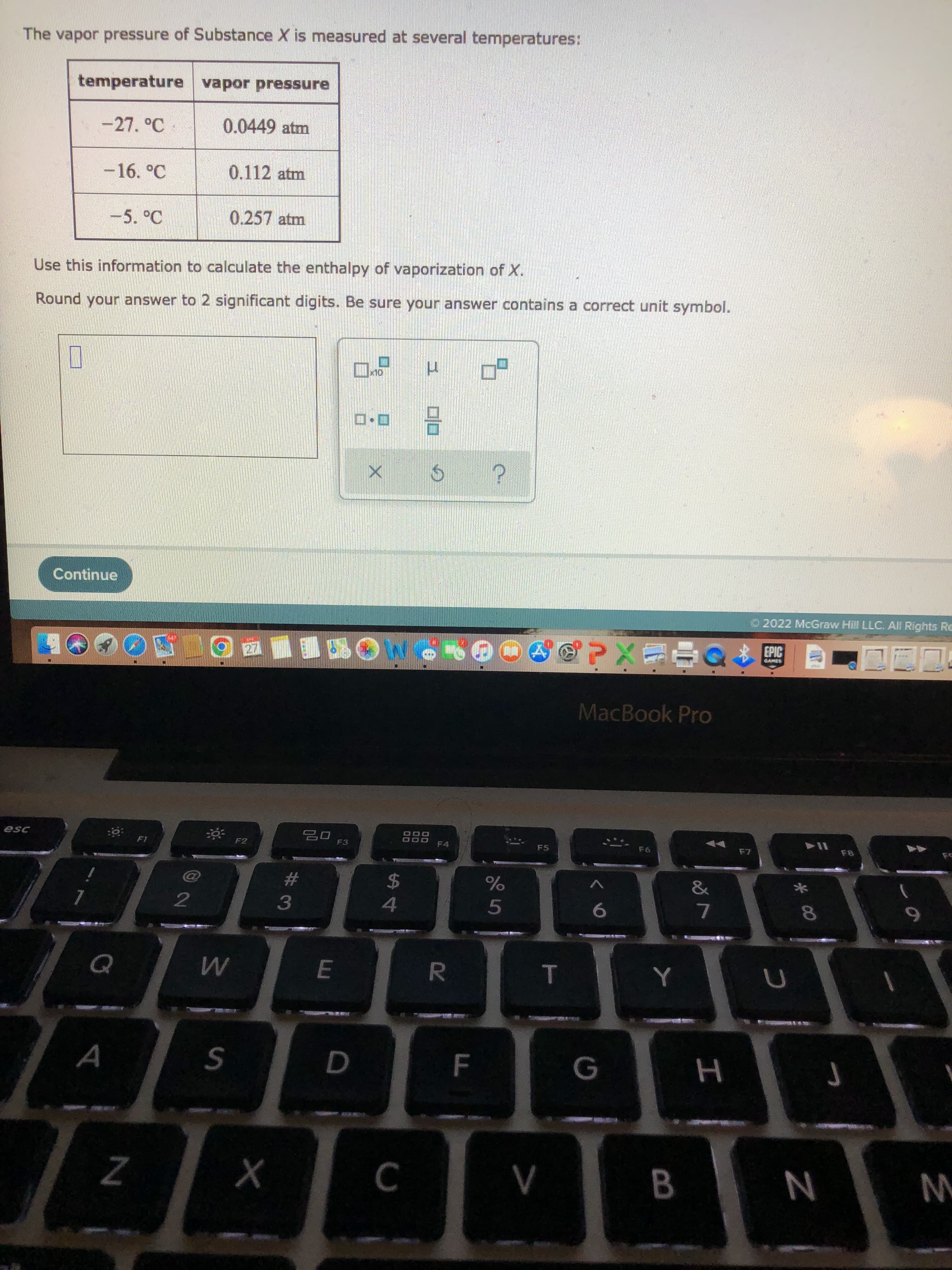
The vapor pressure of Substance X is measured at several temperatures:
temperature vapor pressure
-27. °C
0.0449 atm
-16. °C
0.112 atm
-5.°C
0.257 atm
Use this information to calculate the enthalpy of vaporization of X.
Round your answer to 2 significant digits. Be sure your answer contains a correct unit symbol.
Continue
O 2022 McGraw Hill LLC. All Rights Re
EPIC
MacBook Pro
ロ号
F3
F4
F5
F2
24
4.
23
7.
6.
3.
5.
A
G
D.
B.
N 8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning
