Q: MASS % of Base Peak IR 100 50 Transmittance 100 S 50 4000 'H NMR 300 MHz 7.0 60 6.5 70 6.0 PC/DEPT…
A: Some basic spectroscopic methods are used to confirm the presence of organic compound- 1- IR…
Q: 3. Given: NaCl 1 NiCl₂ + 2 NaOH → 1 Ni(OH)₂ + 2 A student combines 2.44 g of NiCl₂ with 6.44 g NaOH.…
A:
Q: QUESTION 3 Correctly name the following compound. Br H₂C NO₂
A: We have write the IUPAC name of the given compound
Q: For a reaction below at 600 K, the equilibrium constant is Kp is 11.5. Suppose that 2.450 g of PC15…
A:
Q: Calculate the standard cell potential of a voltaic cell that uses the half-cell reactions given…
A: Given that - Zn ----> Zn2+ + 2e- VZn0 = -0.763 V Cu2+ + 2e- ------> Cu VCu0 = +0.340…
Q: Lithium metal has a threshold frequency of 6.94 x 10¹4 s with a wavelength of 129 nm strikes a…
A:
Q: 4. What is the expected product of the following reaction? A) OCH 3 B) OH CH 3 C) CH3OH H+ OCH 3 D)…
A: Here protonation occur at 3 position because the generated carbocation is stabilized by 2p-2p…
Q: Consider a molecule with the molecular formula C5H8O. Draw the structure of the molecule based on…
A: The structure for the given molecular formula C5H8O is given below
Q: e. exothermic, because increasing temperature decreases the equilibrium constants, and more products…
A: The value of equilibrium constant indicates towards the extent to which the reaction has occurred.…
Q: Calculate the enthalpy of combustion (kJ/mol) of cetane (C16H34), an analogous component in diesel…
A:
Q: Acetic acid partially dissociates according to the reaction below. HC₂H3O2 + H₂O(1) H3O+ + C₂H30₂ ⇒…
A:
Q: What is the maximum number of electrons within an atom that can have the quantum numbers n = 4, l =…
A:
Q: Consider a molecule with the molecular formula C7H8O2 Draw the structure of the molecule based on…
A: In this problem we need to figure out the correct structure of the compound with molecular formula…
Q: 3.1 Propose a detailed mechanism for the reaction below: AICI 3 CI
A:
Q: A sample of element X has two naturally occurring isotopes. The isotope X-45 makes up 35.8% of the…
A:
Q: Calculate accurately using the equation w = sqrt(k/mu) the expected ratio ω(OD str.)/ ω(OH str.) for…
A:
Q: 5) A certain compound has the following percent composition: 57.1% C, 4.8% H, and 38.1 % O. If the…
A:
Q: c) Suggest and explain in detail one heat treatment procedure that can strengthen or harden the…
A: A metal when it is hardend,it is heated to a point where the element transform into a solution.
Q: Write the lewis electron dow structure of Magnesium Iodide. Determine also whether Magnesium Iodide…
A: Lewis electron dot structure is representation of valence shell electrons of an atom, molecule or…
Q: sider a chem reacti R has two states separated by 2.0x 10-22 J. P has a doubly degenerate level that…
A: The partition function of a system, Q, measures the possibility of a system occupying a state i. A…
Q: 5. Using pictures and words, explain why triflate, CF3SO3, is such a good leaving group.
A: To explain why triflate, CF3SO3- is such a good leaving group using pictures and words.
Q: Considering the C-NMR below, choose the compound from the list provided that gave rise to the…
A: The carbons with different environment will give different signal in C-13 spectrum. This shows the…
Q: How many of the atoms/ions have the incorrect configuration listed next to it. A. Be: [He]2s² B.…
A: Here we are required to find the number of atoms having correct the configuration.
Q: Question1 12 UI 12 Provide the correct IUPAC name for the compound shown here. SH
A: For IUPAC naming select the functional group & principle chain i.e longest possible carbon…
Q: For the reversible reaction PCI5 PCl3 + Cl₂, the equilibrium constant Kc is 1.00, If at the…
A: The reaction involved is PCl5 <------> PCl3 + Cl2 Kc of the reaction = 1.00 Volume of vessel =…
Q: 3. Which of the labelled hydrogens in vitamin C (L-ascorbic acid) is the most acidic? Explain your…
A: An acidic hydrogen is a hydrogen that has a tendency to leave easily as H+ ion.
Q: Find the [H+] concentration when it has a Ph of 12.2
A:
Q: Draw a valence-bond (balloon-animal) orbital picture of an ethyl cation. With reference to your…
A:
Q: 3. List two major sources of error associated with pH measurements.
A: pH = -log[H+] pH is defined as a type of scale which is used to measure the acidity and basicity of…
Q: 7. In potentiometric measurement, any change in cell potential is due to a change in the potential…
A: Given- Potentiometric titration statement To find- the statement is true or false
Q: Propose a detailed mechanism for the reaction below. لعلام , AICI3
A:
Q: True or False Metallic mercury is gray in nature.
A: Given statement is : Metallic mercury is gray in nature? Predict whether the given statement is…
Q: Examine the diagram below: In Energy I Which of the following statements is false? Reaction…
A: Given diagram is :
Q: Examine the galvanic cell below. Ni Volts 1.0 M t 1.0 M Ni (NO₂)₂ KNO3 † 1.0 M AgNO₂ The reaction…
A: Given, Galvanic cell:
Q: At 60 °C, Kw for water is 1.0 × 10¹³. At 60 °C, a weak acid has a Ka of 1.3 x 10-4. What is the Kb…
A: Acid can be defined as a substance that give hydrogen ions in an aqueous solution.
Q: For the equilibrium reaction N2(g) + 3H2(g) « 2NH3(g) DH = -92 kJ, Describe how the equilibrium…
A:
Q: 0= H₂C CH3 •CH3 cro3 H₂504, H₂0
A: Given incomplete reaction is : Give the product of the reaction ?
Q: 1.1 Provide IUPAC names of the following aromatic compounds: OH Br Br 6
A: IUPAC is International Union of Pure and Applied Chemistry. This nomenclature is accepted all over…
Q: fox H₂C CH3 · CH3. NaBH4 20
A: This reaction is an example of reduction reaction by which we get alcohol end product from given…
Q: 2. For each pair of molecules, circle the term―enantiomers, diastereomers, constitutional isomers,…
A:
Q: H3C ОН CH3 -CH3 CH3
A:
Q: Determine the moler solubility (s) of in a buffered solution with Ag₂ C03 a pH of 3.70 using the…
A: Given the pH of the solution = 3.70 => pOH = 14 - pH = 14 - 3.70 = 10.30 => [OH-(aq)] =…
Q: 10. The pH response pf glass electrode is limited entirely to the area of special glass membrane…
A: A question based on electrochemical cells that is to be accomplished.
Q: 2.2 Draw all the important resonance contributors for the arenium ions formed from each of these…
A:
Q: True or False Arsenic sulfide is basic in nature.
A: To answer: Arsenic sulfide is basic in nature.True or False
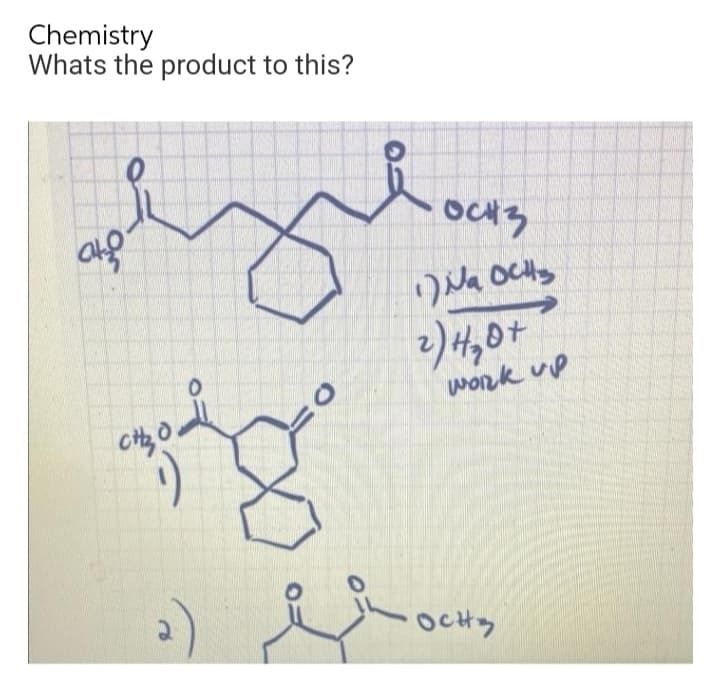
Q: OEt OEt Diethyl heptanedioate (a 1,7-diester) 2007 Thon Higher Education 1. Na OEt, ethanol 2. H30+…
A: The given reaction is an example of Dieckmann condensation, which is an intramolecular condensation…
Q: 1. Boiling temperatures for chlorobenzene are 283.2 K at 5332 Pa and 405.4 K at normal pressure…
A:
Q: Which does not belong to the group?* 3CuS + 2NO₃¹⁻ + 8H₃O ⇌ 3Cu²⁺ + 2NO +3S + 12H₂O 3HgS + 2NO₃¹⁻…
A:
Q: For the equilibrium reaction N2(g) + 3H2(g) « 2NH3(g) DH = -92 kJ, Describe how the equilibrium…
A:
Q: what is the na-me, 1/2, 01 12 = day:
A:
Step by step
Solved in 2 steps with 2 images

- SCE // PO43- (aq) , Ag3PO4 (saturated) / Ag h a s a n I n d i c a t o r e l e c t r o d e t h a t i s a c h o o s e : l i q u i d m e m b r an e e l e ct r o d e m et a l l i c e l e c tr o d e of t he s e co n d k in d g l a ss el e c t ro d e m e t a l li c el e c t ro d e of t h e f i r st k i n dWrite down the summary of the selection Rules for Pericyclic reactions.Calculate ΔrG° for the following reaction at 387 K. HCN(g) + 2H2(g) → CH3NH2(g) ΔrH°= -158.0kJmol-1; ΔrS°= -219.9JK-1 mol-1
- Calculate the Keq for the following reaction, given that Eo Cd2+/Cd = -0.403 V and Eo Zn2+/Zn = -0.763. Overall rxn: Cd2+ + Zn <-> Zn2+ + CdI get using the Arrhenius equation but even after calculating the e-[82.64 KJ/mol)/(8.314 J/mol.K × 346.63 K)] it doesn't equal up to 3.518× 10-13 s-1. Where did it come from if it didn't from from just that?What is the standard free energy change for the ionization of ethanolamine, if its kb is 3.20e-5.
- Calculate the standard free energy of the following reactionsat 25°C, using standard free energies of formation. a. C2H4(g) + 3O2(g) ----> 2CO2(g) + 2H2O(l )b. CaCO3(s) + 2H+(aq) ----> Ca2+(aq) + H2O(l ) + CO2(g)Which of the following reaction coordinate diagrams represents SN1 and E1 reactions? A B C DCalculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4
- Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?The deltaHs of cobalt (II) nitrate is +47.7 kJ/mol. What is the deltaHf of solid cobalt (II) nitrate? deltaHf(NO3-(aq)) = -205.00 kJ/mol deltaHf(Co2+(aq)) = -58.2 kJ/molinvolvedinthereaction? 4.Theenthalpyforthereactionbelowis-25kJ: 2A-A+B=B→2A-B-A a.Giventhefollowingbondenergies,findthemissingvalue. b. Sketch a potential energy diagram for this reaction:

