Q: Which has the highest entropy? Explain your answer in a separate sheet. a. H2O(s) at −50°C…
A: Which has the highest entropy ? Explain. a) H2O(s) at -50 °C b) H2O(g) at 150 °C c) H2O(l) at 100…
Q: Calculate the pH of a 0.020 M solution of phenylacetic acid, C6H5CH2COOH. What will be the pH if the…
A:
Q: с. H2N° HO, SOCI2 (еxcess) N-CH3 CH3
A: Detail mechanistic pathway is given below to find out the final product
Q: In a gas chromatographic run, anthracene peak was eluted at 7.50 min. The adjusted retention time of…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: Draw in the missing structures A and B, and then choose what could function as reagent C to complete…
A: In this question, we have to draw the missing structure A,B and the reagent C.
Q: The mass of solute per 100 ml of solution is abbreviated as (m/v). Mass is not technically the same…
A:
Q: A solution has a PH of 1.75. We conclude that A. The solution is very acidic B. The solution is only…
A: pH The pH of the solution is the negative logarithm of H+ ion concentration. pH = -log[H+] Note:…
Q: A chemist titrates 180.0 mL of a 0.4899M potassium hydroxide (KOH) solution with 0.5114M HCl…
A:
Q: 1. Give one example of chemical derived benzene. 2. Give the uses and source 3. Show the synthesis…
A: 1. Example for benzene derivatives are phenol, toluene and aniline. 2. Uses of Phenol - lower…
Q: Draw the major organic product of the following reaction conditions. CH3 1. Os04, H20 2. NalO4 ČH3
A: Detail mechanistic pathway is given below to find out the major product
Q: This molecule will undergoa bimolecular nucleophilic substitution reaction with hydraxide ion.…
A:
Q: Give the IUPAC name for each compound.
A:
Q: Identify the conjugate acid-base pairs in the following reactions: a. H2PO4- + H2O ↔ H3O+ + HPO42-…
A:
Q: 21. Which substance will lower the melting point of ice the most? a. NaCI b. KNO3 c. Ca Cl2 d. KBr
A: formula to calculate the depression in the freezing point is delta T = i Kf m here i is the van't…
Q: 0.250 mol of N2 is placed into a 0.750 L container. What is the density of the gas? 84.1 g/L 4.67…
A:
Q: CHO Cro3, H2SO4 (excess) ČH2OH HOʻHa Ph 1) O3, 2) Zn, H20 3) NaOH, H20 heat, (dehydration)
A:
Q: Calculate the molar concentration of Ba2+ in saturated BaC2O4 solution. The pH was kept at 4.5. Ksp…
A: Given: pH = 4.5 Ksp of BaC2O4 = 1.0x10-6 Ka1 of H2C2O4 = 5.4x10-2 Ka2 of H2C2O4 = 5.42x10-5
Q: In each of the molecules drawn below one chemical bond is colored red. Decide whether this bond is…
A: if there is an appreciable electronegativity difference between the combining atoms of a bond , then…
Q: several Ph. steps Br OH Please explain the mechanism and steps of the synthesis. Thank you!
A:
Q: (a) the fission reaction of uranium-235 with a neutron to produce xenon-144 and strontium-90 chemPad…
A:
Q: 3. Fill the boxes with the missing reactants, reagents, or the major product(s) expected from each…
A:
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A:
Q: The concentration of CO- in a saturated solution of CuCO, is 1.60x10- mol/L at a certain…
A: * Solubility product is the product of molar concentrations of ion present in solution, when ion…
Q: O CHEMICAL REACTIONS Using the Avogadro Number An aluminum atom has a mass of 4.48 × 10 -23 g and a…
A: Answer 1. 27.0 g Answer 2. 1.85 × 105 moles
Q: NH2 NH2 NH2 a.
A:
Q: What is the volume, in milliliters, of each of the following diluted solutions? A 1.2 MM HClHCl…
A:
Q: What is the [H3O+] is a solution that has the pH of 3.400?
A: Given: pH of the solution = 3.400
Q: is a reactant-favored process what is true about this reaction? a. reactants predominate over…
A: At equilibrium, rate of forward reaction is equal to rate of Backward reaction. If reaction is…
Q: Consider the following reaction: N2(g) + 3H2(g) ⇄ 2NH3(g) A) Determine ∆Hrxn˚ and ∆Srxn˚ using the…
A:
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Entropy is the degree of disorderness. Increasing order of entropy, solid < liquid <…
Q: Be sure to answer all parts. A freshly isolated sample of 90Y was found to have an activity of 2.9 x…
A:
Q: Give the IUPAC name for each compound.
A:
Q: 1) Upon dissolving 90.0g of KNO3 in 100g of water at room temperature (20° to 25°C), a precipitate…
A:
Q: Part A How much concentrated solution would it take to prepare 2.95 L of 0.585 M HCl upon dilution…
A:
Q: Draw a voltaic cell for the given reaction (please include anode, cathode, and flow of electrons)…
A:
Q: Na" "SCH3 CH,CN Br HCI CH,OH in
A: Favorable conditions for SN1: 1) Strong nucleophile 2) Aprotic solvent Favorable conditions for SN2:…
Q: OH OEt H. chemoselectivity
A: ->LiBH4 is a reducing agent which can selectively reduce ester and lactones to the Alcohols.
Q: Use your solubility table in the back of the lab manual to complete the following tables. Sr2+ Fe3+…
A: We prepeared chart on the basis of water solublity and by the help of solublity chart. The prepeard…
Q: What is the IUPAC name of the following compound? ball & stick v + labels
A:
Q: Calculate the solubility at 25 °C of Zn (OH), in pure water and in a 0.0100M ZnSO, solution. You'lIl…
A: From alesk data, Ksp of Zn(OH)2=3.0×10-17 Molar mass of Zn(OH)2=99.424 g mol-1
Q: CH, H20 H2SO4 H,C- -CH3 H,C
A:
Q: The activation energy for the gas phase isomerization of dimethyl maleate is 111 kJ.…
A: Activation energy =111 KJ =111000 J T1=678K K1=3.77×10-4 K2=3.08×10-3 T2=?
Q: Predict the major, organic product for the following reaction sequence. Be sure your answer accounts…
A: In the given reaction here we have to take care about the stereochemistry of the product species.
Q: A Zn wire and Ag/AgCl reference electrode saturated with KCI (E = 0.197 V) are placed into a…
A:
Q: Explain the following trend in the absorptions corresponding to the most energetic C–H stretching…
A: Note : Absorption frequency is proportional to bond strength. Alkyne C-H bond strength is more than…
Q: Name it とセーBい
A: Naming is Done according To CIP rules in which heavier atom given more priority.
Q: 2- The concentration of SO, in a saturated solution of Hg2SO4 is 5.54x10-3 mol/L at a certain…
A: Given- Concentration of SO42- = 5.54 × 10-3 Mole/L Ksp of the compound=?
Q: A 0.56g sample of gas is contained in 250.0 mL cylinder. It's pressure is 983 mmHg and it's…
A:
Q: A 50.00-mL aliquot of 0.05000 M NaOH is titrated with 0.1000 M HCl. Calculate the pH of the solution…
A:
Q: delta S° for a reaction can be calculated from the following equation. a. S° = (moles…
A: Entropy is a measure of the randomness of the system. The entropy change in a chemical reaction can…
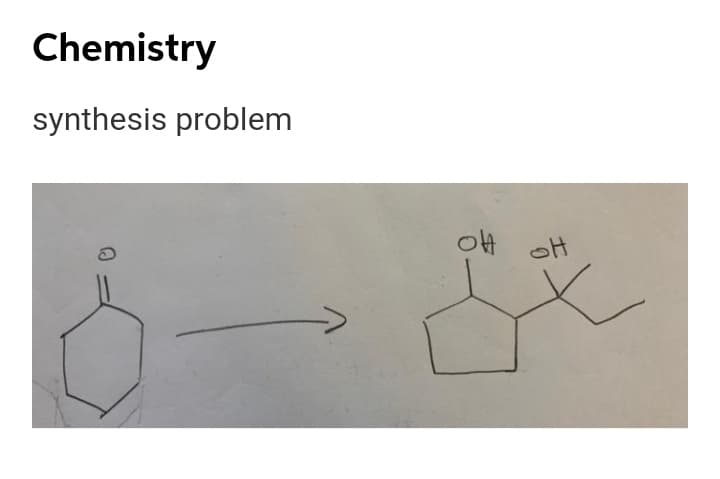
Step by step
Solved in 2 steps with 2 images

- Write the IUPAC name of the following compounds. Don’t use CIS/TRANS or E/Z in naming the compounds. NOTE: needs answers immediately. Will send a good rate right away as well.Please help me understand this practice proIn laboratory, Dr. Nur found out that the hydrolysis of nitriles (R—CN) when heated under reflux with dilute sulfuric acid yielded a major compound A. When compound A reacts with element N it showed that two products are produced (salt and gas). Meanwhile, the reaction of compound A with methanol yielded aromatic scent of compound B. She also noticed two products (compounds C and D) were obtained when she react compound B with reagent Lithium Aluminium Hydride (LiAlH4). Dr. Nur was observed compound C can be prepared naturally in the presence of enzymes in yeast through anaerobic conditions. Further investigation showed when compounds C and D can react with acidified potassium permanganate will produce compound (compounds E and F) with similar functional group. Dr. Nur also discovered compound E will produce silver mirror when react with reagent M and vice versa from compound F. Identify the possible expanded structure of compound A, B, C, D, E and F. Predict the…

