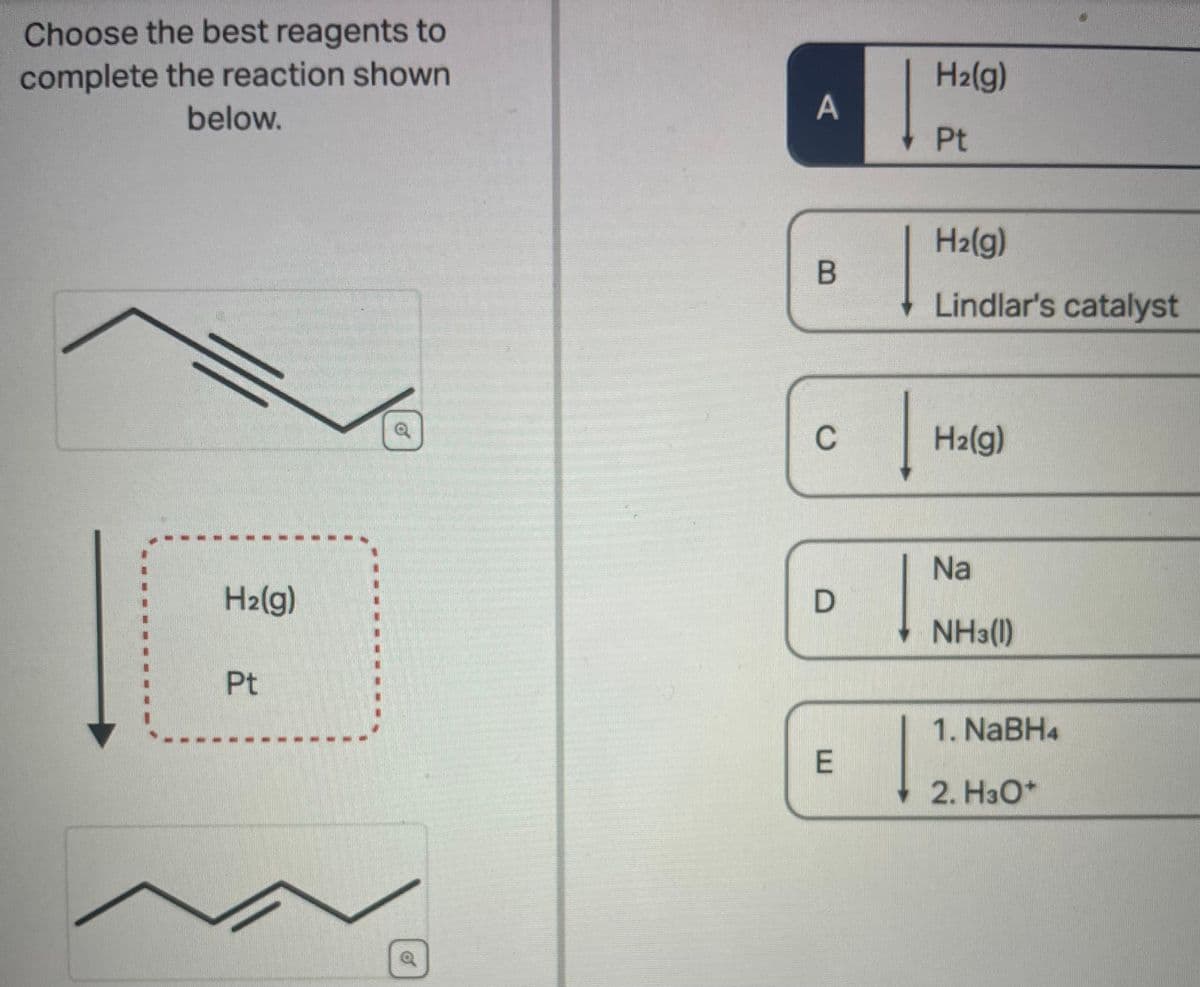
Choose the best reagents to complete the reaction shown below. H₂(g) Pt Q G A B C D E | H₂(g) Pt H₂(g) Lindlar's catalyst H₂(g) Na NH3(1) 1. NaBH4 2. H3O+
Q: Draw the major product of this reaction. Ignore any inorganic byproducts. HBr (1 equiv) H₂O₂
A:
Q: Problem 24: What is the major predicted product of the reaction shown? O ole D B 1.00 2. H₂O* ??? O…
A: The claisen condensation is a carbon - carbon bond forming reaction that occurs between two esters…
Q: Draw the final product formed in the reaction below. HO- O OH A
A: We have given the organic reaction and we have to find out the product of the reaction.Maleic acid…
Q: You have two containers at a pressure of 1.00 bar. One contains 1.00 mol of H₂O(g) at 450 K, and the…
A: Given,For H2O (g): mole(n)=1 mol, Po=1 bar P=20 bar, temperature(T)=450KFor H2O (l): mole(n)=1 mol,…
Q: Part B How much heat is required to convert 20.0 g of ethanol at -145 °C to the vapor phase at 78…
A:
Q: How many atoms are in 5.00 moles of O? O O 23 5.02 x 10 atoms 24 1.51 x 10 atoms O 3.01 × 1023 atoms…
A: The concept of the Avogardo number is used here. Because the moles of O are given and we are asked…
Q: A voltaic cell is constructed in which the anode is a Cd | Cd²+ half cell and the cathode is a Co |…
A:
Q: A chemistry graduate student is studying the rate of this reaction: 2H1 (g) - H₂ (g) +1₂ (g) She…
A: The objective of this question is to determine the rate law for the given reaction and calculate the…
Q: Select the product of the following reaction. и O O ОН ОН OH NaBH4, CH3OH
A: Given,The reaction:
Q: List 3 criteria that is important to consider when choosing a recrystallization solvent In a TLC…
A: Recrystallization is an experimental technique of separating pure compound from the solution…
Q: Question 7 Which of the following cells is most likely to be an example of a galvanic cell? A) Zn(s)…
A: Answer:-This question is answered by using the simple concept of Galvanic cell which is also called…
Q: Suppose a 500. mL flask is filled with 0.30 mol of Br₂, 1.2 mol of OCI, and 0.60 mol of BrCl. The…
A: The objective is the computation of equilibrium molarity of Br2, i.e., [Br2]. Equilibrium molarity…
Q: a mixture containing 0.750M HCl(g) and 0.330 M O2 (g) is allowed to come to equilibrium at 480C. At…
A: The equilibrium constant Kc - The equilibrium constant of concentration (denoted by Kc) of a…
Q: i mean how do i draw the structure of 2,3,8-trichloro-3,7-dimethyldecan-5-ol .
A: Identify the parent chain name and write carbon continuously based on that.Number the carbons and…
Q: Draw the product formed when the structure shown below undergoes solvolysis in CH3CH₂OH with heat.…
A: In this solvolysis reaction Alkyl halides reacts with ethanol to form an ether.Here's a…
Q: 10) Which of the following pairs of structures represent resonance structures? Please select any and…
A: Resonance structures share identical molecular formulas and atom connections. While the arrangement…
Q: 15. Which one of the following molecules is most unlikely to adopt the chair conformation? A)…
A: First of all made all molecules in chair form .try to put relative configuration in stable form as…
Q: 3. Ammonium ion (NH4*) is an acid that can dissociate to form ammonia (NH3) and H* in aqueous…
A:
Q: A galvanic (voltaic) cell consists of an inert platinum electrode in a solution containing 1.0 M…
A: The given cell is a voltaic cell or a galvanic cell. A galvanic cell is a cell that uses chemical…
Q: This dipeptide is designated as: *H3N-C- H3C-CH O Leu-Val Olle-Val Val-lle Yal-Leu CH3 18 C-N-C H3C…
A: When two amino acids are joined together, then elimination of water takes place and amide linkage…
Q: Which of following molecular formulas, once the BEST Lewis structure has been solved, will always…
A: Polarity results from the uneven partial charge distribution between various atoms in a compound.…
Q: Calculate the pH of a 5.0 x 10^-3 M KOH solution. A. 2.30 B. 12.70 C. 11.70 D. 1.30 E. 10.70
A: Answer:pOH tells about the strength of hydroxide ion in the solution and its value is equal to:Also,
Q: Please write the major organic product
A: In Micheal's addition reaction, a nucleophilic addition reaction takes place at the carbon-carbon…
Q: 4-hydroxycoumarin (s) and ammonium acetate (s) react to form 4-aminocoumarin (s) and water (l). What…
A:
Q: Enter your answer in the provided box. Enter your answer in scientific notation. A first-order…
A: Given:Rate constant (K) for first-order decomposition reaction = 0.00393 yr-1We have to determine…
Q: What will happen if a small amount of hydrohloric acid is added to 0.1 M solution of HF? A. The…
A: The objective of the question is to predict the change in the value of percent ionization.
Q: Draw the resonance structure indicated by the curved arrows. Assign formal charges. H H- HO: H H…
A: Resonance structures are the sets of Lewis structures that describe the delocalization of electrons…
Q: Identify the following electron transport intermediate. : O : H the superoxide free radical …
A: The correct response to the previous question is option D, specifically water. This is indicated by…
Q: 30. Give the major elimination product for the reactions below Br Br Br Br 2 NH₂ 2 OH
A: The reaction that involves the elimination of the leaving group along with the removal of hydrogen…
Q: QUESTION 2 The structure for 2-ethyl-1-butene contains how many hydrogen atoms? OA. 10 B. 11 O C. 12…
A: To determine the number of hydrogen atoms in the 2-ethyl-1-butene, first write the complete…
Q: Draw the organic product of the following reaction (either enantiomer). Copy the SMILES code…
A: When an alkene reacts with a halogen in the presence of H2O, a halohydrin will be formed.
Q: What is the Molarirt of 4.10 grams of magnesium chloride in 300 mL of solution
A:
Q: Consider the reaction. 4HCI(g) + O₂(g) 2H₂O(g) + 2Cl₂(g) Using the standard thermodynamic data in…
A: "Since you have posted multiple questions, we will provide the solution only to the first…
Q: How would you prepare 207 mL of 0.214 M NaCl solution using an available 1.90 M solution? Dilute…
A: V1 = 207 mLM1 = 0.214 MM2 = 1.90 MV2 = ?
Q: ܝ ܬ ܬ
A: Given are organic reactions. Given are pericyclic reactions.The name of given reactions is…
Q: The movie below shows some molecules in a tiny sample of a mixture of gases. 1 2 3 4 5 6 7 8 9 10 11…
A: In the movieGreen balls = Chlorine = ClWhite balls = Hydrogen = HTwo green balls attached = Cl2Two…
Q: After running various experiments, you determine that the mechanism for the following reaction is…
A: When a nucleophile attacks on an alkyl halide molecule and halide is substituted by nucleophile then…
Q: 5. The general formula for an alkene is a. CnH2n b. CnH2n-2 c. CnH2n+2 d. CnHn e. C₂nHn 6. Which of…
A: Alkenes and alkynes are two types of hydrocarbons, which are organic compounds composed of hydrogen…
Q: 2 C₂H6 (g) + 7 02 (g) →4 CO2 (g) + 6 H₂O (1) AHrxn-3121 kJ How many grams of C₂H6 are required in…
A: Given that2C2H6(g) + 7O2(g) ---> 4 CO2(g) + 6 H2O(l) ∆Hrxn = -3121 kJHow many grams of C2 are…
Q: In the titration of 35 mL of 0.2 M CH3NH2 (Kb=4.6 * 10^-4) w/ 0.2 M HCl, calculate the pH under the…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Complete the road map for the synthesis of a triglyceride of Lauric acid. ACI a) NaOH b) H3O+ ^g a)…
A: For the synthesis of triglycerides of lauric acid 3-chloroprop-1-ene reacts with sodium alkoxide of…
Q: I Br CI CI NaOEt NaOH NaOH t-BuOK
A: E2 reactions occur in one step , without involving any intermediate. Since, E2 reactions proceed…
Q: 4. Explain the reaction shown below. Include the following pieces in your explanation. a. Identify…
A: Given that, the reaction is:
Q: how many of the following elements can form compounds with an expanded octet P, Kr, As, B A) 2 B)4…
A: The list of the given elements is: Phosphorus (P), Krypton (Kr), Arsenic (As), and Boron (B). The…
Q: Draw full mechanism for below reaction? explain the stereochemistry. H2, Pd/C DA HA-HA HSPr Н SPr…
A: An alkene is electron-rich. But when it is attached to an electron-withdrawing group, it undergoes…
Q: 1) Based on the Material Reaction Scheme Safety Data Sheets (MSDS) for the compounds, which of the…
A: The given reaction is between anthracene and maleic anhydride to form an adduct. Toluene is the…
Q: (a) Rank each proton shown below in terms of relative chemical shift in the 'H NMR spectrum. (1 =…
A: Proton NMR spectroscopy is mainly used for the identification of the complete structure of an…
Q: 20. From what you know about alkenes and cis and trans isomers, explain the structure of the cis and…
A: The cis-trans isomerization is a type of stereoisomerism. The molecule which contains a double bond…
Q: Bq What is the specific activity in if 2.94 kg of an isotope emits 4.07 x 107 B particles per…
A:
Q: A 50.0 mg sample of an unknown radioactive substance was placed in storage and its mass measured…
A: Radioactive decay is a first order reaction. Half-life ( t1/2 ) formula for first order reaction or…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- In the Hell–Volhard–Zelinskii reaction, only a catalytic amount of PBr3 is necessary because of the equilibrium below. Review the mechanism for the reaction of a carboxylic acid with thionyl chloride and propose a mechanism for the equilibrium.Complete the following reactions ( DO NOT BALANCE )( all in aqueous medium) a ) KCl + ___ → _ppt__ + KNO3 b ) Cu (NO3)2 + ___ → _ppt____ + NaNO3 c ) ___ + ___ → CaCO3 + ___ d) ___ + ____ → _ppt__ + H2 (g) e) CaCO3 + HNO3 → f) BaSO4 + HNO3 →The Kc-value of the reaction 8A (g) + 4B (g) ⇌ 4A2B (g) is 20,263.What is the Kc-value of the reaction below?A2B (g) ⇌ 2A (g) + B (g) Report your answer in scientific notation with three sig figs. (For example, 0.001542 would be 1.54E-3)
- I just was wondering why its not 2e- for the second reaction and just e-2I– (aq) + H2O2 (aq) + 2H3O + (aq) → I2 (aq) + 4H2O (l) (slow) C6H8O6 (aq) + 2H2O (l) + I2 (aq) → C6H6O6 (aq) + 2H3O + (aq) + 2I– (aq) (very fast) I2 (aq) + I– (aq) ⇌ I – 3 (aq) I3- (aq) + starch → blue I3- · starch complex (aq) (fast) A different 0.100 L reaction mixture is prepared with initial concentrations of 1.0×10−2 M iodide I– , 2.0 × 10−3 M ascorbic acid C6H8O6 , and 0.135 M hydrogen peroxide H2O2 . If the first two reactions above procede at the same rate (they’re limited by the first one!), which of these reactants will be completely consumed first?Reagent is NOT (CH3)2CuLi. Which one works from the answer bank? iPrMgBr is (CH3)2CHMgBr
- Fill in Q,R,S,T,U and V with appropriate chemical reagants to make these reactions possible.A +2B + C → 2D + D Data and rates for the reaction are given below. (question 1.jpg) a) What is the reaction grade according to A, B and C? b) What is the value of R5 in terms of R1?5.9. In the polymerization of H2N(CH2 )10 COOH to form nylon-11, what is the molecular weight of the species that has the largest weight fraction in the reaction mixture at 99% conversion ? [Ans. 18,300]
- here are the two reactions for this worksheetLi+(aq) + e- ⇌ Li(s) -3.040Rb+ + e- ⇌ Rb (s) -2.98please help me, i will absolutely give a thumbs up asap, I really need help with this whole worksheet, i dont have any questions left on chegg, i would really appreciate your help, thank you in advance.If 0.70 g of FeCl3 is used to iodinate 8.42 g of benzene with sufficient ICl, what is the average number of reactions that each FeCl3 molecule must catalyze?Is the reaction [Co(CN)5]3- + Co(NH3)5(OH)]2+likely to proceed by an inner-sphere or out-sphere mechanism? Explain your answer.

