Q: Predict the major products of the following organic reaction: lift ? Some important notes: • Draw…
A: Diels-Alder reaction is a (4+2) cycloaddition reaction between electron rich species called diene…
Q: Match the IR spectra with its corresponding structure and explain reasoning.
A: IR spectroscopy is mainly used for the identification of functional groups present in organic…
Q: D Question 4 Consider an object that is in the shape of a cube with a mass of 45.8 grams. If the…
A: Answer:Density of an object is the ratio of its mass and volume as shown below:Here:
Q: During this course you will use a basteriallysis buffer One of the ingredients of his buffer as…
A: Triton X-100, also known as octylphenol ethoxylate or polyethylene glycol octylphenyl ether, is a…
Q: Draw the organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as…
A: Bronsted–Lowry acid base theory : An acid is a proton (H⁺) donor, and a base is a proton acceptor.…
Q: A person weighs 150.0 lb, and the correct dosage of a drug is given as 1.50 mg per kilogram of body…
A: Note: 2.20 lb = 1 kgWeight of a person = 150.0 lb = (150.0/2.20) kg = 68.18 kgCorrect drug dosage =…
Q: Propose an efficient synthesis for the following transformation: The transformation above can be…
A: To carry out the transformation detailed stepwise reaction pathway is given below
Q: The extraction of aluminum metal from the aluminum hydroxide in bauxite ore by the Hall-Héroult…
A:
Q: ▼ Part C Enter the formula for the compound calcium oxide. Express your answer as a chemical…
A: We have to write the systematic names of the given inorganic compounds
Q: sp Calculate the solubility of PbBr, in 0.10 M NaBr. The K of PbBr, is 4.6 X 10% at 25 °C. (Answer:…
A:
Q: Chemical energy is released or absorbed from reactions in various forms. The most easily measurable…
A: Given:∆fH°(A) = -225kJ/mol∆fH°(B) = -393 kJ/mol∆fH°(C) = 209 kJ/mol∆fH°(D) = -491 kJ/mol
Q: مسلم NaOME, HOMe
A: An enolate is an organic ion or molecule that contains a negatively charged oxygen atom bonded to a…
Q: For the reaction of A B, the Keq at 25°C is 0.0206 and the Keq at 50°C is 52.361. What is the delta…
A: Dear student since you have posted multiple questions, As per BARTLEBY QnA guidelines we will solve…
Q: Part 2) Accurate Solutions Creating a conductivity calibration curve 1. Using the dried NaCl from…
A: The simple concept for calculation of ppm concentration of solids has a general equality formula:1…
Q: Q1: It has been planned to use Thorium fuel (Th232) in a nuclear power station instead of Uranium…
A:
Q: 3. Propose synthesis for the following molecules st a. CH₂OH b. C.
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts of…
Q: The structure of a unit cell of an oxide of niobium is depicted here. Niobiums are dark and oxygens…
A: A cube has 8 corners, 6 faces and 12 edges.An atom that is located at the corner of a unit cell is…
Q: The pH of a given solution at 25 °C is 10.25. What is the pOH of the solution at the same…
A: The relationship between pH and pOH of a solution at :pH + pOH = 14
Q: One of the following reactions is redox (oxidation-reduction). Determine which is redox and why.…
A: The chemical reactions in which both oxidation and redox reactions take place simultaneously are…
Q: Which of the following sets of quantum numbers are NOT allowed? Choose one or more: 0 n=2, l=0, me…
A: Quantum numbers are a set of four parameters that describe the properties and characteristics of…
Q: 3. What starting material would be necessary to prepare the following compound 00 by the Diels-Alder…
A: An electrocyclic reaction, which involves cycloaddition of electrons of the conjugated diene and…
Q: QUESTION 14 Draw the dot structure for SC2 and state the type of bonds on the central atom. 2 single…
A:
Q: 100 4000 4000 A 3371 2971 3299 1000 2956 2997 3000 B BAVENUMBER 2132 2000 1500 1500 C 1000 1000 D nn
A: IR spectroscopy is used to determine the functional groups present in the sample. Different…
Q: What is the chemical shift in ppm of a carbon that resonates at 1325 Hz on a 250 MHz instrument?
A:
Q: Learning Goal: To understand the relation between the strength of an acid or a base and its pK and…
A: pH = 3Concentration = 0.083MpKa =?
Q: 12. Glycerin, C3H8O3, is a nonvolatile liquid. What is the vapor pressure of a solution made by…
A: The liquid molecules tend to convert into the vapour phase. In a closed container, the pressure of…
Q: 1. How many grams of Zn2+ are present in 2.11 grams of zinc iodide? grams Zn2+. 2. How many grams…
A: 1:Mass of zinc iodide= 2.11 gWe have to find the grams of Zn2+present2:Mass of Zn2+ = 4.84 gWe…
Q: Quantum numbers arise naturally from the mathematics used to describe the possible states of an…
A:
Q: A student diluted 13.6 mL of blue dye solution to 20.0 mL using distilled water. The concentration…
A: As we know ,Moles the solute remains same on dilution but volume of the solution increases.Dilution…
Q: 9. A modified Ringer's Irrigation has the following formula: 8.6 g 0.3 g 0.33 g Sodium chloride…
A: Answer :---- Here, we have to calculate the specific gravity of the irrigation solution, we need to…
Q: Provide the correct systematic name for the compound shown here. CH3 CH₂CH3
A:
Q: 1. Draw the products (with arrow formalism) OH Br Indicate the direction of equilibrium + Na C…
A: An organic reaction or rearrangement results in the conversion of one compound to another. A…
Q: According to the following reaction, how many grams of hydrogen gas will be formed upon the complete…
A: Given:Mass of hydrobromic acid = 27.5 g Calculate the mass of hydrogen gasThe balanced chemical…
Q: 1.30. Determine the expressions for the following, assuming that the ideal gas law holds. an (27),…
A: Ideal Gas equation is given by:PV = nRTwhere, P is pressureV is Volumen is number of milesR is…
Q: Draw the major product of this elimination. Consider regiochemistry and stereochemistry. Ignore…
A: This reaction is the E1 elimination reaction.At first presence of acid, protonation occurs at the…
Q: Problem 11: Determine the molarity and normality of 15% sulfuric acid (H₂SO4): a. By mass b. By…
A: By mass:15% sulfuric acid means that 15 g of H2SO4 is dissolved in 100 g of solution.The molar mass…
Q: (2) 1a. Fix the following structure which is a neutral molecule. You cannot add atoms. H H-C-C-N H…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: List four Physical factors that affect the rate of diffusion and how they will affect it. 2.What…
A: 1. Answer :---- Four physical factors that affect the rate of diffusion are :-Temperature :- Here,…
Q: 1- Draw the major product(s) for the following reactions. + HCI 40°C HCI 0°C CI C _A
A: We know alkenes due to the pi bond are electron-rich and thus can act as a good source of…
Q: Which of the curly arrows are incorrectly drawn in the following reaction mechanism? Br W W and Z O…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: Draw the two possible products produced in this E2 elimination. Ignore any inorganic byproducts.
A: In E2 elimination reaction, the abstraction of acidic proton by the base and loss of leaving group…
Q: Which of the listed molecule will react as an acid (will loose the most acidic hydrogen) if mixed…
A: It is asked to find the acid that reacts with NaOH.An acid is a substance that loses an ionizing…
Q: Explain a boiling point analysis for Sodium Propionate. Draw the lewis structure for the compound…
A:
Q: Draw the Newman projection so that it corresponds to the molecule and conformation shown when viewed…
A:
Q: When a particular substance crystallizes, it forms a body-centered cubic unit cell. It has a density…
A: Density of cubic unit cell = 3.87 g cm-3Unit cell edge = 475 pm = 4.75 X 10-10 meter = 4.75 X 10-8…
Q: 2. Name the following aromatic compounds: NH₂ HO H
A:
Q: Identify the condensed formula of the following structure: XX CI (CH3)2CHCHCICH(CH3)2…
A: The given structure is shown belowWe have to give the condensed formula of the given structure.
Q: Use vapor pressure. A student leaving campus for spring break wants to make sure the air in his dorm…
A: Given the temperature of room, T = 22 oC + 273.15 = 295.15 KThe dimension of the room = 3.41 m ×…
Q: The constant volume calorimeter is used to measure the heat of combustion of a reaction. Constant…
A: A constant volume calorimeter is a device used to measure the heat of combustion of a reaction. It…
Q: The metal alloy such as the silver-colored hubcap cover of a carwheel is an example of heterogenous…
A: Homogeneous mixture: Homogeneous mixture contains one phase and has a uniform composition throughout…
Give detailed Solution with explanation needed of all...don't give Handwritten answer
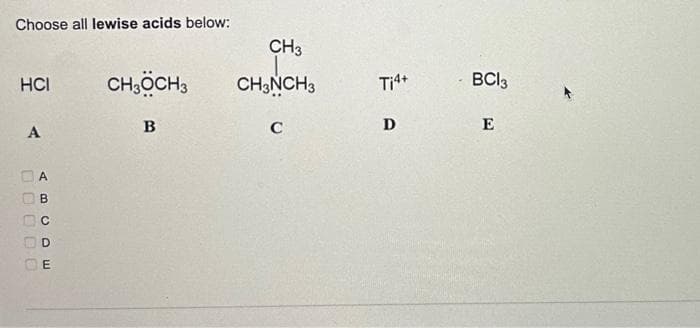
Step by step
Solved in 3 steps

- Arrange the following in increasing order of basic strength :C6H5NH2, C6H5NHCH3, C6H5N(CH3)2What steps would you need to take to calculate the pKa1 and pKa2 of something? For example: hydrosulfuric acid H2S(aq) (-38.6 ΔH^0 f kJ mol^-1).1. Which of the ff. groups is an electron donating group by resonance? A. COOCH3 B. COCH3 C. N(CH3) 3+ D. NHCH3 2. Looking at the diagram. Rank the ff. hydrogen atoms in the structure below based on the order of INCREASING acidity A. H4 < H3 < H1 < H2 B. H1 < H2 < H3 < H4 C. H2 < H3 < H1 < H4 D. H2 < H1 < H3 < H4
- What is the value of Kb for the formate anion, HCOO-? Ka(HCOOH) = 2.1 × 10-4HCN (aq) + SO4^2- (aq) -->2 part question please help! a) Which of the following is the strongest acid? a. CH3COCH3 (pKa = 19.3) b. CH3COOH (pKa = 4.76) c. CH3(CH2)2CH3 (pKa = 50) d. CH3CH2OH (pKa = 16.00) e. CH3(CH2)3C≡CH (pKa = 25) b) A carbon-carbon double bond contains the following bonding orbitals: a. one π and one π* b. two π c. one σ and one π d. one σ and one π* e. two σ
- Given the table below, what is the most probable identity of the acid?Acid KaHypochlorous acid 3.0 x 10⁻⁸Nitrous acid 7.1 x 10⁻⁴Hydrogen cyanide 6.2 x 10⁻10Propionic acid 1.3 x 10⁻⁵Which arrow (x, y, or z) points toward the most basic atom in the molecule to the right? Which structural feature plays the most significant role in influencing the relative acidities of the indicated protons? a. inductive effect b. size c. resonance d. hybridizationHow many of the following elements to form compounds with an extended Octect Pb Kr Si B
- Rank the following acids in order of strength. H2S, CHCl3, H2Se, CH2Ch2Draw and identity thr hybridized atoms (sp/sp2), draw reaonance structures/ polar bonds, and acidic hydrogens and basic positions for the compound Omeprazole.How is SO3 more acidic than MgO? SO3 + H2O = H2SO4 so it can give two (H+) but MgO + H2O = Mg(OH)2, wouldn't it therefore also be able to give 2 (H+) making it just as acidic as SO3? can someone explain?



